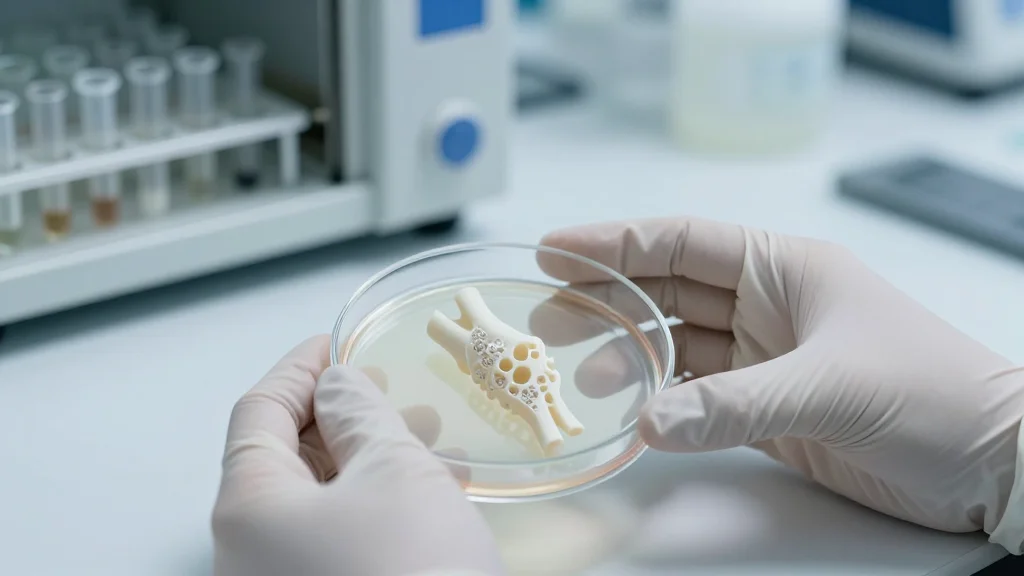
Living, 3D-Printed Biological Knee Replacement Advances to Preclinical Testing
Key Takeaways
- •NOVAKnee cleared for preclinical testing under ARPA‑H’s NITRO program.
- •Biodegradable 3D‑printed scaffold seeded with patient‑derived stem cells.
- •Implant aims to regenerate cartilage, extending joint lifespan beyond 20 years.
- •Could lower revision surgery rates, especially for patients under 60.
- •Commercialization planned via NOVAJoint Orthopedics, targeting FDA approval by 2028.
Pulse Analysis
Knee arthroplasty is one of the most common surgeries in the United States, with roughly 800,000 procedures performed annually and projections of two million by 2030. Conventional implants, built from metal and plastic, typically last 15 to 20 years before degradation or loosening forces patients into costly revision surgeries. Younger, active patients face especially high revision risks—up to 35% within five years—making the current technology a poor fit for a growing demographic that demands longevity and mobility.
The NOVAKnee platform, funded by the Advanced Research Projects Agency for Health (ARPA‑H), tackles these shortcomings by marrying 3‑D‑printed biodegradable polymers with autologous or induced pluripotent stem cells. The scaffold provides immediate load‑bearing strength, then gradually resorbs as seeded cells lay down native cartilage and bone, essentially growing a living joint in situ. After two years of prototype development, the team met all technical milestones, earning the first NITRO‑program approval for preclinical work. Engineering hurdles—balancing mechanical integrity with controlled degradation—were solved through novel polymer blends and patient‑specific geometry, positioning the implant for seamless integration into existing surgical workflows.
If preclinical trials confirm safety and efficacy, NOVAKnee could disrupt the orthopedic market by reducing the lifetime cost of joint care and expanding the eligible patient pool. The formation of NOVAJoint Orthopedics signals a clear path to commercialization, with a pre‑IND already filed and FDA approval targeted for 2028. Successful adoption would not only improve quality of life for millions suffering osteoarthritis but also generate substantial revenue streams for biotech firms and insurers willing to back regenerative solutions.
Living, 3D-printed biological knee replacement advances to preclinical testing
Comments
Want to join the conversation?