Key Takeaways
- •OP‑3136 selectively inhibits KAT6A, an epigenetic writer linked to MYC
- •Phase 1/2 trial combines OP‑3136 with SERDs like palazestrant
- •Preliminary efficacy data expected later this year
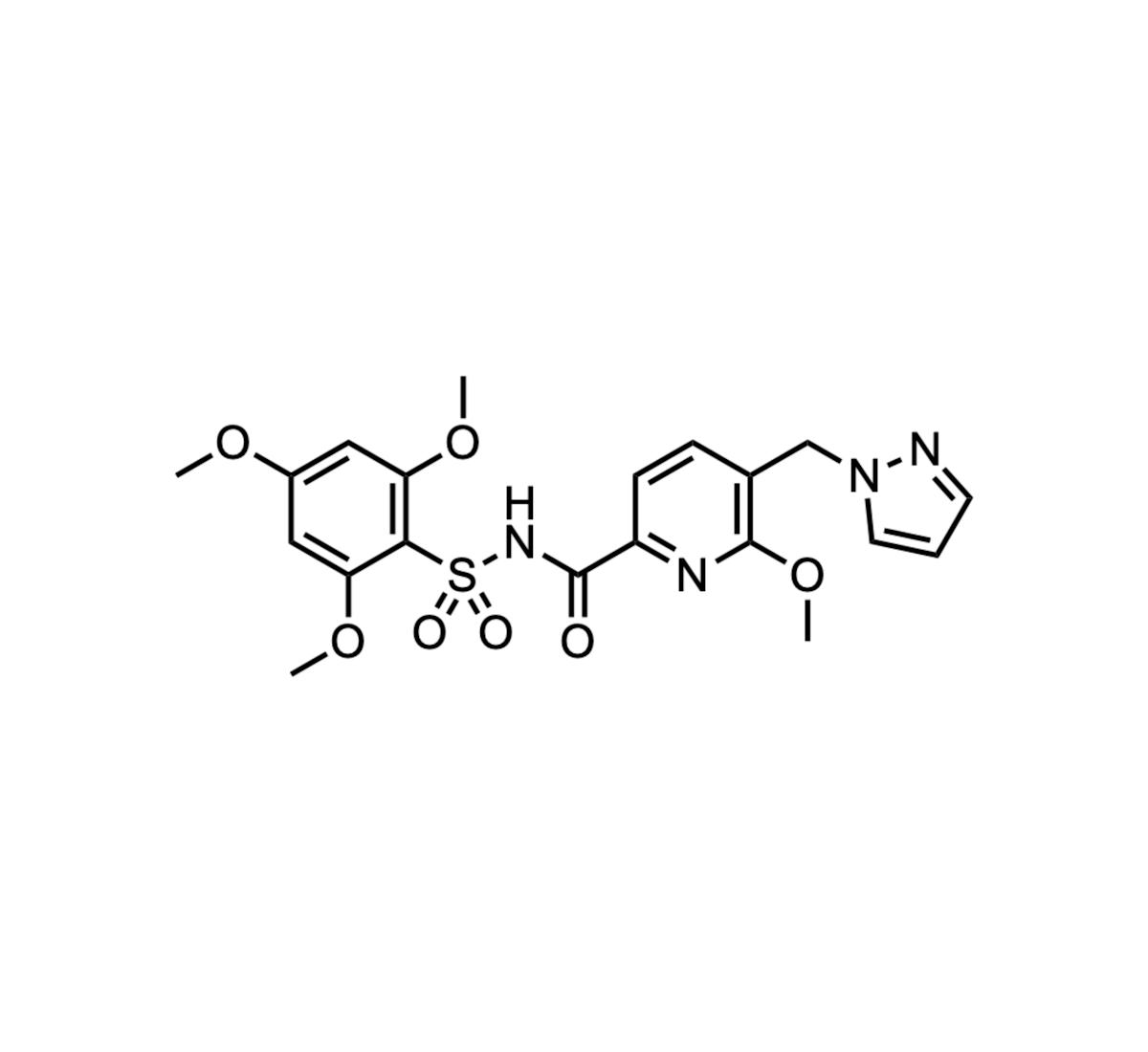
- •Acyl‑sulfonamide moiety mimics acetyl‑CoA pyrophosphate, enhancing target binding
- •KAT6A inhibition could address resistance mechanisms in hormone‑receptor‑positive breast cancer
Pulse Analysis
The lysine acetyltransferase KAT6A has risen to prominence as an epigenetic writer that fuels oncogenic transcription programs, notably by amplifying MYC and other growth drivers. By transferring acetyl groups from acetyl‑CoA to histone tails, KAT6A reshapes chromatin accessibility, creating a permissive environment for tumor proliferation. OP‑3136, Olema’s first‑in‑class KAT6A‑selective inhibitor, exploits an acyl‑sulfonamide scaffold that mimics the pyrophosphate of acetyl‑CoA, delivering high affinity and specificity while sparing related acetyltransferases. This mechanistic precision distinguishes it from broader epigenetic modulators.
Clinically, OP‑3136 entered a combined Phase 1/2 study targeting patients with advanced, hormone‑receptor‑positive breast cancer who have progressed on standard endocrine therapy. The trial pairs the inhibitor with selective estrogen receptor degraders (SERDs), including Olema’s own palazestrant (OP‑1250), to simultaneously shut down estrogen signaling and the downstream epigenetic support that drives resistance. Early safety signals appear manageable, and the sponsor anticipates first efficacy readouts later in 2026. If the combination demonstrates meaningful tumor shrinkage, it could set a new therapeutic benchmark for refractory disease.
From a market perspective, a successful KAT6A inhibitor would fill a notable gap in the breast‑cancer pipeline, where most approved agents target receptors or downstream kinases rather than chromatin regulators. Investors are watching Olema closely, as a breakthrough could unlock a multi‑billion‑dollar opportunity and stimulate partnerships with larger oncology players. Moreover, the acyl‑sulfonamide chemistry may be adaptable to other KAT family members, expanding the platform’s relevance beyond breast cancer. Regulatory agencies are likely to scrutinize the novel mechanism, but favorable data could accelerate the path to approval.
OP-3136
Comments
Want to join the conversation?