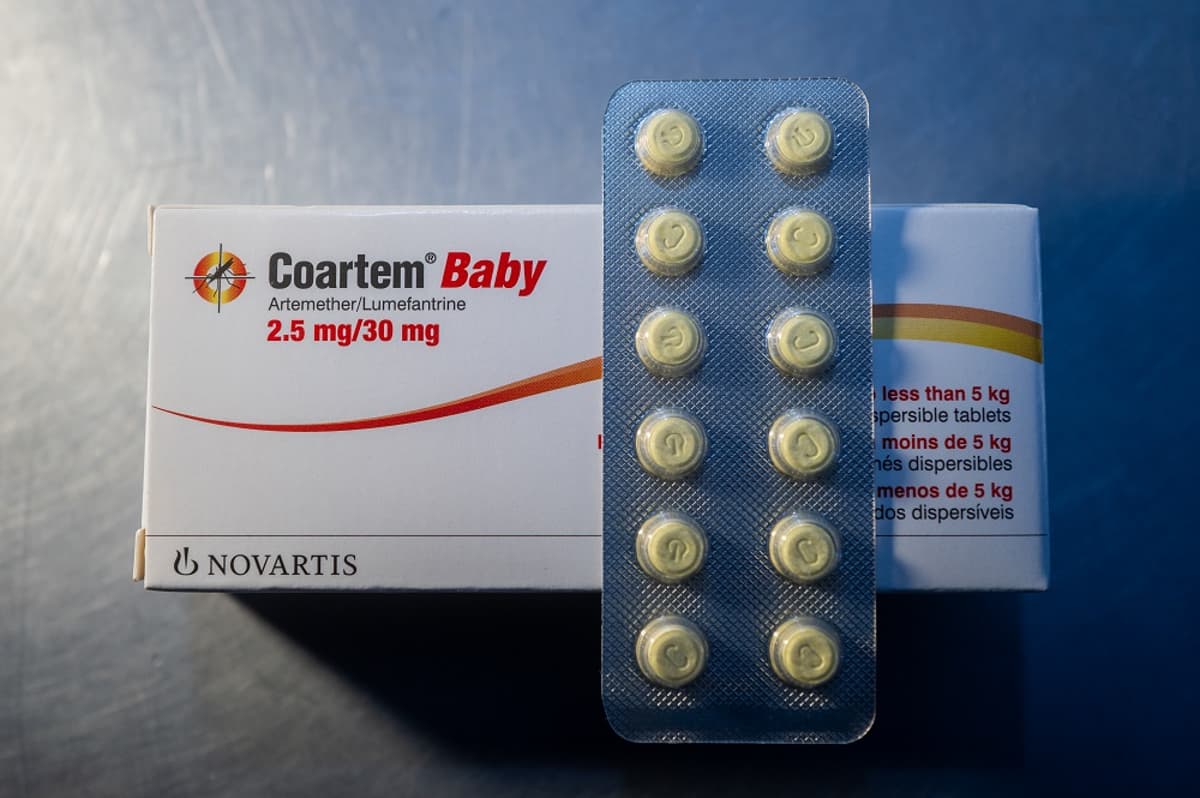
Novartis Antimalarial Clears Key WHO Review to Get the Combo Drug to Babies
Companies Mentioned
Why It Matters
WHO pre‑qualification unlocks access to a child‑friendly malaria treatment in low‑capacity countries, potentially reducing infant malaria mortality and drug‑resistance risks. The initiative also showcases a sustainable, not‑for‑profit model for essential medicines in underserved regions.
Key Takeaways
- •WHO pre‑qualification enables procurement in countries lacking regulatory capacity
- •Coartem Baby dissolves in liquid, sweet cherry flavor aids infant dosing
- •Formulation uses artemether‑lumefantrine to reduce resistance risk
- •Novartis supplies drug on not‑for‑profit basis to public‑sector buyers
- •Swissmedic approval and African reviews paved path for WHO clearance
Pulse Analysis
Malaria remains a leading cause of death among children under five in sub‑Saharan Africa, accounting for an estimated 200,000 fatalities annually. While artemisinin‑based combination therapies have transformed adult treatment, infants weighing 2‑5 kilograms have faced off‑label dosing, leading to suboptimal adherence and heightened side‑effect risk. The WHO’s pre‑qualification of Coartem Baby marks a pivotal shift, providing a quality‑assured, pediatric‑specific solution that can be rapidly adopted by ministries of health lacking strong drug‑evaluation frameworks.
Coartem Baby’s development was a joint effort between Novartis and the nonprofit Medicines for Malaria Venture. By reformulating artemether and lumefantrine into a sweet, cherry‑flavored liquid that dissolves in breast milk or water, the product simplifies administration and improves palatability for infants. The not‑for‑profit supply model ensures that public‑sector purchasers and donor‑funded programs can acquire the drug without price barriers, while Novartis recoups costs through volume‑based agreements rather than profit margins. This approach mirrors recent philanthropic strategies in global health, where manufacturers prioritize impact over revenue in high‑need markets.
The broader implications extend beyond immediate disease control. WHO pre‑qualification serves as a de‑facto endorsement for countries without mature regulatory agencies, accelerating market entry and encouraging local procurement. Successful rollout could stimulate further pediatric‑focused drug development, a historically neglected segment in infectious‑disease pipelines. Moreover, the liquid formulation may set a new standard for other antimalarial combinations, fostering a generation of child‑friendly therapeutics that align with WHO’s goal of eliminating malaria deaths by 2030.
Novartis Antimalarial Clears Key WHO Review to Get the Combo Drug to Babies
Comments
Want to join the conversation?
Loading comments...