Pliant Therapeutics Announces Presentation of Updated Data From the Phase 1 Trial of PLN-101095 in Patients with ICI-Refractory Solid Tumors at the 2026 AACR Annual Meeting
Companies Mentioned
Why It Matters
The data suggest PLN‑101095 could overcome checkpoint‑inhibitor resistance, addressing a high‑unmet need and potentially reshaping the therapeutic landscape for refractory solid tumors.
Key Takeaways
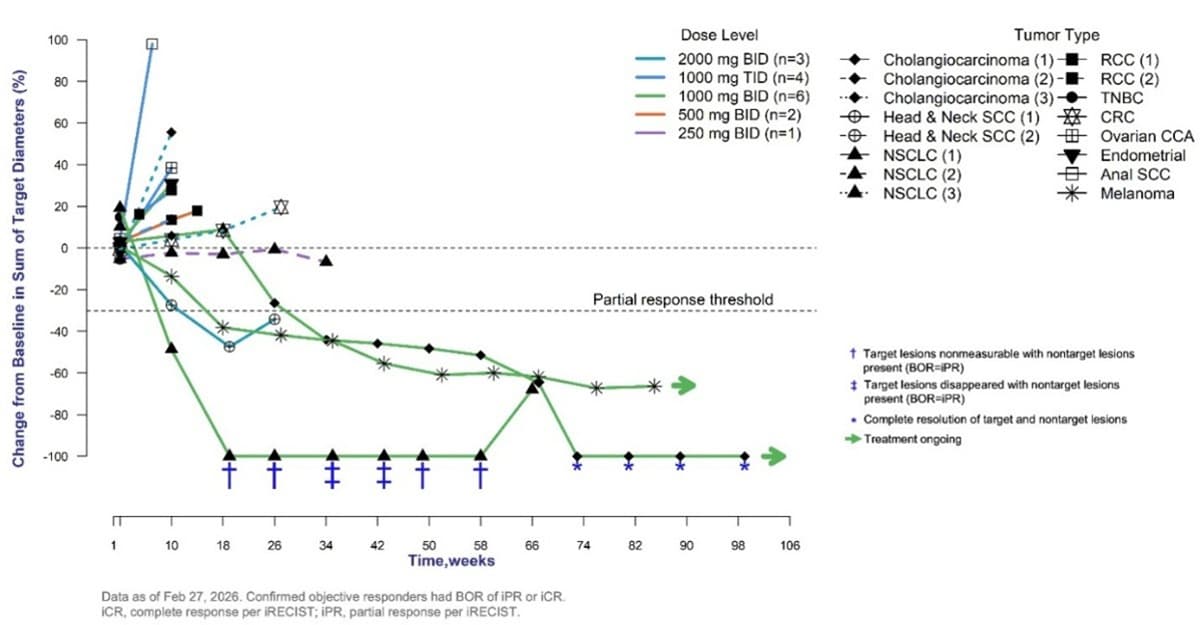
- •PLN‑101095 + pembrolizumab achieved 89% average tumor shrinkage.
- •Median treatment duration for responders reached 19 months.
- •IFN‑γ rose 4‑13× in responders, indicating biomarker potential.
- •One complete response observed across four tumor types.
- •Phase 1b expansion enrolling NSCLC, high‑TMB, and renal cell carcinoma.
Pulse Analysis
Immune‑checkpoint inhibitors have transformed oncology, yet a sizable subset of patients develop resistance driven by TGF‑β‑mediated immunosuppression. Pliant Therapeutics’ oral small‑molecule PLN‑101095 targets integrins αvβ8 and αvβ1, blocking the upstream activation of TGF‑β and restoring immune surveillance. By pairing this integrin blockade with pembrolizumab, the company aims to re‑sensitize tumors that have become refractory to standard checkpoint therapy, a strategy that could fill a critical gap in current treatment algorithms.
The Phase 1a/1b data presented at AACR reveal compelling early signals. Responders experienced an average 89% reduction in baseline tumor size and remained on therapy for a median of 19 months, far exceeding typical durations in heavily pre‑treated cohorts. Biomarker profiling showed dramatic 4‑ to 13‑fold spikes in plasma IFN‑γ and sustained PD‑L1 elevation, suggesting that IFN‑γ may serve as an early pharmacodynamic read‑out of effective TGF‑β inhibition. Safety was manageable, with only two discontinuations and mostly low‑grade rash, supporting the feasibility of continued dose escalation.
Looking ahead, Pliant has launched a Phase 1b indication‑expansion trial focusing on non‑small cell lung cancer, high‑tumor‑mutational‑burden tumors, and clear‑cell renal cell carcinoma. If these cohorts replicate the initial responses, PLN‑101095 could become a differentiated option in the crowded checkpoint‑inhibitor market, offering a novel mechanism to overcome resistance. Investors will be watching the upcoming 2027 interim readout closely, as successful validation could drive significant valuation uplift for the company and accelerate partnership or acquisition interest from larger oncology players.
Pliant Therapeutics Announces Presentation of Updated Data from the Phase 1 Trial of PLN-101095 in Patients with ICI-Refractory Solid Tumors at the 2026 AACR Annual Meeting
Comments
Want to join the conversation?
Loading comments...