Video•Mar 1, 2026
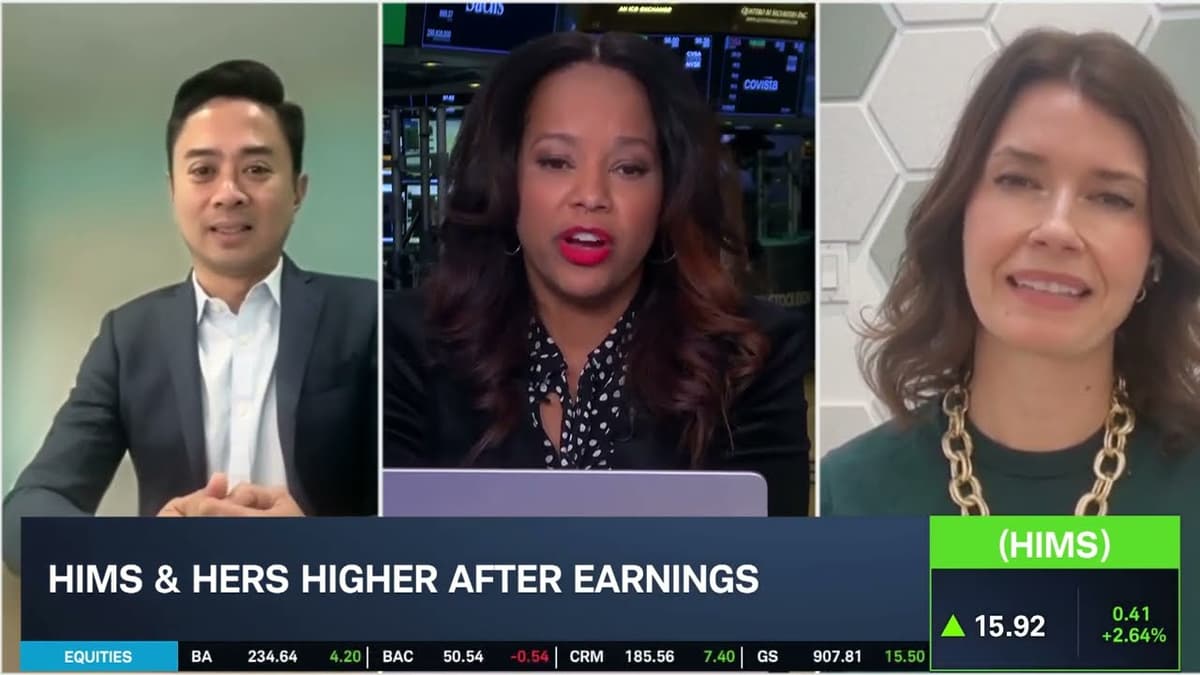
HIMS Core Business Faces Challenges in Guidance, GLP-1 Prices & NVO
Hims & Hers reported Q4 results after the bell, with mixed performance, drawing investor focus on its GLP‑1 program and forward guidance. The company posted EPS of $0.08, double expectations, but revenue of $617 million fell short of the $619 million forecast. For Q1, guidance of $600‑$625 million disappointed analysts who had modeled $654 million, reflecting pressure from GLP‑1 price erosion and regulatory scrutiny. Analysts Christine Short highlighted a 30%+ growth in the core sexual‑health and dermatology segments, noting that only a small fraction of the 2.5 million subscribers use GLP‑1. Brian Tanquilut reiterated a “hold” rating, citing the Novo Nordisk patent lawsuit and the fading GLP‑1 halo as near‑term valuation caps, while emphasizing the company’s shift toward a holistic, personalized‑medicine model. The market’s initial sell‑off has softened as investors reassess the pivot away from GLP‑1 dependence. However, ongoing legal liabilities and uncertain GLP‑1 pricing dynamics keep the stock’s upside limited, making execution of the broader telehealth strategy critical for long‑term growth.
By Schwab Network
Video•Feb 27, 2026
AI Insights in Pharma with Protai - Life Sciences Today Podcast Episode 50
In episode 50 of the Life Sciences Today podcast, host Danny Lieberman sits down with Carol Pesner, CTO and co‑founder of ProtAI, to discuss the company’s AI‑driven approach to drug discovery. ProtAI fuses structural proteomics with advanced artificial intelligence to...
By Healthcare IT Today

Video•Feb 27, 2026
LIVE: WHO Meeting on 2026-27 Northern Hemisphere Influenza Vaccine Composition
The World Health Organization convened an information meeting to finalize the composition of the 2026‑27 Northern Hemisphere influenza vaccine. Senior officials, including Dr. Chikui Hikawazu and Dr. Maria Van Kokov, highlighted the critical role of the Global Influenza Surveillance and...
By World Health Organization (WHO)

Video•Feb 26, 2026
Walk With Me: Joseph Wu, Cardiologist Studying Stem Cells and Heart Disease
Joseph Wu, a Stanford professor of medicine and radiology, leads the Stanford Cardiovascular Institute in developing patient‑specific cardiac cells derived from a person’s own blood. By reprogramming blood cells into pluripotent stem cells and then coaxing them to become beating...
By Stanford Medicine

Video•Feb 25, 2026
Malta as a Living Lab for Precision Oncology | MedTech World Middle East 2026
At MedTech World Middle East 2026, Prof. Christian Scerri unveiled a plan to turn Malta into a living lab for precision oncology, leveraging its single national health system, compact ecosystem, and EU‑aligned regulatory framework. He outlined a roadmap that includes...
By MedTech World

Video•Feb 25, 2026
GCC MedTech Boom: Market, Funding & Scaling | MedTech World Middle East 2026
At MedTech World Middle East 2026, a panel of GCC health leaders highlighted the region’s rapid shift from a pure healthcare consumer to a burgeoning hub for MedTech, biotech, and digital health innovation. National visions and transformation strategies are driving...
By MedTech World

Video•Feb 25, 2026
Citi Bearish On Dr Reddy's; CLSA Positive On HAL, Says Company Has Strongest Pipeline Of Orders
Citi’s research team issued a bearish stance on Dr Reddy’s Laboratories, citing concerns that the company’s generic version of semaglutide may not capture significant market share. In contrast, CLSA upgraded Hindustan Aeronautics Limited (HAL) to a preferred pick, highlighting its strongest...
By CNBC-TV18

Video•Feb 24, 2026
The Surprising Impact of Drug Price Negotiation on Clinical Trials
The podcast examines a new Health Affairs paper that tracks how the Inflation Reduction Act’s Medicare drug‑price negotiation provision has affected biopharmaceutical clinical‑trial activity. Dr. So Young Kang and co‑authors compare industry‑sponsored trial initiations from 2015‑2024, focusing on firms directly hit...
By Health Affairs

Video•Feb 24, 2026
The Conjugation Conundrum: The Realities of Conjugated LNP Manufacturing
The discussion centers on the emerging complexities of conjugated lipid nanoparticle (LNP) manufacturing, a shift from standard platform processes toward targeted RNA delivery. Sujit explains that adding a biologic ligand to LNPs forces manufacturers to revisit every process parameter—viscosity, shear...
By Life Science Connect

Video•Feb 24, 2026
The “Art” Of the Linker: Complexity, Biodegradability, and Scale in Active LNP R&D
The panel examined the emerging “active” delivery paradigm for lipid nanoparticle (LNP) therapeutics, focusing on the added layers of complexity introduced by ligand‑modified formulations and the need for robust R&D pipelines. Participants contrasted passive LNPs with active, ligand‑decorated versions, highlighting...
By Life Science Connect

Video•Feb 24, 2026
The Challenges Novel Lipids Pose For mRNA-LNP Manufacturing
Novel lipid chemistries are accelerating mRNA‑LNP innovation, but they also upend established manufacturing platforms. According to CMC consultant Sujit Jain, each new lipid class forces a fresh round of process development, demanding new impurity profiling strategies and vendor qualification. The...
By Life Science Connect

Video•Feb 24, 2026
Novel Lipid Chemistries & Their Impact on Passive LNP Delivery
In a recent Advancing RNA Live session, Dominik Witzigmann of NanoVation and John Zuris of Stealth Co dissected emerging lipid chemistries that enhance passive lipid nanoparticle (LNP) delivery. They highlighted breakthroughs in ionizable lipids, helper lipids, and PEG‑lipids that improve...
By Life Science Connect

Video•Feb 24, 2026
How Microfluidics & QbD Are Maturing LNP Manufacturing
In a recent discussion, CMC consultant Sujit Jain and NanoVation CEO Dominik Witzigmann highlighted how microfluidic platforms combined with Quality‑by‑Design (QbD) principles are transforming lipid nanoparticle (LNP) production for mRNA therapeutics. They noted that continuous‑flow microfluidics now enable precise control...
By Life Science Connect

Video•Feb 24, 2026
Primates, Patents, & Progress: Advancements In Rational LNP Design
In a recent Advancing RNA Live segment, Dominik Witzigmann of NanoVation Therapeutics and John Zuris of Stealth Co discussed the latest scientific breakthroughs shaping lipid nanoparticle (LNP) design. They highlighted rational, data‑driven approaches that improve particle stability, targeting precision, and...
By Life Science Connect

Video•Feb 24, 2026
The Evolving CMC Landscape for mRNA-LNPs
In a recent Advancing RNA Live segment, CMC consultant Sujit Jain outlined the current maturity levels across process development, analytical methods, and supply‑chain logistics for mRNA‑LNP products. He highlighted that COVID‑19 vaccines and liver‑targeted therapeutics now operate on a reproducible,...
By Life Science Connect