
STAT+: Drug Meant to Make Gene Therapy Safer May Also Make It Less Effective
A trial by Encoded Therapeutics investigating a gene‑therapy for Dravet syndrome found that adding the immune‑suppressant sirolimus may blunt therapeutic benefit. The study enrolled 21 children, most of whom received standard steroids, while a high‑dose subgroup also received sirolimus. Researchers observed reduced efficacy in the sirolimus cohort despite its intended role in preventing immune reactions to viral vectors. The results suggest a trade‑off between safety and effectiveness in gene‑therapy protocols.

STAT+: Why Marty Makary Was the Worst FDA Commissioner in 25 Years
FDA Commissioner Marty Makary resigned on Tuesday amid mounting pressure from staff and lawmakers, with critics labeling him the agency’s worst leader in the past 25 years. Observers cite his lack of understanding of the FDA’s mission, a wave of...

STAT+: Makary Departs FDA Amid Turmoil as Diamantas, Agency’s Top Food Official, Steps In
FDA Commissioner Marty Makary announced his resignation, prompting President Donald Trump to confirm the departure during a press briefing before his China trip. Makary cited personal difficulties, and the administration named Kyle Diamantas, the agency’s senior food regulator, as acting commissioner....
STAT+: Capsida Says It Still Doesn’t Know What Caused Gene Therapy Death
Capsida Biotherapeutics announced it still cannot determine the cause of a child’s death that occurred during the September 2025 CAP-002 gene‑therapy trial, the first brain‑penetrating treatment of its kind. The investigation is stalled because the hospital conducting the study has...
STAT+: Medicare Is Spending Far Less than Expected on New Alzheimer’s Drugs
Medicare’s uptake of the newly approved Alzheimer’s therapies Leqembi and Kisunla is far lower than projected, prompting the agency to forecast minimal spending through 2026‑2027. Earlier estimates had anticipated billions in annual costs, but real‑world adoption has stalled. The drugs...

STAT+: Five Years After Disaster, a Rare Disease Community Gets New Chance at Treatment
Astellas Pharma has re‑initiated its gene‑therapy trial for X‑linked myotubular myopathy (XLMTM) five years after a previous study was halted by safety concerns. Early observations from the first participant, Joshua "JJ" Gonzalez, indicate a dramatic reduction in airway suctioning, suggesting...

The Biotechs Wear Prada
STAT’s annual "The Readout" tradition of turning Met Gala outfits into biotech logos continued in 2026. This year’s dress code, “Fashion is Art,” dovetails with pharmaceutical firms’ push to treat corporate logos as artistic statements. The article highlights Janelle Monáe’s Met Gala appearance...

STAT+: FDA Revisits a Rare Cancer Treatment It Rejected a Few Months Ago
The FDA has announced it will re‑evaluate a rare‑cancer therapy it dismissed just months earlier, citing new data submitted by the drug’s sponsor. The treatment, aimed at a subtype of metastatic sarcoma, originally failed to meet the agency’s efficacy benchmarks...
STAT+: Next-Gen Duchenne Drug From Entrada Disappoints
Entrada Therapeutics reported that its next‑generation exon‑skipping drug for Duchenne muscular dystrophy failed to achieve its primary efficacy endpoints in an early‑stage trial. The study showed only a modest rise in dystrophin levels, far below the thresholds set by the...

STAT+: Novo Nordisk’s Less-Bad News on Its Wegovy Pill Boosts Earnings and Share Price
Novo Nordisk reported first‑quarter results that showed a modest improvement in its outlook for the obesity drug Wegovy. The company narrowed its adjusted sales decline forecast to 4%‑12% for the year, up from a prior 5%‑13% range, and aligned its...

STAT+: Even at a Meeting in Rome, FDA Shifts Are Top of Mind for Gene Therapy Field
At a gene‑therapy summit in Rome, Tim Hunt highlighted recent FDA approvals of rare‑disease treatments from Rocket Pharmaceuticals and Regeneron as signs of progress. He also flagged the departure of Vinay Prasad, the FDA’s top regulator for cell and gene therapies,...

STAT+: Are Analysts Too Quick to Gloss over Lilly’s Liver Case?
Analysts are being criticized for downplaying a recent liver safety issue at Eli Lilly, raising concerns about market oversight. Meanwhile, biotech earnings showed mixed results, with Vertex shelving an mRNA cystic fibrosis candidate and Pfizer and Alkermes delivering near‑consensus Q1 numbers....

STAT+: Cytokinetics Drug Myqorzo Meets Twin Efficacy Goals in Study of Genetic Heart Disease
Cytokinetics announced that its Phase 3 ACACIA trial met both primary efficacy endpoints for Myqorzo in patients with non‑obstructive hypertrophic cardiomyopathy, showing significant symptom relief and improved cardiovascular fitness. Myqorzo is already approved for the obstructive form of HCM, and this...

UCB to Acquire Maker of Antibody Treatments for Autoimmune Diseases
UCB announced a definitive agreement to acquire a privately held biotech that develops monoclonal antibody treatments for autoimmune diseases. The target company’s portfolio includes candidates for rheumatoid arthritis, lupus and multiple sclerosis, complementing UCB’s existing immunology assets. Financial terms were...
STAT+: Biotech Raises $42 Million to Run Huntington’s Disease Trial
Latus Bio, a gene‑therapy startup founded by CHOP scientist Beverly Davidson, announced a $42 million financing round to launch its first human trials. The company is advancing two programs this year: a CLN2 disease therapy with data expected by year‑end, and...
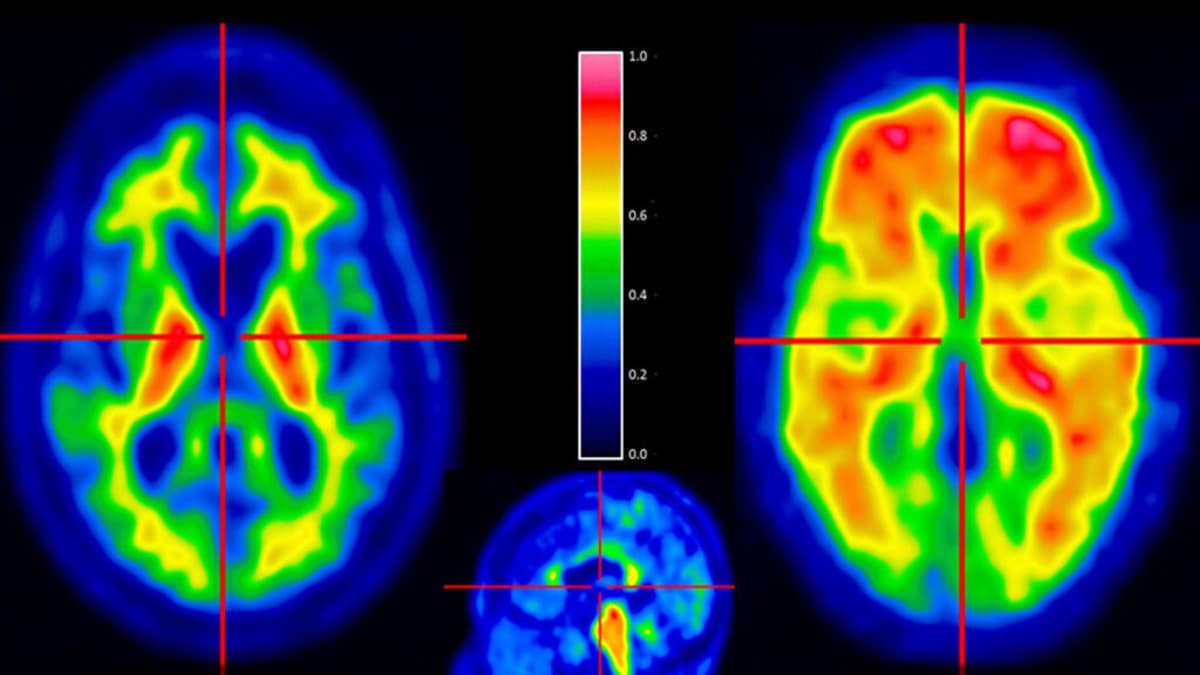
STAT+: Axsome Wins FDA Nod for Alzheimer’s Agitation
Axsome Therapeutics announced that the U.S. Food and Drug Administration has granted a regulatory nod for its investigational therapy aimed at treating agitation in Alzheimer’s disease. The agency’s decision clears the path for accelerated clinical development, potentially moving the drug...
STAT+: Her Daughter Mila Got a Bespoke Medicine. Now She’s Starting a New Biotech to Make More
Julia Vitarello, whose eight‑year‑old daughter Mila was treated with a tailor‑made gene therapy, announced she is launching a new biotech to scale individualized medicines. Her previous company, EveryONE Medicines, folded after FDA guidance on custom therapies proved insufficient for investors....

Remembering J. Craig Venter: A Relentless Scientist Who Changed Biotech — and Was All Too Easily Misunderstood
J. Craig Venter, the pioneering genomics entrepreneur who died at 79, reshaped biotechnology by accelerating the human genome sequence and building the first synthetic cell. His private‑sector efforts, including Celera and Synthetic Genomics, turned DNA sequencing into an industrial process. Venter’s...
STAT+: Five-Way Obesity Drug Is Super Effective — in Mice
Researchers have unveiled a preclinical obesity candidate that combines five distinct mechanisms, delivering dramatic weight loss in mouse models and outperforming current GLP‑1 therapies. The multi‑modal approach targets appetite, metabolism, gut hormones, energy expenditure, and adipose tissue remodeling, achieving up...

STAT+: Boehringer’s Obesity Drug Shows 16.6% Weight Loss; More Data Needed
Boehringer Ingelheim reported that its experimental obesity drug produced an average 16.6% weight loss in a mid‑stage trial, signaling strong efficacy against a disease affecting over 100 million Americans. The study involved roughly 300 participants over 68 weeks and showed improvements...
STAT+: Erasca Touts Strong, Though Preliminary, Results in Trial of Pancreatic and Lung Cancer Therapy
Erasca announced that its oral RAS‑targeting drug ERAS‑0015 produced tumor shrinkage in 40% of patients with advanced pancreatic cancer and 62% of those with advanced non‑small‑cell lung cancer. The early‑stage data, gathered from trials in the United States and China,...

STAT+: Astellas Retries XLMTM Gene Therapy After Deaths
Astellas Pharma announced it will restart its next‑generation gene therapy trial for X‑linked myotubular myopathy (XLMTM) after pausing the program following two patient deaths. Meanwhile, Intellia Therapeutics reported its one‑time CRISPR treatment lonvo‑z reduced hereditary angioedema attacks by 87% in...
STAT+: Oruka’s Long-Acting Psoriasis Therapy Posts Strong Results in Mid-Stage Study
Oruka Therapeutics reported that its long‑acting injectable, ORKA‑001, achieved complete skin clearance in 63% of plaque‑psoriasis patients during a mid‑stage trial. The data suggest the drug can be administered just once per year, a stark contrast to current biologics that...

STAT+: Veradermics’ Hair Loss Drug Succeeds in Late-Stage Trial
Veradermics announced that its oral hair loss drug VDPHL01 met primary endpoints in a Phase III trial. Over six months, participants taking the pill grew 30‑33 hairs per square centimeter versus seven in the placebo group. Patient‑reported improvement reached 79‑86%, and...

STAT+: FDA to Speed up Review of Three Psychedelics as Mental Health Treatments
The FDA announced it will grant priority‑review vouchers to accelerate the evaluation of three psychedelic therapies—Compass Pathways’ psilocybin for treatment‑resistant depression, Usona Institute’s psilocybin for major depressive disorder, and Transcend Therapeutics’ MDMA‑like compound for PTSD. The move is part of...

STAT+: Sanofi Research Priorities in Flux as New CEO Logs In
Sanofi’s new chief executive, Belén Garijo, assumes leadership amid questions about the French drugmaker’s research direction. The company recently intensified its immunology portfolio, but recent trial disappointments have dampened expectations. Garijo is expected to reassess R&D allocations, potentially shifting focus...

STAT+: Can Erasca Be Biotech’s Next Big Thing? We’ll See
Erasca, a biotech startup valued at roughly $7 billion, is developing ERAS‑0015, a pan‑RAS inhibitor aimed at treating pancreatic cancer. The company positions itself as a cost‑effective alternative to RevMed, whose market cap exceeds $30 billion after reporting a 13.2‑month median overall...

STAT+: At AACR, Talk of Chinese Biotech, Oncology’s Comms Issue, and More
Revolution Medicines highlighted two key updates at the AACR meeting: promising frontline pancreatic cancer data for its RAS inhibitor daraxonrasib and the introduction of a novel compound, RM-055. RM-055 is described as a catalytic inhibitor that can strip a phosphate...

STAT+: Gene Therapy Trial for Deafness Adds Evidence to Drug’s Efficacy
Researchers have reported that a gene‑therapy injection dramatically improved hearing in a Chinese clinical trial, with 90% of participants noting significant gains. The study, published in Nature, includes both children and adults, such as a 32‑year‑old who regained functional hearing....

STAT+: From Revolution Medicines, More Strong Data on KRAS Drug and a Glimpse of a ‘Novel Class’ Beyond It
Revolution Medicines presented new clinical data on its next‑generation KRAS inhibitor daraxonrasib at the AACR meeting, highlighting strong first‑line activity and synergistic effects in combination regimens for advanced pancreatic cancer. The company also unveiled pre‑clinical results for RM‑055, a candidate...

STAT+: Kyverna Therapeutics Plans to Submit Cell Therapy for Stiff Person Syndrome for FDA Approval
Kyverna Therapeutics announced that its personalized CAR‑T cell therapy improved mobility and reduced disability in patients with stiff person syndrome, a rare autoimmune neurological disorder. The company plans to file an FDA submission by mid‑2026, which would make it the...

STAT+: Key GOP Senators Push Back on Trump’s Plan to Cut NIH, Reorganize HHS
During a Senate appropriations health subcommittee hearing, bipartisan senators questioned Health Secretary Robert F. Kennedy Jr. about the White House’s 2027 budget proposal that would slash the Department of Health and Human Services (HHS) by 12%. The plan calls for...
STAT+: Insurers Refuse to Join Medicare Pilot Offering Weight Loss Drugs to Seniors at Steep Discount
The Trump administration negotiated a $245‑per‑month price with Eli Lilly and Novo Nordisk for obesity drugs, intending to offer seniors a $50 monthly copay through a Medicare pilot called BALANCE. Insurers refused to join, arguing the program would strain their finances. Consequently,...

STAT+: Tortugas Neuroscience Launches with Hopes to Develop Drugs for Brain Disorders, Other Conditions
Tortugas Neuroscience launched with a $106 million financing round led by Cure Ventures, The Column Group and AN Ventures. The startup has licensed two schizophrenia and tinnitus candidates from China’s Jiangsu Hansoh and two additional therapies for focal epilepsy and encephalopathies...
STAT+: BioAge Says Experimental Pill Aimed at Reducing Heart Risks Significantly Reduced Inflammation
BioAge Labs reported that its investigational cardiovascular‑risk pill BGE‑102 dramatically lowered inflammation in a Phase 1 trial. A 60‑mg dose cut high‑sensitivity C‑reactive protein (hs‑CRP) by 85% after one week, with the effect persisting through three weeks. The same reduction was...

STAT+: At AACR, a Provocative Use of CAR-T, Merck’s New Thing and Cancer’s Geography Problem
At the AACR 2026 meeting, early‑phase data showed Merck‑partnered CAR‑T therapy Carvykti eliciting deep responses in 20 high‑risk smoldering multiple myeloma patients, suggesting a preventive angle for a precursor disease. The conference also featured Merck’s first glimpse of an oncology...

STAT+: Former Banker Brought on to Revitalize Pfizer’s Strategy Departs His Post
Pfizer announced that Andrew Baum, the former Citi analyst who joined as executive vice president and chief strategy and innovation officer in June 2024, is leaving his post. Baum will remain as an adviser to CEO Albert Bourla through the...
STAT+: In Early Trial, CAR-T Results Raise Hope of Preventing Multiple Myeloma in High-Risk Patients
A phase‑I trial of CAR‑T therapy in high‑risk smoldering multiple myeloma reported that all 20 participants achieved undetectable disease after treatment. The results, presented at the AACR meeting, suggest the possibility of preventing progression to active myeloma, a deeper response...

STAT+: Eli Lilly’s $3.25 Billion Acquisition of Kelonia Therapeutics Caps Startup’s Tortuous Ride
Eli Lilly announced a $3.25 billion acquisition of Kelonia Therapeutics, a small biotech focused on cell therapies for cancer and autoimmune diseases. The deal may include additional payments if Kelonia meets clinical, regulatory, and commercial milestones. Kelonia, formerly Elcano Therapeutics, survived on...
STAT+: Finally Cracking KRAS as a Druggable Target
Revolution Medicines announced that its KRAS inhibitor daraxonrasib produced tumor shrinkage in a Phase 2 trial for pancreatic cancer, marking the first clear efficacy signal for an oral drug targeting the once‑undruggable KRAS protein. The result follows a wave of...

STAT+: Extended Use of Nektar Therapeutics Drug Shows Promise in Alopecia
Nektar Therapeutics announced that its experimental oral drug rezpeg produced significant hair regrowth in patients with severe alopecia areata. After a year of treatment, 27% of participants reached a SALT Score 20, meaning at least 80% of the scalp was covered...

STAT+: The Race to Catch KRAS, Pancreatic Cancer’s ‘Greasy Ball,’ and Create the Most Promising Drug in Decades
A new wave of KRAS‑targeted therapies is reshaping pancreatic cancer treatment after decades of failure. Revolution Medicines’ daraxonrasib, a next‑generation KRAS inhibitor, delivered dramatic survival benefits for patient Leanna Stokes, who enrolled in a clinical trial. The drug’s success has...

STAT+: FDA Eyes Expanding Testosterone Therapy for Libido
The FDA is reviewing data that could broaden testosterone‑replacement therapy to include low libido as an approved indication, a move that would extend the drug’s market beyond hypogonadism. If cleared, the label change could add roughly $1.5 billion in annual U.S....

STAT+: Researchers Behind GLP-1 Obesity Drugs Advance New Approach: Drop GLP-1 as a Target
Researchers who helped create GLP‑1 obesity drugs such as Eli Lilly’s Zepbound are now testing a different strategy. A team led by Richard DiMarchi and Matthias Tschöp engineered a molecule that activates GIP and glucagon receptors, showing weight‑loss results in...
STAT+: Cochrane Review Reignites Alzheimer’s Amyloid Wars
The FDA announced it will convene an external advisory panel to revisit rules on compounded peptides, with meetings slated for July and a follow‑up before February 2027. A new Cochrane review has reignited controversy over amyloid‑targeting Alzheimer’s therapies, questioning their...

STAT+: Roche to Launch Another Elevidys Trial, with Eyes on European Approval
Roche announced a new Phase 3 trial of Elevidys, the gene‑therapy for Duchenne muscular dystrophy, targeting European approval after a negative EMA review last year. The study will enroll roughly 100 boys in the early stages of the disease and compare...

STAT+: Novartis CEO Joins Anthropic’s Board
Novartis chief executive Vasant Panda has been appointed to the board of AI startup Anthropic, underscoring the growing convergence of pharma and generative‑AI. At the same time, Revolution Medicines secured $2 billion through simultaneous equity and debt offerings after its experimental pancreatic‑cancer...

STAT+: New Bain Biotech Startup, Building on BMS Drugs, Gets a Name and a CEO
Bain Capital Life Sciences has launched a new biotech venture, Beeline Medicines, backed by a $300 million investment and five drug assets licensed from Bristol Myers Squibb. The startup will focus on inflammatory and immune‑mediated diseases, beginning with an oral candidate for...

Opinion: My Brother Can’t Access a Just-Approved Breakthrough Drug for His Rare Disease
A newly FDA‑approved breakthrough drug promises to address the neurological degeneration that has long plagued patients with Hunter syndrome, a rare lysosomal disorder. While the approval marks a scientific milestone, patients like the author’s 28‑year‑old twin brother still face barriers...

For Ben Sasse, Revolution Medicines’ Pancreatic Cancer Trial Felt Like His Best, only Option
Former U.S. senator Ben Sasse was diagnosed with metastatic pancreatic cancer in December and promptly enrolled in an early‑phase trial of Revolution Medicines' targeted drug daraxonrasib. The therapy, positioned as a first‑line option, aims to extend both the quantity and...