Disorder Drives One of Nature’s Most Complex Machines
•March 9, 2026
0
Why It Matters
Understanding the NPC’s dynamic gating mechanism opens pathways for targeted therapies that can block pathogens or deliver drugs to the nucleus, a critical frontier in disease treatment.
Key Takeaways
- •High‑speed AFM captures millisecond NPC dynamics
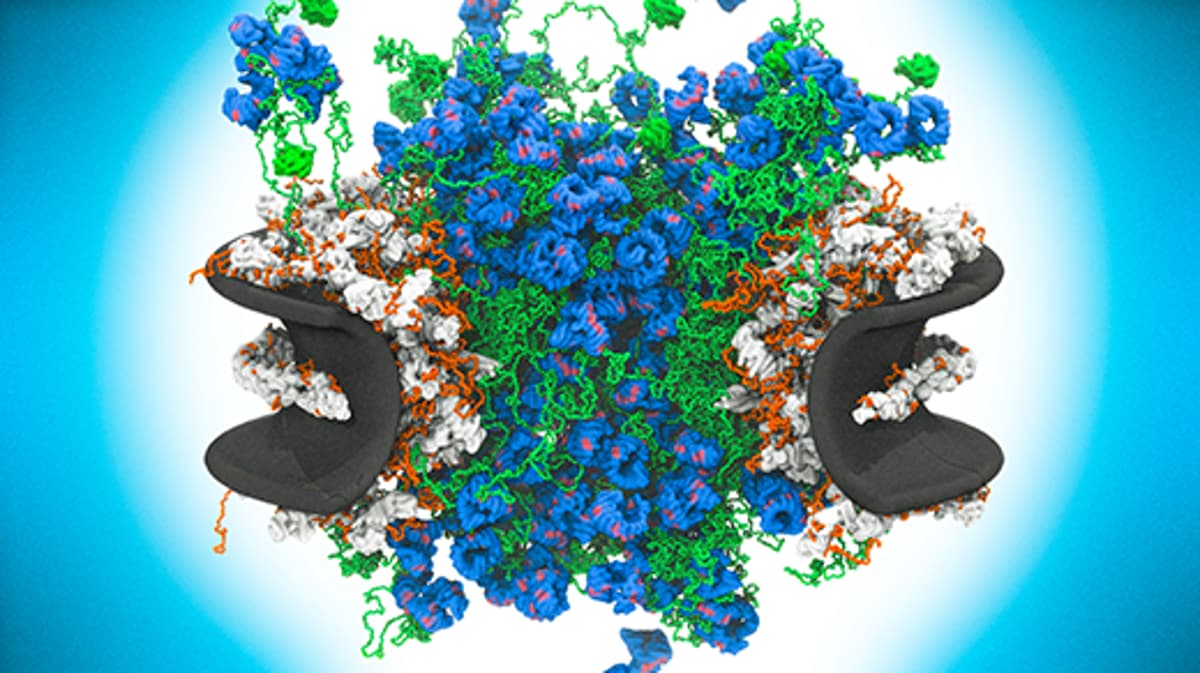
- •Central plug consists of karyopherins and cargo
- •Findings favor brush‑like “virtual gate” model
- •NPC dysfunction linked to cancer, viruses, neuro disorders
- •Synthetic pores reproduce native NPC transport behavior
Pulse Analysis
The nuclear pore complex (NPC) has long been a structural enigma, with static images masking the frantic motion at its core. Recent advances in high‑speed atomic force microscopy have finally pierced this veil, delivering millisecond‑resolution movies that show the transport channel’s intrinsically disordered FG‑nucleoporins flailing like seaweed while a dense, mobile plug of karyopherin proteins darts through the center. This dynamic portrait overturns the decades‑old gel‑mesh hypothesis, aligning instead with the “virtual gate” concept where rapid, brush‑like fluctuations dictate selectivity and throughput.
Beyond basic biology, the implications for medicine are profound. The NPC serves as the cell’s primary conduit for genetic information and regulatory proteins; its malfunction is increasingly implicated in oncogenesis, viral hijacking, and neurodevelopmental disorders. By mapping how transport factors temporarily block or facilitate passage, researchers can envision novel interventions—small molecules or engineered proteins that either reinforce the gate against viral entry or shepherd therapeutic cargos directly into the nucleus, enhancing gene‑editing or anti‑cancer strategies.
The study also demonstrates the power of synthetic biology to validate complex cellular mechanisms. Recreating NPC‑sized pores with tethered nucleoporins reproduced the central plug phenomenon, confirming that the observed dynamics are intrinsic to the protein composition rather than an artifact of the imaging technique. As imaging technologies like Minflux and cryo‑EM continue to evolve, a near‑atomic, real‑time view of the NPC may soon become routine, accelerating drug discovery and deepening our grasp of cellular logistics.
Disorder Drives One of Nature’s Most Complex Machines
0
Comments
Want to join the conversation?
Loading comments...