Newly Identified Barrier Cells Seal Off Choroid Plexus From CSF, Rest of Brain
Why It Matters
The discovery revises the fundamental understanding of central nervous system compartmentalization, influencing how scientists approach neuroinflammation, drug delivery, and immune surveillance in the brain.
Key Takeaways
- •Fibroblasts form tight junction barrier at choroid plexus base.
- •Barrier present in mice and human postmortem brains.
- •Inflammation makes barrier leaky, enabling immune cell entry.
- •Barrier challenges notion that only endothelial/epithelial cells create brain borders.
- •Single‑cell RNA sequencing revealed CLDN11‑expressing fibroblast subpopulation.
Pulse Analysis
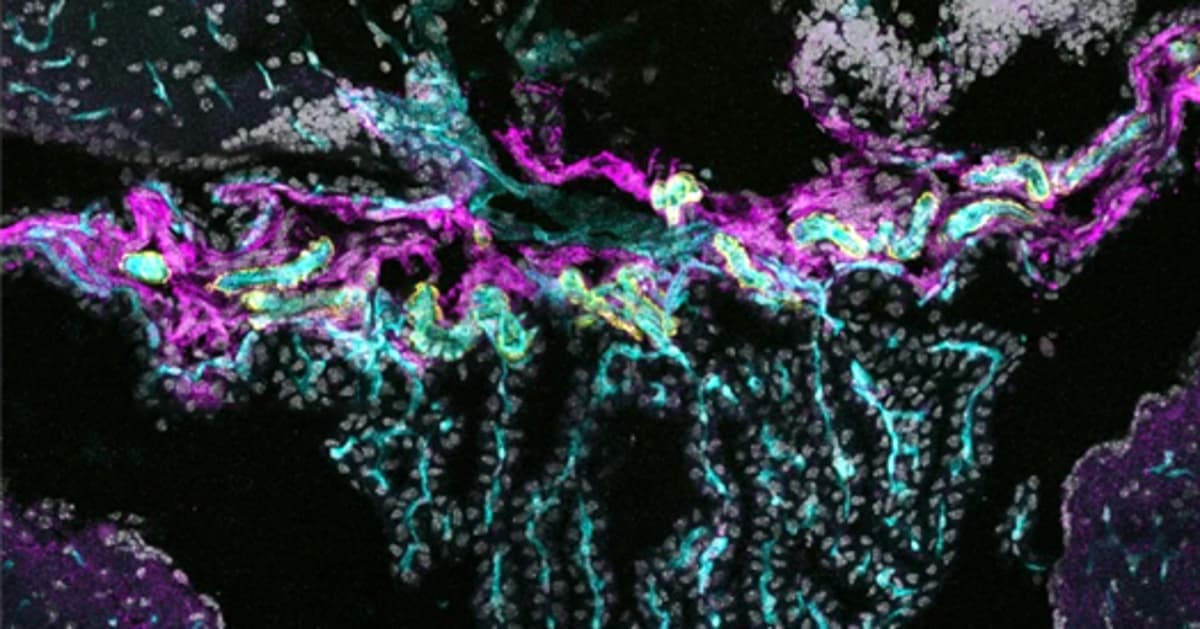
The brain’s protective architecture has long been defined by the blood‑brain barrier and the blood‑CSF interface, both built primarily from endothelial and epithelial cells. Recent single‑cell RNA sequencing of the choroid plexus uncovered a distinct fibroblast cohort expressing the tight‑junction protein CLDN11. Electron microscopy confirmed that these cells cluster at the plexus base, forming adherens and tight‑junction complexes that effectively seal the region from circulating cerebrospinal fluid. This structural insight expands the catalog of cellular players that maintain central nervous system homeostasis.
Functionally, the newly described barrier behaves like a dynamic gatekeeper. Under basal conditions, tracer studies show that substances injected into the bloodstream cannot penetrate the choroid plexus, while ventricular tracers remain confined, confirming barrier integrity. However, exposure to lipopolysaccharide—a bacterial component that triggers inflammation—disrupts the seal, allowing immune cells to traverse from blood to brain tissue. This leaky response implicates the fibroblast barrier as a potential primary route for immune surveillance and neuroinflammatory processes, prompting a shift in focus for researchers studying brain‑immune interactions.
Looking ahead, the presence of analogous fibroblast barriers in human brains suggests translational relevance. Understanding how these cells coordinate with other meningeal and vascular barriers could inform strategies to modulate drug delivery across the CNS or to mitigate pathological inflammation. Moreover, targeting the molecular regulators of fibroblast tight‑junction formation may offer novel therapeutic avenues for conditions such as meningitis, multiple sclerosis, or neurodegenerative diseases where barrier dysfunction plays a critical role. The discovery thus not only revises basic neuroanatomy but also opens a frontier for clinical innovation.
Newly identified barrier cells seal off choroid plexus from CSF, rest of brain
Comments
Want to join the conversation?
Loading comments...