Novartis' Oral BTK Drug Moves the Needle in CINDU
Companies Mentioned
Why It Matters
Rhapsido’s potential approval fills a major therapeutic gap in CINDU, offering a disease‑modifying option beyond antihistamines. It also diversifies Novartis’s dermatology portfolio and could generate a new high‑margin revenue stream.
Key Takeaways
- •Rhapsido shows higher complete response rates vs placebo
- •First targeted therapy potential for chronic inducible urticaria
- •Novartis filed worldwide approvals for symptomatic dermographism
- •CINDU affects 0.5% global population, 29 million cases
- •Rhapsido could avoid competition with Dupixent in CSU
Pulse Analysis
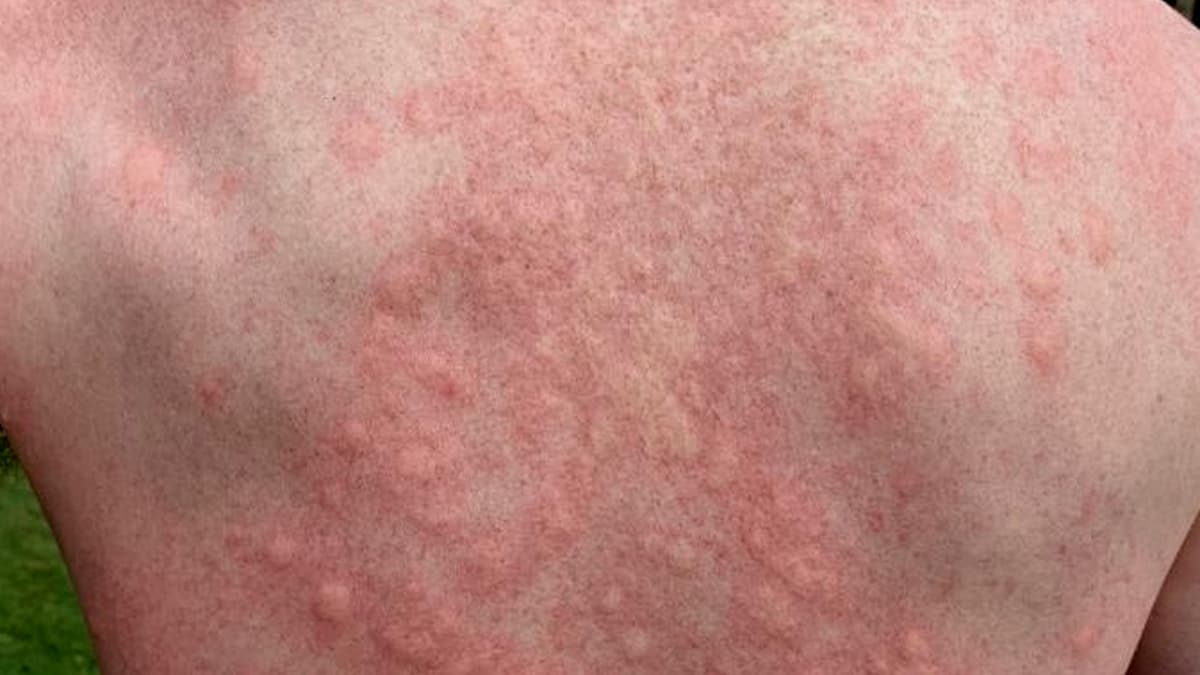
Chronic inducible urticaria (CINDU) remains an underserved segment of the dermatology market, affecting roughly half a percent of people worldwide and manifesting as hives triggered by physical stimuli. Patients typically rely on avoidance strategies and second‑generation antihistamines, with off‑label biologics like Xolair offering limited relief. The phase 3 RemIND trial, enrolling 364 antihistamine‑refractory participants, demonstrated that remibrutinib markedly improves complete response rates across symptomatic dermographism, cold urticaria, and cholinergic urticaria, signaling a breakthrough in disease‑specific therapy.
Novartis’s rapid filing for symptomatic dermographism and upcoming submissions for the other CINDU subtypes underscore a strategic push to capture a niche yet sizable market. By securing a first‑in‑class indication, Rhapsido can sidestep direct competition with Sanofi‑Regeneron’s Dupixent, which dominates the chronic spontaneous urticaria (CSU) arena. This differentiation not only broadens Novartis’s dermatology pipeline but also promises higher pricing power, given the lack of existing targeted options for CINDU patients.
The success of Rhapsido may catalyze broader interest in oral BTK inhibitors for allergic and inflammatory skin disorders. As Novartis leverages early uptake data from its CSU launch, the company anticipates scaling sales to rival its historic dermatology hits. Meanwhile, competitors are advancing alternative modalities—such as Sanofi’s KIT inhibitor SAR449028—highlighting a burgeoning race to dominate the urticaria space. For investors and clinicians alike, Rhapsido’s progress signals both a therapeutic advance for patients and a potential new growth engine for Novartis.
Novartis' oral BTK drug moves the needle in CINDU
Comments
Want to join the conversation?
Loading comments...