Vaginal Drug Delivery Had A Funding Problem—Merck Changed That
Companies Mentioned
Why It Matters
The Merck‑Calla Lily deal validates a trillion‑dollar women’s health opportunity and could unlock funding for technologies that have long been overlooked, reshaping the med‑tech investment landscape.
Key Takeaways
- •Merck partners with Calla Lily to advance Callavid vaginal drug platform
- •Callavid targets $22 billion market across IVF, miscarriage, preterm birth
- •Funding gap stems from decades of under‑studied women’s health research
- •AI models inherit bias from historic exclusion of women in clinical data
- •Calla Lily raised ~$10 million without institutional investors, highlighting capital barriers
Pulse Analysis
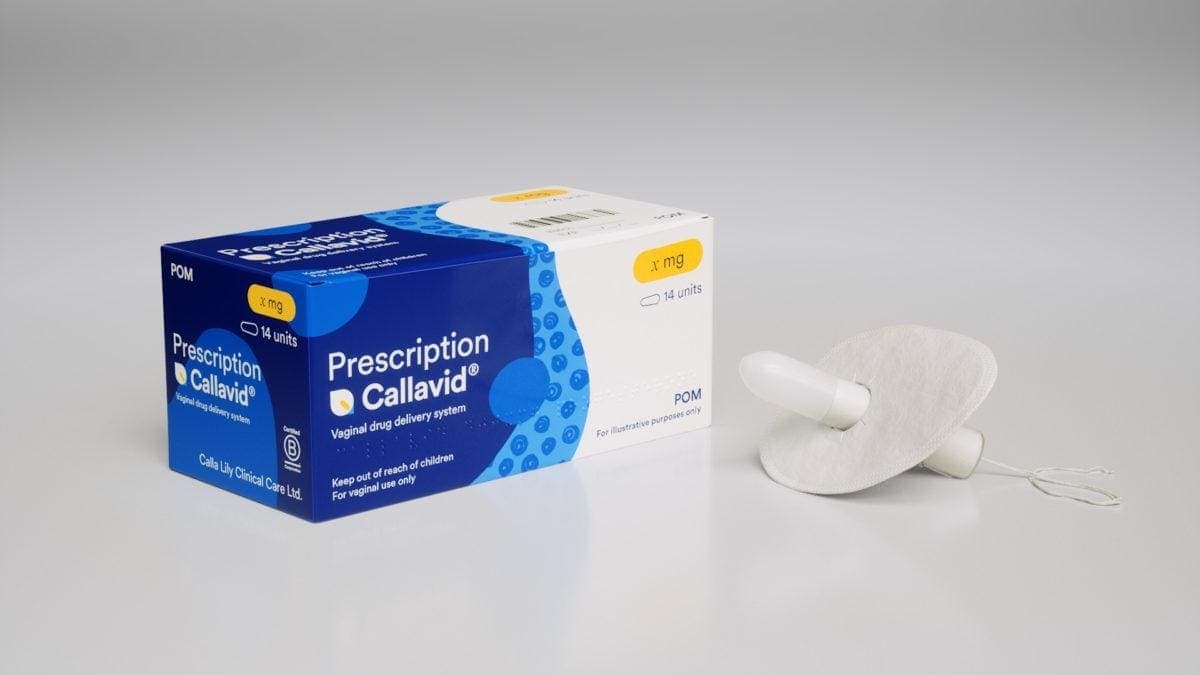
The collaboration between Merck and Calla Lily marks a watershed moment for women’s health innovation, marrying a proven pharmaceutical giant’s resources with a niche, leak‑proof delivery system. Callavid’s tampon‑shaped design solves a long‑standing problem: medication loss and patient discomfort that have hampered the efficacy of vaginal pessaries and suppositories. By securing FDA 510(k) clearance and a portfolio of patents across the U.S., Europe, Japan and China, the platform is positioned to capture a $22 billion market that spans IVF luteal‑phase support, miscarriage prevention, preterm‑birth mitigation, and pre‑eclampsia therapies. This partnership not only accelerates clinical trials but also signals to the broader industry that women‑centric solutions can deliver robust commercial returns.
Beyond the immediate commercial upside, the Merck deal highlights systemic funding disparities that have plagued women’s health for decades. Only about 9 % of NIH grants target female‑specific research despite women comprising over half of the global population, a gap that has produced blind spots in both drug development and emerging AI models trained on skewed data. The resulting inefficiencies cost the UK’s NHS roughly £236 million ($319 million) annually in avoidable losses, illustrating the economic toll of neglect. By channeling capital into Callavid, Merck is effectively betting on correcting these historic imbalances, which could spur a wave of investment into other under‑funded therapeutic areas.
The broader implications extend to the venture ecosystem, where traditional VC criteria often sideline founders with non‑linear career paths, particularly women in healthcare. Calla Lily’s ability to raise nearly $10 million—$7 million from private angels and $2.5 million in non‑dilutive grants—without institutional backing underscores the resilience required to navigate a biased funding landscape. Initiatives like the Material Change Institute aim to diversify the investor pipeline, ensuring that domain experts like Dr. Lara Zibners can both shape and benefit from capital allocation. As Merck’s endorsement validates the commercial promise of gender‑focused med‑tech, the industry may witness a recalibration of investment theses, fostering a more inclusive and financially rewarding future for women’s health innovations.
Vaginal Drug Delivery Had A Funding Problem—Merck Changed That
Comments
Want to join the conversation?
Loading comments...