
Lecture 3.2.8: FDA Digital Health & CE Mark Pathways
The lecture walks developers through the regulatory maze for software‑based medical devices, comparing the U.S. FDA framework with the European CE‑mark pathway. It defines "software as a medical device" (SaMD), outlines the FDA’s three‑tier risk classification, and explains how each tier dictates the appropriate market‑entry route. Key insights include the three U.S. pathways—510(k) clearance for predicate‑based tools, the de novo route for novel low‑to‑moderate risk products, and the PMA for high‑risk devices—along with their typical timelines (3‑6 months, 6‑12 months, and 12‑24 months respectively). The EU’s MDR focuses heavily on documentation, quality‑system compliance, and notified‑body audits, with Rule 11 classifying SaMD into low, medium, and high risk. Both regions now demand a Software Bill of Materials (SBOM) and rigorous post‑market surveillance. Illustrative examples such as an AI‑powered skin‑cancer detection app (high‑risk Tier 3) and a medication‑reminder app (low‑risk Tier 1) highlight how intended‑use language can shift a product’s classification. The 2026 TEMA pilot is introduced as a rolling‑review mechanism to accelerate high‑impact AI tools, while the speaker stresses the importance of aligning technical files to satisfy both FDA and MDR requirements. For innovators, harmonizing documentation to meet the stricter MDR standards can cover roughly 80% of FDA expectations, streamlining global market entry, reducing time‑to‑value, and protecting investment. Mastering regulatory strategy is thus as critical as building the predictive model itself.

Lecture 3.2.5: Signal Preprocessing ECG, PPG + Feature Extraction, Windowing & HRV Spectral Features
The lecture walks through converting raw ECG and PPG voltages into actionable physiological metrics, focusing on preprocessing, feature extraction, and heart‑rate‑variability (HRV) spectral analysis. Aksha outlines three dominant noise sources—power‑line interference, baseline wander, and EMG artifacts—and recommends a “Goldilocks” filter chain:...

Lecture 3.2.3: Transfer Learning & Domain Adaptation , Class Imbalance & Augmentation
The lecture focuses on practical strategies—transfer learning, domain adaptation, class‑imbalance handling, and data augmentation—to build reliable AI systems for healthcare, where data are often noisy, biased, and scarce. Key insights include leveraging pre‑trained models by freezing early convolutional layers and fine‑tuning...

Lecture 3.2.2: U Net Segmentation Variants
The lecture introduces U‑Net segmentation variants for medical imaging, emphasizing need for pixel‑wise tumor delineation rather than simple presence detection. It reviews core U‑Net architecture—contracting encoder for context, expanding decoder for localization, and skip connections that transmit high‑resolution details. It then...

2.2.3 | Stakeholder Mapping | Masters in Health Economics
The video introduces stakeholder mapping as a core tool in health economics, defining it as a systematic way to list every individual or group that can affect or be affected by health policy decisions. It explains why mapping matters: it...

2.2.4 Theory of Change | Masters in Health Econmics
The video introduces Theory of Change (TOC) as a roadmap that connects health program inputs, activities, outputs, outcomes, and ultimate impact. It explains that TOC answers the how and why a program should work, turning abstract goals into a logical...

2.2.5 | Fiscal Incidence Methods | Masters in Health Economics
The lecture introduces fiscal incidence – the study of who actually bears the economic burden of taxes and government spending – and explains why it matters for health‑economics policy analysis. It then presents the difference‑in‑differences (DiD) technique as the standard tool...

Lecture 3.5.5 | Human-Robot Interaction & Cognitive Load | Masters in Medical Robotics
The lecture introduces human‑robot interaction (HRI) and cognitive load as intertwined design challenges for medical robotics and other domains. Effective HRI requires robots to convey intent, status, and data in a clear, predictable manner, preventing distraction. High cognitive load—when users must...

1.3.2 Regression Methods | Masters in Global Health Economics
The video introduces regression techniques tailored for health‑economics research, emphasizing how econometric tools move beyond simple correlations to uncover causal policy impacts. It outlines the multivariate model framework, where the outcome (Y) is linked to a treatment variable (X1) and...

1.3.2 | Regression Discontinuity Design | Masters in Health Economics
The video introduces regression discontinuity design (RDD) as a quasi‑experimental tool for health‑policy evaluation, explaining how statutory cutoffs such as age‑65 Medicare eligibility or birth‑weight thresholds generate locally random assignment of treatment. It distinguishes sharp RDD, where the probability of treatment...

Lecture 3.1.12: Partner Scoping & Secondary Data Discovery
Partner scoping and secondary data discovery are presented as the first step in health data science, emphasizing that researchers should ask whether needed data already exist before launching expensive primary surveys. The lecture outlines the three cornerstone repositories for low‑...
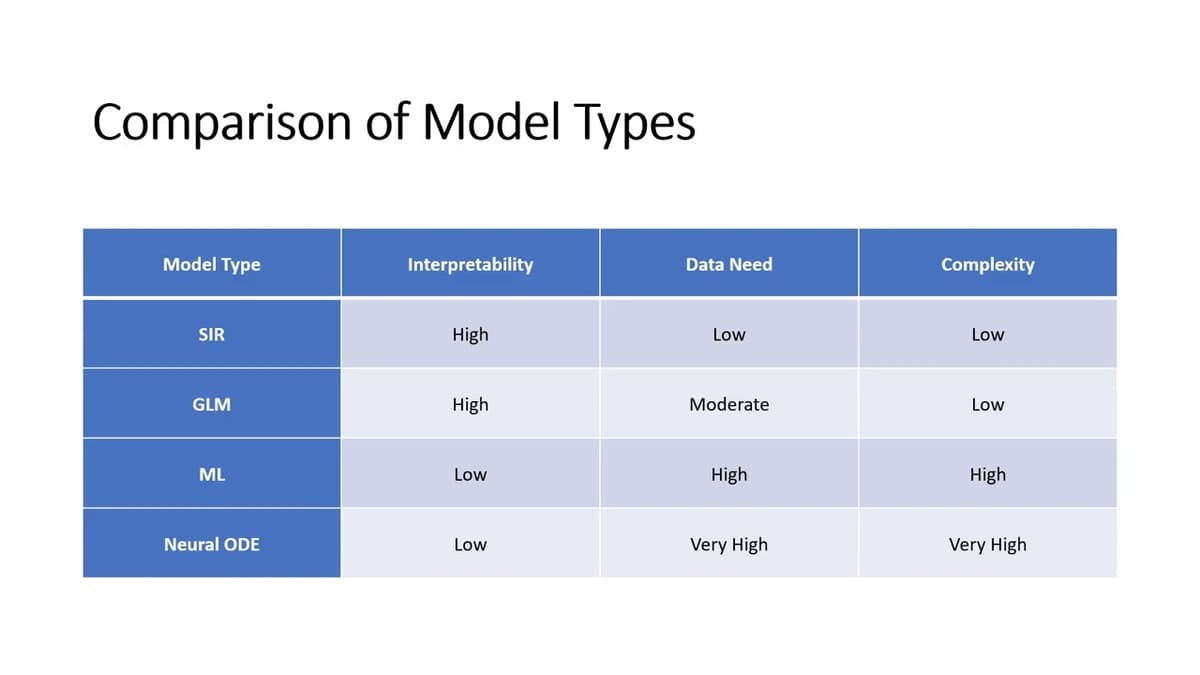
Lecture 1: Disease Modelling Introduction
The lecture provides a foundational overview of disease modeling, aimed at public‑health, data‑science, and AI students, and explains why models are essential for turning health data into policy decisions. It categorizes four principal model families—mechanistic (e.g., SIR), statistical (GLM), machine‑learning (random...

Lecture 3.1.11: Cloud Dashboard
The lecture explains how cloud‑based dashboards bridge the gap between sophisticated analytical models and non‑technical stakeholders. By converting Jupyter notebooks into interactive web interfaces, data scientists can deliver actionable insights with a single click, eliminating the need for stakeholders to...

Lecture 3.0.18: CICD & Cloud Cost Estimation Github Action
The lecture reframes data‑science projects as medical devices, emphasizing three pillars: a reproducible scaffold, continuous integration/deployment (CI/CD) for safety, and a realistic cloud‑cost budget. By mirroring device housing, safety circuits, and manufacturing budgets, students are urged to treat codebases with...

Lecture 4: Survival Analysis Case Study (Kaplan-Meier, Log-Rank, Cox in R)
The lecture walks through a published breast‑cancer survival study, illustrating how non‑parametric (Kaplan‑Meier, log‑rank) and semi‑parametric (Cox proportional hazards) techniques are implemented in R to handle censored time‑to‑event data. It explains why the authors chose these methods: Kaplan‑Meier for...