A DNA 'On-Off' Switch? Light and Redox Cues Reversibly Link Strands for Nanotech
•February 12, 2026
0
Why It Matters
The ability to reversibly link DNA strands on demand provides precise, stimuli‑responsive control essential for next‑generation nanomedicine and dynamic material design.
Key Takeaways
- •Thioguanosine enables light‑triggered DNA crosslinking.
- •Crosslinks reversible via redox or light without helix distortion.
- •Method preserves DNA thermal stability and native structure.
- •Enables dynamic nanomaterials and intracellular drug delivery.
- •Expands nucleic‑acid chemistry toolbox for programmable nanomachines.
Pulse Analysis
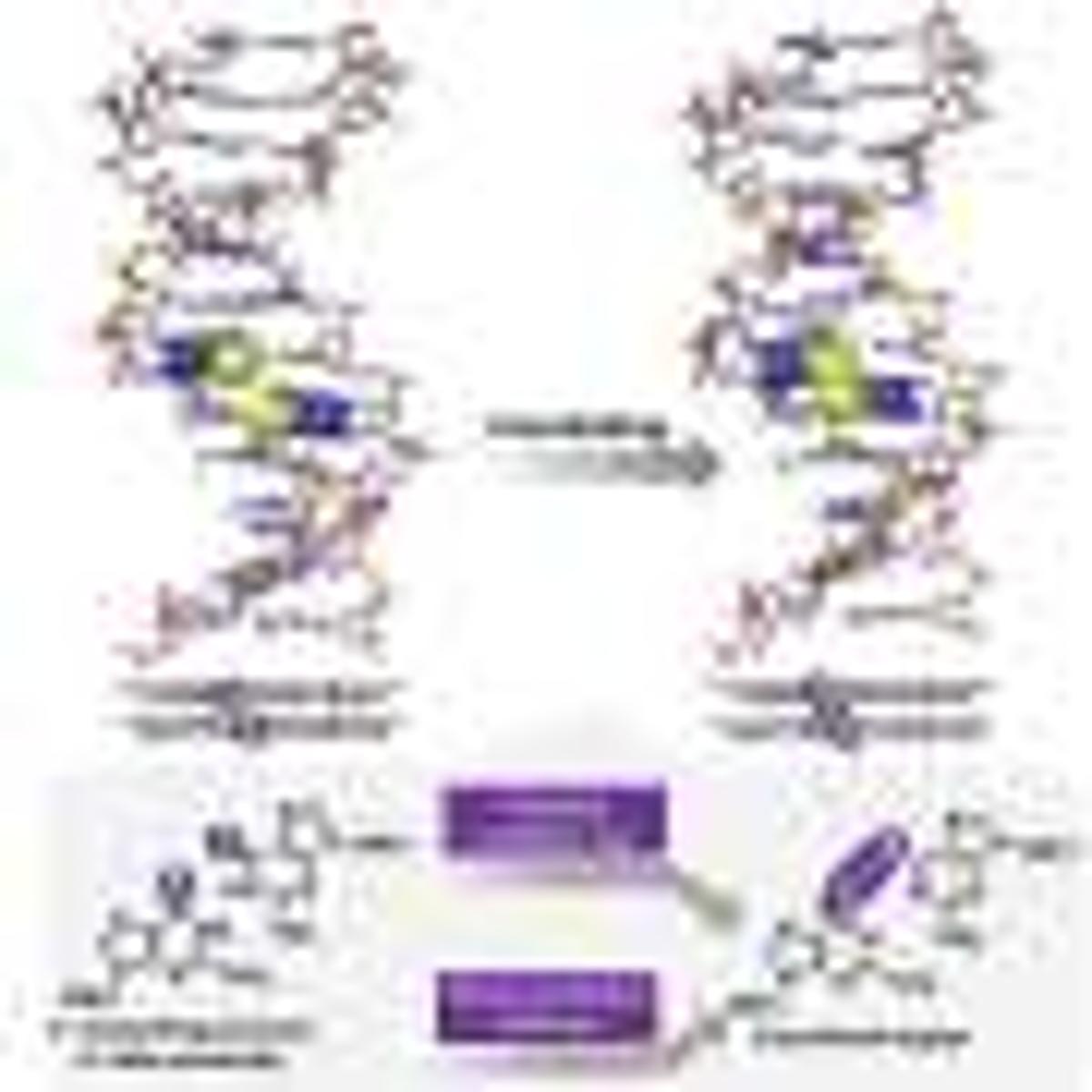
DNA’s utility is rapidly evolving from a passive genetic repository to an active engineering platform. By exploiting the predictable base‑pairing and three‑dimensional rigidity of duplexes, scientists can position reactive groups with nanometer precision. The recent discovery that thioguanosine undergoes a photo‑induced electron‑transfer reaction within the helix adds a powerful, controllable crosslinking handle, enabling chemists to lock strands together only when illumination or a mild oxidant is applied.
What sets this system apart is its reversibility. Redox agents or specific wavelengths of light can cleave the newly formed bonds, restoring the original duplex without compromising structural integrity. This dynamic behavior translates into materials that can adapt, self‑heal, or release therapeutic payloads in response to cellular cues. For drug‑delivery applications, the crosslink acts as a protective cage that disassembles inside target cells, improving payload stability and reducing off‑target effects.
Looking ahead, the thioguanosine‑based switch opens avenues for programmable DNA nanomachines that execute complex tasks, such as logic‑gated assembly or on‑demand catalysis. Integration with existing DNA origami techniques could yield smart scaffolds for biosensing, tissue engineering, and synthetic biology. As the market for nucleic‑acid therapeutics and nanomaterials expands, technologies offering precise, reversible control are poised to become foundational components of next‑generation biotech solutions.
A DNA 'on-off' switch? Light and redox cues reversibly link strands for nanotech
0
Comments
Want to join the conversation?
Loading comments...