🎯Today's Pharma Pulse
Updated 1h agoWhat's happening: Cell‑free PL‑display platform accelerates peptide screening ten‑fold
Researchers at the Innovation Center of NanoMedicine unveiled PL‑display, a cell‑free system that immobilizes individual peptides on magnetic beads, delivering more than ten‑fold efficiency gains versus traditional cell‑based displays. The method remains functional under high‑temperature, high‑salt and toxic‑protein conditions, broadening screening capabilities.
Also developing:
- •Sanofi’s rilzabrutinib receives orphan drug designation in Japan for IgG4‑related disease
- •Roche’s fenebrutinib shows positive Phase III results as first BTK inhibitor for relapsing and primary progressive multiple sclerosis (FENhance 1)
- •Lynk Pharmaceuticals announces positive Phase III topline data for zemprocitinib in moderate‑to‑severe atopic dermatitis
- •InnoCare Pharma’s next‑generation TRK inhibitor zurletrectinib receives priority review in China for pediatric solid tumors
Video•Mar 1, 2026
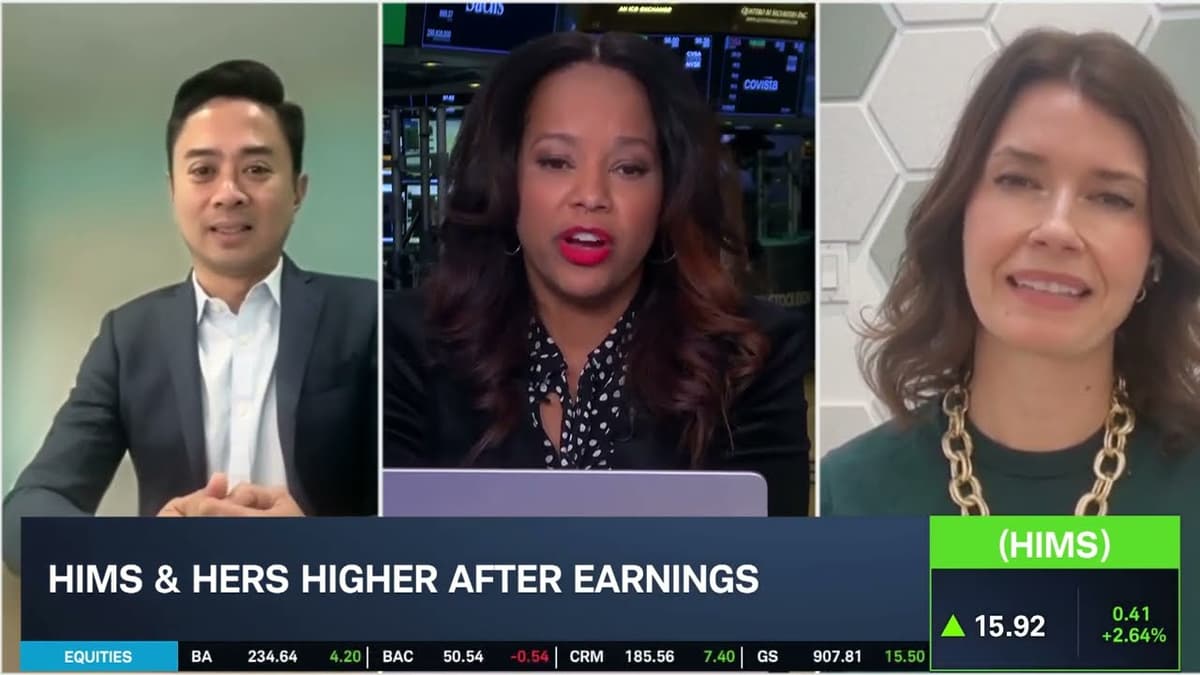
HIMS Core Business Faces Challenges in Guidance, GLP-1 Prices & NVO
Hims & Hers reported Q4 results after the bell, with mixed performance, drawing investor focus on its GLP‑1 program and forward guidance. The company posted EPS of $0.08, double expectations, but revenue of $617 million fell short of the $619 million forecast. For Q1, guidance of $600‑$625 million disappointed analysts who had modeled $654 million, reflecting pressure from GLP‑1 price erosion and regulatory scrutiny. Analysts Christine Short highlighted a 30%+ growth in the core sexual‑health and dermatology segments, noting that only a small fraction of the 2.5 million subscribers use GLP‑1. Brian Tanquilut reiterated a “hold” rating, citing the Novo Nordisk patent lawsuit and the fading GLP‑1 halo as near‑term valuation caps, while emphasizing the company’s shift toward a holistic, personalized‑medicine model. The market’s initial sell‑off has softened as investors reassess the pivot away from GLP‑1 dependence. However, ongoing legal liabilities and uncertain GLP‑1 pricing dynamics keep the stock’s upside limited, making execution of the broader telehealth strategy critical for long‑term growth.
By Schwab Network

News•Mar 1, 2026
Drug Discovery Bottleneck? Cell-Free Platform Screens Peptides Faster, Even in Harsh Conditions
Researchers at the Innovation Center of NanoMedicine unveiled PL‑display, a cell‑free platform that immobilizes individual peptides on magnetic beads for rapid screening. The method delivers over ten‑fold efficiency gains versus traditional cell‑based displays and can operate under high‑temperature, high‑salt, or...
By Phys.org – Nanotechnology
News•Mar 1, 2026
GoodRx Launches Employer Program to Help Subsidize High-Cost Brand Drugs
GoodRx introduced GoodRx Employer Direct, a new service allowing employers to directly subsidize the manufacturer‑sponsored price of high‑cost brand medications such as GLP‑1 drugs without adding them to health‑plan formularies. The model, first piloted with retailer Hy‑Vee, lets employers contribute...
By MedCity News

Podcast•Mar 1, 2026•0 min
🎧 What Investors Get Wrong About Hims & Hers, Ep. 2 with Paul Cerro
In this episode, the hosts dissect why investors misunderstand Hims & Hers' (HEMS) gross margin potential, emphasizing that as a compounder without its own API manufacturing, HEMS cannot achieve margins higher than drug manufacturers. They illustrate how HEMS' margin trajectory...
By GLP-1 Digest

Blog•Mar 1, 2026
Weekly Reads: Longevity Hype Vs. Cool Research, Losing Your Y, FDA Maelstrom
This week’s coverage juxtaposes longevity hype with emerging science, highlighting David Sinclair’s bold claim that aging could be reversed within 10‑20 years, a new Nature paper confirming adult hippocampal neurogenesis, and research showing men lose the Y chromosome as they...
By The Niche

Blog•Feb 28, 2026
Epic's First Pharma Deal
Epic Systems announced its first pharmaceutical customer, Eli Lilly, for the newly launched Epic Discovery platform. The deal marks the inaugural sale of a Health Grid product to a life‑sciences firm, allowing Lilly to streamline clinical trial creation, site identification, activation,...
By Health API Guy

News•Feb 28, 2026
In China, Phase 2 Study of Relma-Cel in R/R MCL Finds Durable Responses
A phase‑2 trial of relmacabtagene autoleucel (relma‑cel), a CD19‑directed CAR‑T therapy, enrolled 59 Chinese patients with relapsed/refractory mantle‑cell lymphoma after BTK‑inhibitor failure. The study reported a 71.2% overall response rate and a 59.3% complete response rate, with median time to...
By AJMC (The American Journal of Managed Care)

News•Feb 28, 2026
Analysis Finds Efficiencies, Savings of Using a Single Bispecific for DLBCL and FL
A new analysis quantifies the operational and financial benefits of using Genmab’s epcoritamab, a dual‑indication bispecific antibody, for both relapsed/refractory diffuse large B‑cell lymphoma (DLBCL) and follicular lymphoma (FL). In a community‑practice model of 100 patients, the study projects 3,110...
By AJMC (The American Journal of Managed Care)

News•Feb 28, 2026
Mixed Immune Signature Identified in Chronic Hand Eczema
A phase‑2b trial enrolling 94 adults with chronic hand eczema (CHE) without etiologic pre‑selection uncovered a mixed immune signature spanning type 2, type 3 and type 1 pathways. Dupilumab, an IL‑4Rα antagonist, delivered a 59.8% mean improvement in modified Total Lesion Symptom Score...
By AJMC (The American Journal of Managed Care)

News•Feb 28, 2026
GLP-1 News Galore; Top 100 Venture Investors; Doug Ingram to Step Down; and More
This week’s Endpoints Weekly highlighted a surge of GLP‑1 developments, including new trial data and expanded indications that reinforce the class’s dominance in obesity and diabetes treatment. The newsletter also released its annual Top 100 venture investors list, showing a notable...
By Endpoints News

News•Feb 28, 2026
EMA’s CHMP Recommends Three New Orphan Drugs, Rebuffs Two FDA-Approved Programs
The EMA’s Committee for Medicinal Products for Human Use (CHMP) recommended six new medicines, including three orphan drugs already approved by the FDA—Ojemda for pediatric low‑grade glioma, Palsonify for acromegaly, and Xolremdi for WHIM syndrome. The agency also gave a...
By BioCentury

News•Feb 28, 2026
Emerging Models Point to a New Operating System for Rare Disease Innovation
BioCentury’s cookie policy details five categories of cookies—strictly necessary, functional, marketing, advertising, and analytics—each serving distinct purposes on its website. Strictly necessary cookies support authentication, registration, and navigation, while functional cookies enable personalization of services. Marketing and advertising cookies help...
By BioCentury
Video•Feb 27, 2026
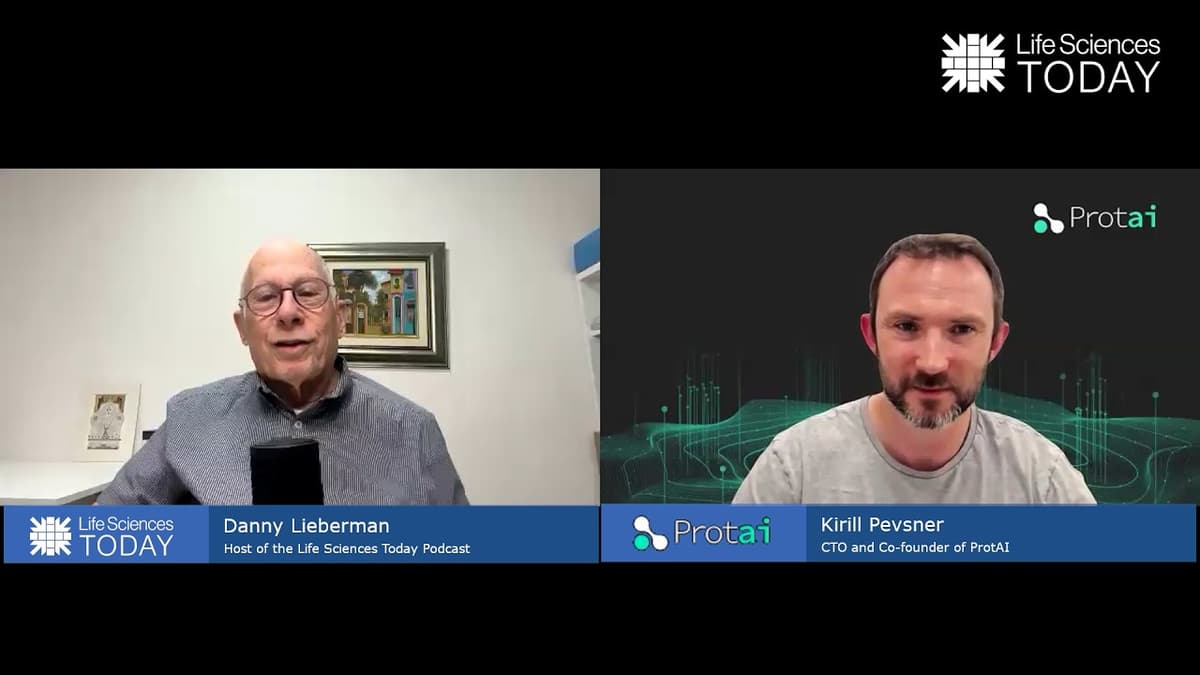
AI Insights in Pharma with Protai - Life Sciences Today Podcast Episode 50
In episode 50 of the Life Sciences Today podcast, host Danny Lieberman sits down with Carol Pesner, CTO and co‑founder of ProtAI, to discuss the company’s AI‑driven approach to drug discovery. ProtAI fuses structural proteomics with advanced artificial intelligence to...
By Healthcare IT Today

Video•Feb 27, 2026
LIVE: WHO Meeting on 2026-27 Northern Hemisphere Influenza Vaccine Composition
The World Health Organization convened an information meeting to finalize the composition of the 2026‑27 Northern Hemisphere influenza vaccine. Senior officials, including Dr. Chikui Hikawazu and Dr. Maria Van Kokov, highlighted the critical role of the Global Influenza Surveillance and...
By World Health Organization (WHO)
![[Comment] Safeguarding Genomic Integrity in Pluripotent Stem-Cell Therapies](/cdn-cgi/image/width=1200,quality=75,format=auto,fit=cover/https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/fba52b2aab9c591bf0e059f57acce9ea.webp)
News•Feb 27, 2026
[Comment] Safeguarding Genomic Integrity in Pluripotent Stem-Cell Therapies
Human pluripotent stem cells (hPSCs) are emerging as a transformative platform for Parkinson’s disease, with recent phase I/II trials showing successful engraftment of hESC‑ and iPSC‑derived dopaminergic neurons. Yet extensive data reveal that cultured hPSCs frequently acquire recurrent genetic lesions—most...
By The Lancet
![[Review] Autosomal Dominant Polycystic Kidney Disease](/cdn-cgi/image/width=1200,quality=75,format=auto,fit=cover/https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/e5c772f073be33a83118f46a1ac91f13.webp)
News•Feb 27, 2026
[Review] Autosomal Dominant Polycystic Kidney Disease
Autosomal dominant polycystic kidney disease (ADPKD) remains the most common hereditary cause of chronic kidney disease, imposing substantial morbidity, mortality, and healthcare costs worldwide. Recent advances in molecular genetics and high‑resolution imaging have sharpened diagnostic criteria and enabled more accurate...
By The Lancet

News•Feb 27, 2026
STAT+: Minnesota Report Shows Large Hospitals Continue to Dominate the 340B Drug Discount Program
Minnesota’s Department of Health reports that hospitals and clinics in the state earned at least $1.34 billion in 2024 from the 340B drug discount program. Participants received $3.045 billion in discounted medicines but paid $1.53 billion plus $165 million in administration fees. The largest...
By STAT News — Pharma

News•Feb 27, 2026
Promoters and Enhancers: Tool Catches Gene-Controlling DNA Sequences Doing Each Other's Jobs
Researchers at Cornell’s Weill Institute introduced QUASARR‑seq, a high‑throughput assay that measures promoter and enhancer activity simultaneously. The study found that most human regulatory elements can function as both promoters and enhancers, following a unified regulatory logic. A bidirectional feedback...
By Phys.org – Biotechnology

Deals•Feb 27, 2026
Cigna Acquires CarepathRx to Expand Hospital Pharmacy Services
Cigna announced the acquisition of CarepathRx, a private‑equity‑backed pharmacy that supplies prescription drugs to roughly 10% of U.S. hospitals. The deal expands Cigna’s vertical integration in the pharmaceutical supply chain, complementing its earlier purchase of Express Scripts.
STAT News — Pharma

Deals•Feb 26, 2026
Asahi Kasei to Acquire German Biotech Aicuris for €780M
Japanese drugmaker Asahi Kasei announced it will acquire German biotech Aicuris for €780 million, gaining its antiviral therapy pritelivir targeting HSV. The deal expands Asahi Kasei's anti‑infectives portfolio and supports its goal of reaching JPY 300 billion in pharma sales by 2030.
pharmaphorum

Video•Feb 26, 2026
Walk With Me: Joseph Wu, Cardiologist Studying Stem Cells and Heart Disease
Joseph Wu, a Stanford professor of medicine and radiology, leads the Stanford Cardiovascular Institute in developing patient‑specific cardiac cells derived from a person’s own blood. By reprogramming blood cells into pluripotent stem cells and then coaxing them to become beating...
By Stanford Medicine

Video•Feb 25, 2026
Malta as a Living Lab for Precision Oncology | MedTech World Middle East 2026
At MedTech World Middle East 2026, Prof. Christian Scerri unveiled a plan to turn Malta into a living lab for precision oncology, leveraging its single national health system, compact ecosystem, and EU‑aligned regulatory framework. He outlined a roadmap that includes...
By MedTech World

Video•Feb 25, 2026
GCC MedTech Boom: Market, Funding & Scaling | MedTech World Middle East 2026
At MedTech World Middle East 2026, a panel of GCC health leaders highlighted the region’s rapid shift from a pure healthcare consumer to a burgeoning hub for MedTech, biotech, and digital health innovation. National visions and transformation strategies are driving...
By MedTech World

Video•Feb 25, 2026
Citi Bearish On Dr Reddy's; CLSA Positive On HAL, Says Company Has Strongest Pipeline Of Orders
Citi’s research team issued a bearish stance on Dr Reddy’s Laboratories, citing concerns that the company’s generic version of semaglutide may not capture significant market share. In contrast, CLSA upgraded Hindustan Aeronautics Limited (HAL) to a preferred pick, highlighting its strongest...
By CNBC-TV18

Deals•Feb 25, 2026
GSK to Acquire 35Pharma for Under $1B
GlaxoSmithKline (GSK) announced the acquisition of privately held 35Pharma, a developer of a pulmonary arterial hypertension drug. The cash deal is valued at just under $1 billion, expanding GSK’s pipeline in respiratory and immunology.
STAT (Biotech)

Video•Feb 24, 2026
The Surprising Impact of Drug Price Negotiation on Clinical Trials
The podcast examines a new Health Affairs paper that tracks how the Inflation Reduction Act’s Medicare drug‑price negotiation provision has affected biopharmaceutical clinical‑trial activity. Dr. So Young Kang and co‑authors compare industry‑sponsored trial initiations from 2015‑2024, focusing on firms directly hit...
By Health Affairs

Video•Feb 24, 2026
The Conjugation Conundrum: The Realities of Conjugated LNP Manufacturing
The discussion centers on the emerging complexities of conjugated lipid nanoparticle (LNP) manufacturing, a shift from standard platform processes toward targeted RNA delivery. Sujit explains that adding a biologic ligand to LNPs forces manufacturers to revisit every process parameter—viscosity, shear...
By Life Science Connect

Video•Feb 24, 2026
The “Art” Of the Linker: Complexity, Biodegradability, and Scale in Active LNP R&D
The panel examined the emerging “active” delivery paradigm for lipid nanoparticle (LNP) therapeutics, focusing on the added layers of complexity introduced by ligand‑modified formulations and the need for robust R&D pipelines. Participants contrasted passive LNPs with active, ligand‑decorated versions, highlighting...
By Life Science Connect