
Avutometinib and Defactinib
The FDA granted accelerated approval to the oral co‑pack Avutometinib and Defactinib for adults with KRAS‑mutated, recurrent low‑grade serous ovarian cancer (LGSOC) after prior therapy. The regimen pairs a RAF/MEK inhibitor with a FAK inhibitor, marking a rare “novel‑novel” combination approval. In the Phase 2 RAMP‑201 trial, the duo achieved a confirmed overall response rate of 44%, with durations of response ranging from 3.3 to 31.1 months. This is the first therapy specifically approved for LGSOC, a distinct ovarian cancer subtype.

Module 3, Section 1: HitID Screens
The module introduces HitID screens, outlining key strategies for early-stage drug discovery. It references recent literature on medicinal chemistry optimization, successful hit‑to‑clinical transitions, DNA‑encoded library (DEL) approaches, ultra‑low‑molecular‑weight crystallographic screening, and fragment‑based drug discovery (FBDD). By consolidating these sources, the...

QPX7728
Xeruborbactam (QPX‑7728) is a broad‑spectrum β‑lactamase inhibitor designed to revive the activity of β‑lactam antibiotics against multidrug‑resistant Gram‑negative bacteria. Developed by Qpex Biopharma and Shionogi, the molecule targets both serine‑ and metallo‑β‑lactamases, addressing a key resistance mechanism. Preclinical data show...

Dose as the Ultimate MPO Endpoint
Tristan Maurer’s Flash Talk framed dose as the definitive multiparametric optimization (MPO) endpoint for small‑molecule drug design. He argued that dose integrates exposure, pharmacology, and mechanism‑driven effects, making it the linchpin for balancing potency, ADME, and safety. The presentation highlighted...

Imatinib
Imatinib (Gleevec®/Glivec®) is an oral ATP‑competitive inhibitor of the BCR‑ABL fusion tyrosine kinase, approved by the FDA in 2001 for Philadelphia chromosome‑positive chronic myeloid leukemia and other malignancies. The drug emerged from high‑throughput screening, structure‑activity relationship optimization, and structure‑based drug...

Thermal Stability Assays as Tools to De-Risk Discovery
Thermal stability assays, especially differential scanning fluorimetry, are gaining traction as early‑stage de‑risking tools in drug discovery. By measuring protein melting temperatures, these assays reveal ligand‑induced stabilization, enabling rapid hit validation and prioritization. The article outlines best‑practice workflows, data‑interpretation guidelines,...

Valbenazine
Valbenazine (Ingrezza®), an oral selective VMAT2 inhibitor from Neurocrine Biosciences, received FDA approval for treating tardive dyskinesia and Huntington’s disease‑associated chorea. In the Phase 3 KINECT‑3 trial, a once‑daily 80 mg dose produced a statistically significant reduction in AIMS dyskinesia scores after...

Zalsupindole
Delix Therapeutics announced the results of a Phase 1b study of zalsupindole, a selective 5‑HT2A receptor partial agonist, in patients with major depressive disorder. The trial, published in the January 2026 issue of ACS Chemical Neuroscience, demonstrated favorable safety, tolerability, and early...

Module 2 Quiz
Drug Hunter’s online curriculum includes a Module 2 Quiz that tests learners on target identification and validation concepts. The quiz is part of a broader course designed for drug discovery professionals and requires full platform access via subscription or sign‑in. It...

NX-1607
Nurix Therapeutics has launched NX-1607, the first orally bioavailable small‑molecule inhibitor of the immune regulator CBL‑B, into a Phase 1a/1b trial for advanced cancers. CBL‑B modulates activation of T, B and NK cells, and NX-1607 locks the protein in an inactive...
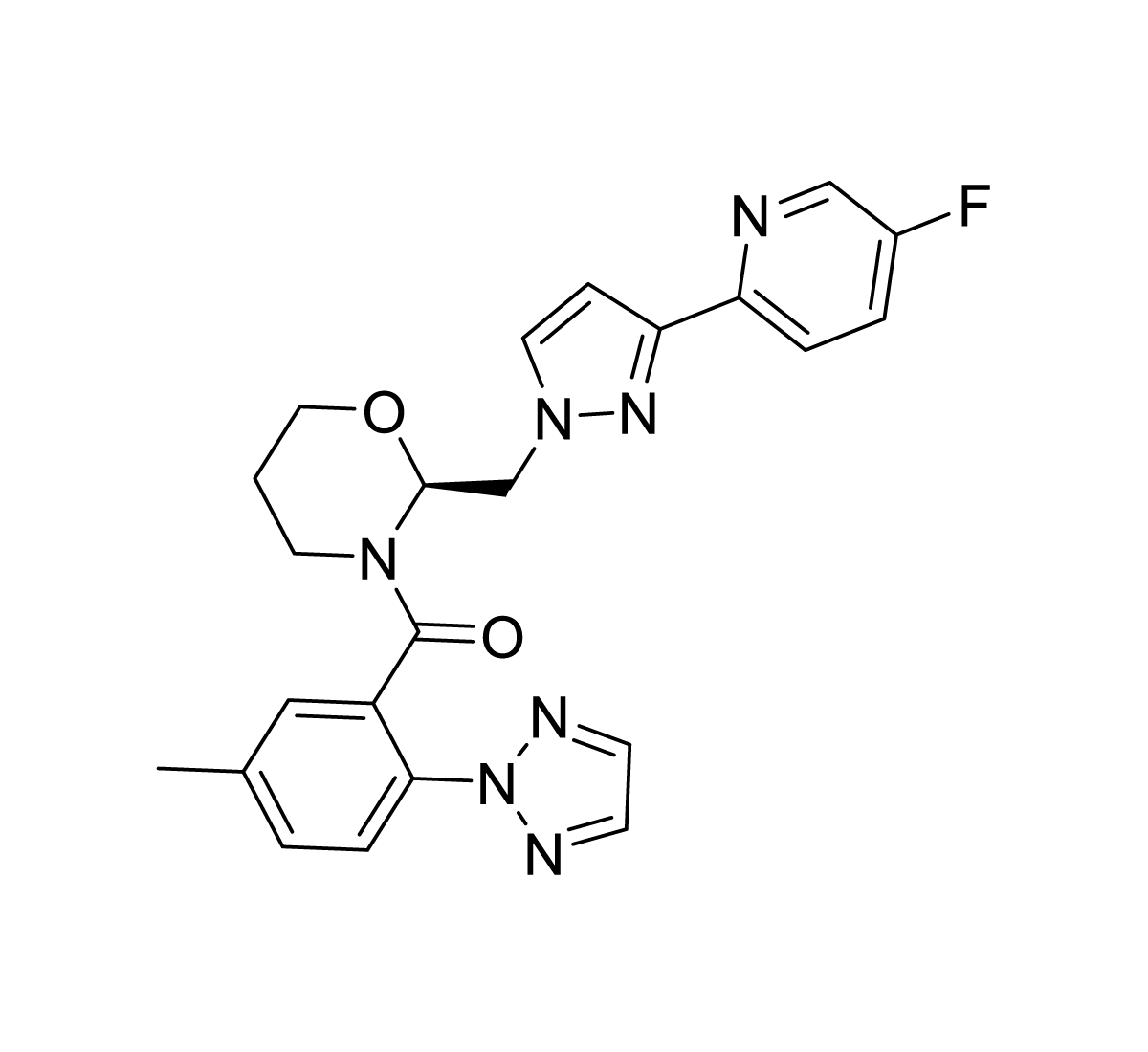
ORN0829
Taisho Pharmaceutical’s vornorexant (TS‑142), marketed as Vorzzz®, received Japanese regulatory approval in August 2025 as a dual orexin‑1/2 receptor antagonist for insomnia. The drug distinguishes itself from existing DORAs through rapid absorption and a short elimination half‑life, aiming to minimize...

Module 2, Section 3: Target Validation
The module on target validation walks through how phenotypic and target‑based strategies intersect in immune‑focused drug discovery. It highlights recent literature on TYK2 pseudokinase stabilization as a mechanism to block T‑cell signaling, and cites Icotrokinra and Deucravacitinib as successful examples....
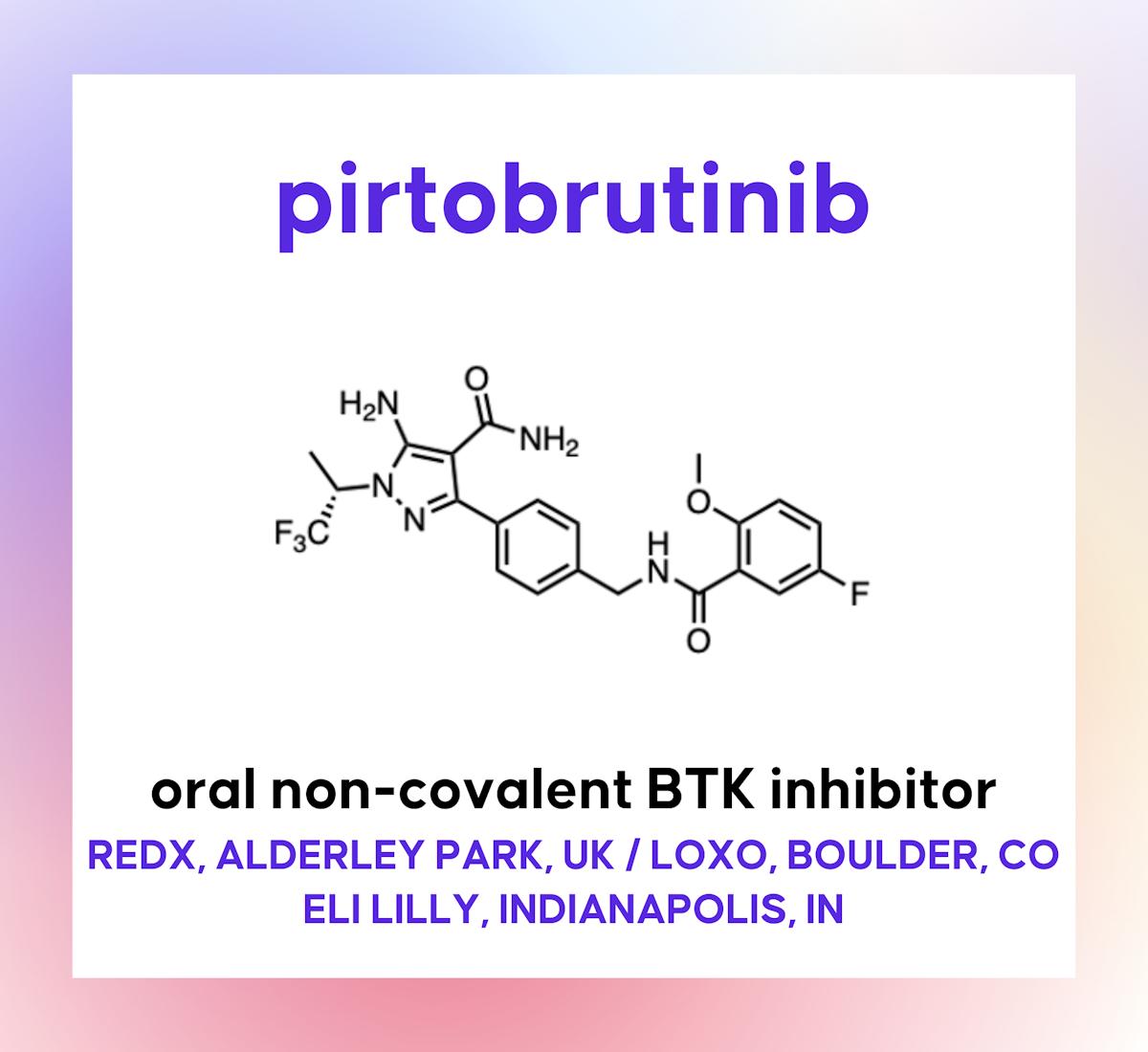
Pirtobrutinib
Late 2025 saw the FDA grant traditional approval to pirtobrutinib, an oral, reversible BTK inhibitor targeting multiple B‑cell malignancies. The drug demonstrated robust efficacy in BTK‑resistant chronic lymphocytic leukemia and small lymphocytic lymphoma, backed by positive Phase 3 data and early...

2025 Non-US Novel Large Molecule Drug Approvals
In 2025, Europe’s EMA, China’s NMPA, and Japan’s PMDA each granted first‑time approvals for novel large‑molecule therapeutics. Oncology accounted for the largest share of these approvals, while endocrinology—driven largely by GLP‑1‑based agents—was the second biggest category. The approvals span a...

INN-Coming: Insights on the Industry’s Latest Disclosures
The WHO’s INN proposed list 134, released in early 2026, reveals several late‑stage drug candidates that were previously hidden from public view. Notably, two NLRP3 inhibitors—abdenoflast and parunoflast—appear to map to Eli Lilly’s newly acquired Ventyx assets VTX2735 and VTX3232, both showing promising...

Module 2, Section 2: The Druggable Interactome
The Module 2, Section 2 lecture introduces the druggable interactome, compiling key resources that map the human druggable genome, protein expression, kinase families, transcription‑factor proteomics, GPCRs, and ion‑channel complexes. It highlights quantitative estimates—over 3,000 proteins deemed druggable and hundreds of actionable kinases—while...

January 2026 Patent Highlights
The January 2026 Patent Highlights roundup spotlights a wave of new intellectual‑property activity across several cutting‑edge drug discovery areas. Notable filings include lysine‑directed covalent inhibitor chemotypes, strategies to balance potency with drug‑like properties, refined target‑selection frameworks, dozens of Polθ synthetic‑lethal patents...

Dotinurad (FYU-981)
Dotinurad (FYU‑981), marketed as Urece®, is a URAT1 inhibitor approved for gout and hyperuricemia in Japan and China. The drug was chemically refined from the older uricosuric benzbromarone to retain potency while eliminating rare hepatotoxic events. Crystalys Therapeutics is now...

Karma-Karma-Karma Chameleon
Balancing potency with oral bioavailability remains a core hurdle as drug candidates grow larger and more complex. Researchers now focus on "chameleonicity"—the ability of a molecule to toggle between polar and lipophilic conformations—to reconcile solubility and permeability. The article outlines...

Module 2, Section 1: Target Selection Strategy
The module outlines a five‑dimensional framework for target selection, linking biological relevance, drugability, disease impact, competitive landscape, and development risk. It contrasts first‑in‑class and best‑in‑class strategies, highlighting how pioneering mechanisms can command premium market positions. The content identifies oncology as...

Tradipitant
Vanda Pharmaceuticals received FDA approval for tradipitant (Nereus®), an oral selective NK1 receptor antagonist, to treat motion‑induced nausea and vomiting. The approval marks the first new drug for motion sickness in more than four decades, highlighting a significant regulatory milestone....

Aceclidine
Aceclidine (Vizz®) received FDA approval in 2025 as an ophthalmic solution for presbyopia, targeting age‑related near‑vision loss. The drug acts as a pupil‑selective muscarinic agonist, inducing miosis without significant ciliary muscle activity, thereby enhancing depth of focus through a pinhole...

Safer CNS Drugs with BrainOnly Pharmacology
The article outlines a growing strategy to develop central nervous system (CNS) therapeutics that remain pharmacologically active only within the brain, termed "BrainOnly" pharmacology. By leveraging selective transport mechanisms, pro‑drug designs, and peripheral clearance pathways, researchers aim to minimize off‑target...

Making a MASH Hit: PNPLA3 and the Rise of Genotype-Driven Therapies
The lipid serine hydrolase PNPLA3, especially its I148M mutant, has emerged as a genetically validated driver of MASLD/MASH, prompting a wave of genotype‑focused drug programs. RNA‑based modalities—Arrowhead’s GalNAc‑siRNA ARO‑PNPLA3 and AstraZeneca/Ionis’ GalNAc‑ASO AZD2693—are in clinical trials aiming to lower mutant...

Vimseltinib
Deciphera Pharmaceuticals received FDA approval for vimseltinib, an oral, selective CSF1R kinase inhibitor, to treat tenosynovial giant cell tumor (TGCT). The drug emerged from structure‑based drug design (SBDD) and extensive SAR optimization, as reported in Bioorganic & Medicinal Chemistry Letters...

The New WIZ-Kid in Protein Degradation
Targeted protein degraders that eliminate the transcription factors WIZ and ZBTB7A are shown to raise fetal hemoglobin (HbF) levels, offering a disease-modifying strategy for sickle cell disease and β‑thalassemia. Novartis disclosed a series of CRBN‑based glue degraders, culminating in the...

Module 1 Quiz
Drug Hunter has launched a Module 1 Quiz to evaluate learners’ grasp of introductory drug‑discovery concepts. The quiz spans all sections of the first module and is accessible through the platform’s subscription model. Users can take the assessment after reviewing the...

Identification Methods for Drug Repurposing: Case Studies in Neurodegeneration
The article outlines modern methods for identifying drug repurposing opportunities, focusing on neurodegenerative diseases such as Alzheimer’s and Parkinson’s. It highlights computational screening, network pharmacology, and real‑world evidence as core techniques, and presents case studies where existing drugs showed disease‑modifying...

It Takes Multiple to Mambo
Recent disclosures illustrate how multimeric design is reshaping drug discovery in the beyond‑Rule‑of‑5 (bRo5) space. GSK’s dimeric BET inhibitor GSK785 uses a rigid diazaspiro linker to achieve >30‑fold BRD4 selectivity, while Eli Lilly’s muvalaplin dimer‑to‑trimer architecture delivers a 10,000‑fold potency boost...

Section 4: The All-Important Dose
The blog post emphasizes dose as a pivotal factor distinguishing first‑in‑class from next‑generation therapeutics. It links total dose and dosing regimen to core drug attributes such as potency, pharmacokinetics, and safety. The discussion covers route of administration, dosing schedules, and...

Lanifibranor (IVA-337)
Lanifibranor (IVA-337) is an oral pan‑PPAR agonist entering Phase 3 trials for metabolic dysfunction‑associated steatohepatitis (MASH/MASLD). The molecule was optimized from high‑throughput screening and its preclinical data were published in the Journal of Hepatology in June 2025. Inventiva Pharma is leading development,...

2025 Novel Large Molecule FDA Drug Approvals
In 2025 the FDA approved fifteen novel large‑molecule therapies, representing 33 % of all new drug approvals that year. The slate was dominated by oncology biologics, including five antibody‑drug conjugates and bispecific antibodies, while non‑cancer indications saw first‑in‑class agents for hereditary...

December 2025 Patent Highlights
Drug Hunter’s December 2025 patent roundup spotlights four high‑impact filings: Dark Blue Therapeutics’ MLLT1/3 degraders, Insilico Medicine’s KRAS(G12V) inhibitors with demonstrated in‑vivo efficacy, Biohaven’s TRPM2 antagonists for pain, and Rome Therapeutics’ LINE‑1 reverse‑transcriptase prodrugs for oncology. These disclosures reflect rapid...

Hepatotoxicity Headaches: One of the Hardest Risks to De-Risk
Drug‑induced liver injury (DILI) now accounts for roughly 22 % of safety‑related clinical trial failures and one‑third of post‑approval market withdrawals, making it a critical bottleneck in pharmaceutical development. The blog highlights three recent de‑risking efforts: BMS’s LPA1 antagonist program that...

Section 3: Ligand Binding Basics
The post outlines core principles of ligand‑target binding essential for drug discovery, emphasizing how small molecules engage protein sites. It highlights the interplay of binding kinetics, thermodynamics, and inhibition mechanisms in shaping potency. The author explains how affinity can be...

2025 Novel Small Molecule FDA Drug Approvals
In 2025 the FDA approved 31 novel small‑molecule drugs, representing 67% of all new drug launches and a slight decline from the previous year. Oncology dominated the portfolio with nine approvals, while 29% were first‑in‑class therapies and 22% received accelerated...

Ziftomenib
Ziftomenib (Komzifti®), an oral menin‑KMT2A inhibitor, received FDA approval in November 2025 for adults with relapsed or refractory acute myeloid leukemia (AML) harboring NPM1 mutations. The drug blocks the menin‑KMT2A protein interaction, disrupting leukemogenic transcription programs. In the Phase 1/2 KOMET‑001...

Aficamten (CK-3773274)
Cytokinetics’ aficamten (Myqorzo®) received FDA approval as an oral cardiac myosin inhibitor for obstructive hypertrophic cardiomyopathy (oHCM). The drug is a next‑generation, reversible, allosteric inhibitor designed to simplify dose‑titration compared with the existing agent mavacamten. In a Phase 3 trial, aficamten...

Delgocitinib
In July 2025, the FDA granted approval to delgocitinib (Anzupgo®), a topical pan‑JAK inhibitor, for adult chronic hand eczema unresponsive or unsuitable for corticosteroids. Developed by Japan Tobacco and Leo Pharma, the formulation delivers the drug directly to affected skin,...

Acoltremon (AR-15512)
Alcon, Aerie Pharmaceuticals, and Avizorex Pharma announced FDA approval of acoltremon (Tryptyr®), the first‑in‑class TRPM8 thermoreceptor agonist for dry eye disease. The ophthalmic solution activates corneal nerves to boost natural tear production, offering a novel mechanism compared with existing anti‑inflammatory...

Treosulfan (NSC 39069)
Treosulfan (Grafapex®) received FDA approval in 2025 when combined with fludarabine as a conditioning regimen for allogeneic stem‑cell transplantation (alloHSCT) in patients aged one year and older with acute myeloid leukemia (AML) or myelodysplastic syndromes (MDS). The approval follows trials...

Camizestrant
Camizestrant is an oral, selective estrogen‑receptor‑alpha (ERα) degrader developed by AstraZeneca for ER‑positive, HER2‑negative, ESR1‑mutated breast cancer. The drug advanced to a Phase 3 trial, with results published in the New England Journal of Medicine in June 2025. The study demonstrated a...

Top 10 Most Popular Drug Hunter Resources of 2025
Drug Hunter’s 2025 resource roundup highlights the ten most accessed tools that support daily drug‑discovery workflows. The suite includes practical PK calculators, bioisostere and covalent inhibitor cheat sheets, a searchable structure and patent database, and a series of 25 Flash...

A New Approach to Drugging MYC: The Application of Translation-Inhibiting Interdictors To MYC-Driven Malignancies
Researchers have unveiled a novel class of translation‑inhibiting interdictors that selectively suppress MYC protein production, offering a viable strategy to target MYC‑driven malignancies. These small‑molecule agents disrupt the initiation of mRNA translation, leading to rapid depletion of MYC levels in...

Top 10 Most Popular Drug Hunter Reviews of 2025
2025 drug discovery emphasized rigorous target selection, with genetics‑driven programs outpacing unvalidated approaches. Immunology saw a breakthrough as small‑molecule degraders and inhibitors of transcription factors IRF5 and STAT6 demonstrated oral activity, challenging the dominance of biologics. Emerging modalities such as...

Blocking the IgG Highway: FcRn Biology, Approved Agents, and the Small Molecule Horizon
The article reviews the role of the neonatal Fc receptor (FcRn) in extending IgG half‑life and outlines how therapeutic blockade of this pathway can reduce pathogenic antibodies in autoimmune diseases. It surveys the currently approved FcRn‑targeting biologics, such as efgartigimod...

Zoliflodacin (ETX-0914)
Zoliflodacin (ETX-0914) received FDA approval as an oral bacterial type II topoisomerase inhibitor for the treatment of urogenital gonorrhea. The drug emerged from rational design of earlier inhibitors and is the product of a partnership among AstraZeneca, Entasis, Innoviva and GARDP....

Top 10 Most Popular Drug Hunter Case Studies of 2025
2025 proved pivotal for drug discovery, with the FDA approving 44 new therapies and several breakthrough candidates advancing to late‑stage trials. The most‑read case studies highlighted oral macrocycles, innovative PK engineering, and first‑in‑class modalities such as the pan‑RAS glue daraxonrasib...

Gepotidacin
Gepotidacin (Blujepa®), an oral bacterial type II topoisomerase inhibitor developed by GSK, received approval in April 2025 for uncomplicated urinary‑tract infections and gonorrhea. The drug emerged from an unbiased antibacterial screening program and represents the first new oral class of antibiotics targeting...

November 2025 Patent Highlights
The November 2025 Patent Highlights post serves as a gateway to Drug Hunter’s most‑read resources, including top‑10 lists of popular articles, reviews, and case studies from the year. It spotlights a detailed review of FcRn biology and the push toward oral...