
Discovery of a Small Molecule HPK1 Inhibitor for Immuno-Oncology
A biotech firm has disclosed a novel small‑molecule inhibitor of hematopoietic progenitor kinase 1 (HPK1) that demonstrates potent immuno‑oncology activity in preclinical models. The compound achieves sub‑micromolar potency, oral bioavailability, and drives up to 70% tumor regression when combined with anti‑PD‑1 antibodies in mice. Early IND‑enabling studies are slated to begin later this year, positioning the drug for a potential first‑in‑class entry into the rapidly expanding checkpoint‑enhancement market. The discovery highlights a new avenue for augmenting T‑cell activation beyond existing checkpoint inhibitors.

AACR San Diego 2026: New Drugs on the Horizon
The AACR 2026 Annual Meeting in San Diego unveiled 11 first‑time disclosed oncology candidates spanning small‑molecule degraders, bispecific antibodies, T‑cell engagers and ADCs. Highlights include NEO‑811, a CRBN‑mediated molecular glue targeting HIF‑1β for VHL‑deficient renal cancer, and AZD8359, a STEAP2‑directed T‑cell...
BMS-986482
Bristol Myers Squibb disclosed BMS-986482, a CRBN‑mediated degrader that targets the IKZF1‑4 transcription factors, at the ACS Spring 2026 First‑Time Disclosures session. The molecule entered a combined Phase 1/2 study aimed at patients with advanced solid tumors, marking BMS’s entry into...

BHV-2100
Researchers from KU Leuven, CISTIM Leuven and Biohaven Therapeutics have announced that an oral TRPM3 antagonist has entered Phase 2 clinical testing for the acute treatment of migraine. The program leveraged a cell‑based high‑throughput screen of more than 200,000 compounds to...

Orforglipron
Orforglitron, an oral non‑peptide GLP‑1 receptor partial agonist developed by Eli Lilly and Chugai, received FDA approval for chronic weight management. The drug distinguishes itself from oral semaglutide by requiring no fasting or special dosing constraints, enabling once‑daily administration. Clinical trials...

Module 3 Quiz
Drug Hunter’s online learning platform has released a Module 3 quiz covering the Hit Discovery section of its pharmaceutical curriculum. The quiz is part of a broader, subscription‑based course that guides users through early‑stage drug‑target identification. Learners must sign in or...
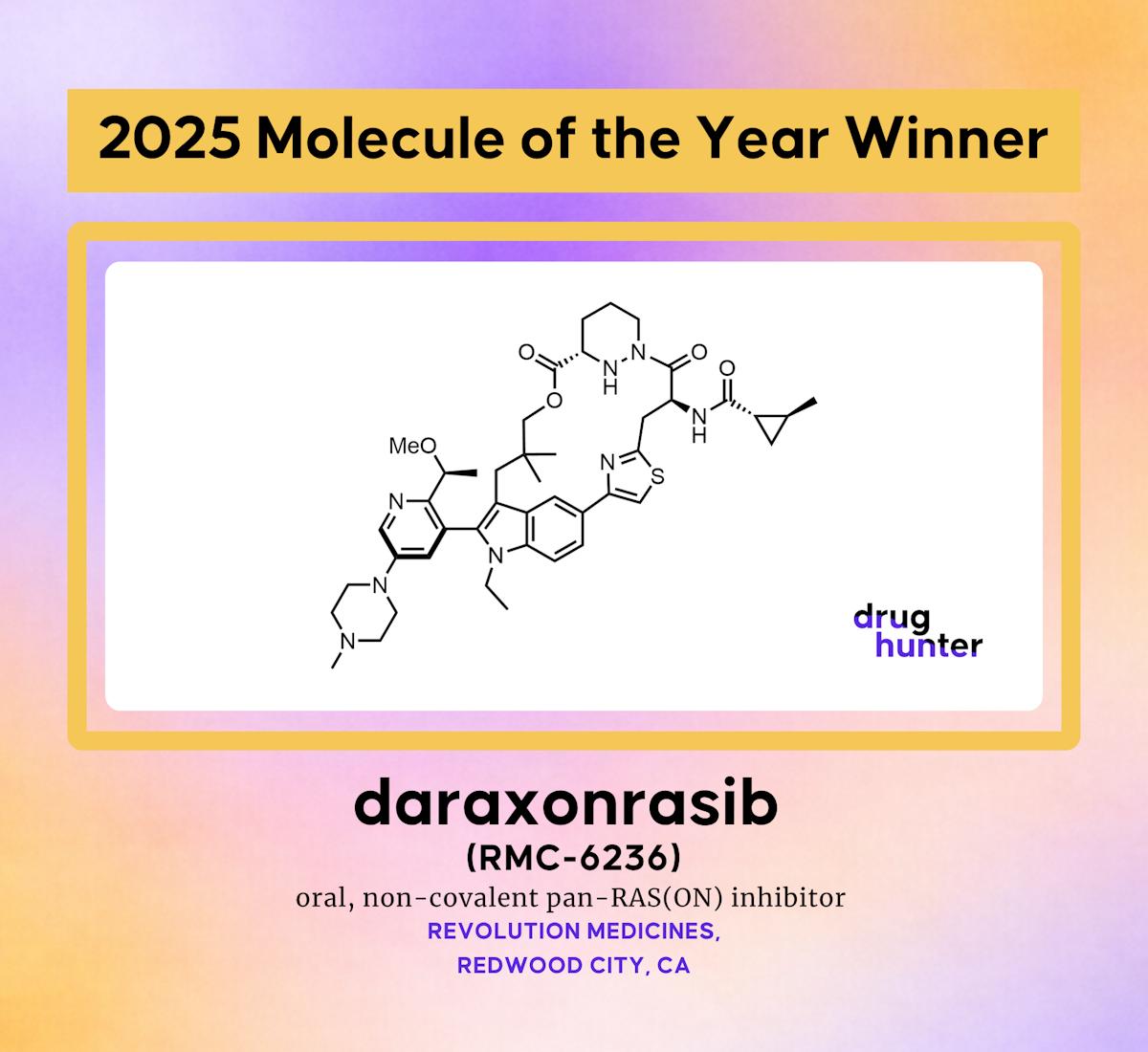
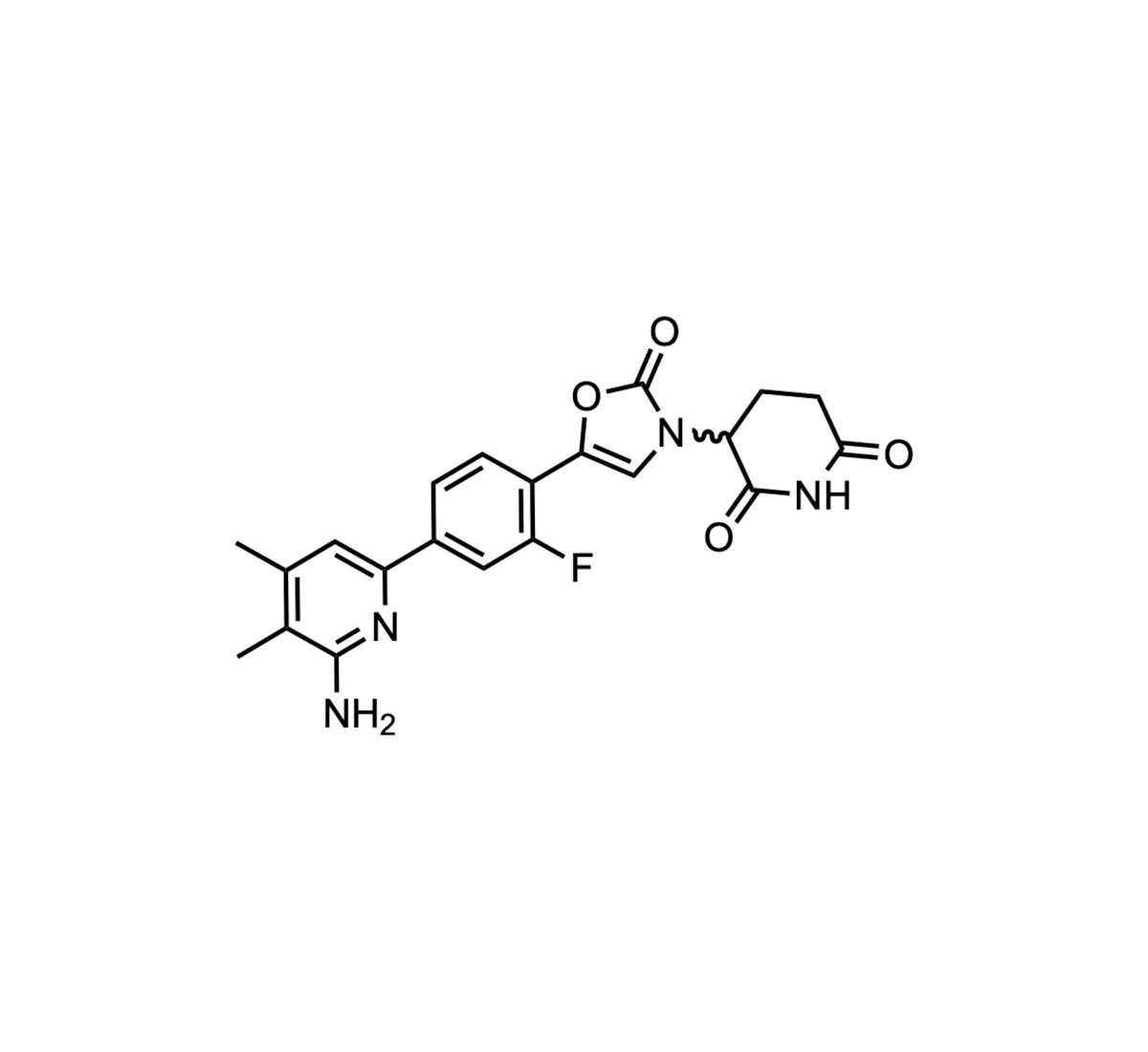
Daraxonrasib (RMC-6236): The 2025 Molecule of the Year
Revolution Medicines’ daraxonrasib (RMC‑6236) was crowned 2025 Molecule of the Year after winning 50% of community votes. The oral, tri‑complex molecular glue inhibitor uniquely targets the active GTP‑bound state of KRAS, NRAS and HRAS, covering both mutant and wild‑type isoforms....
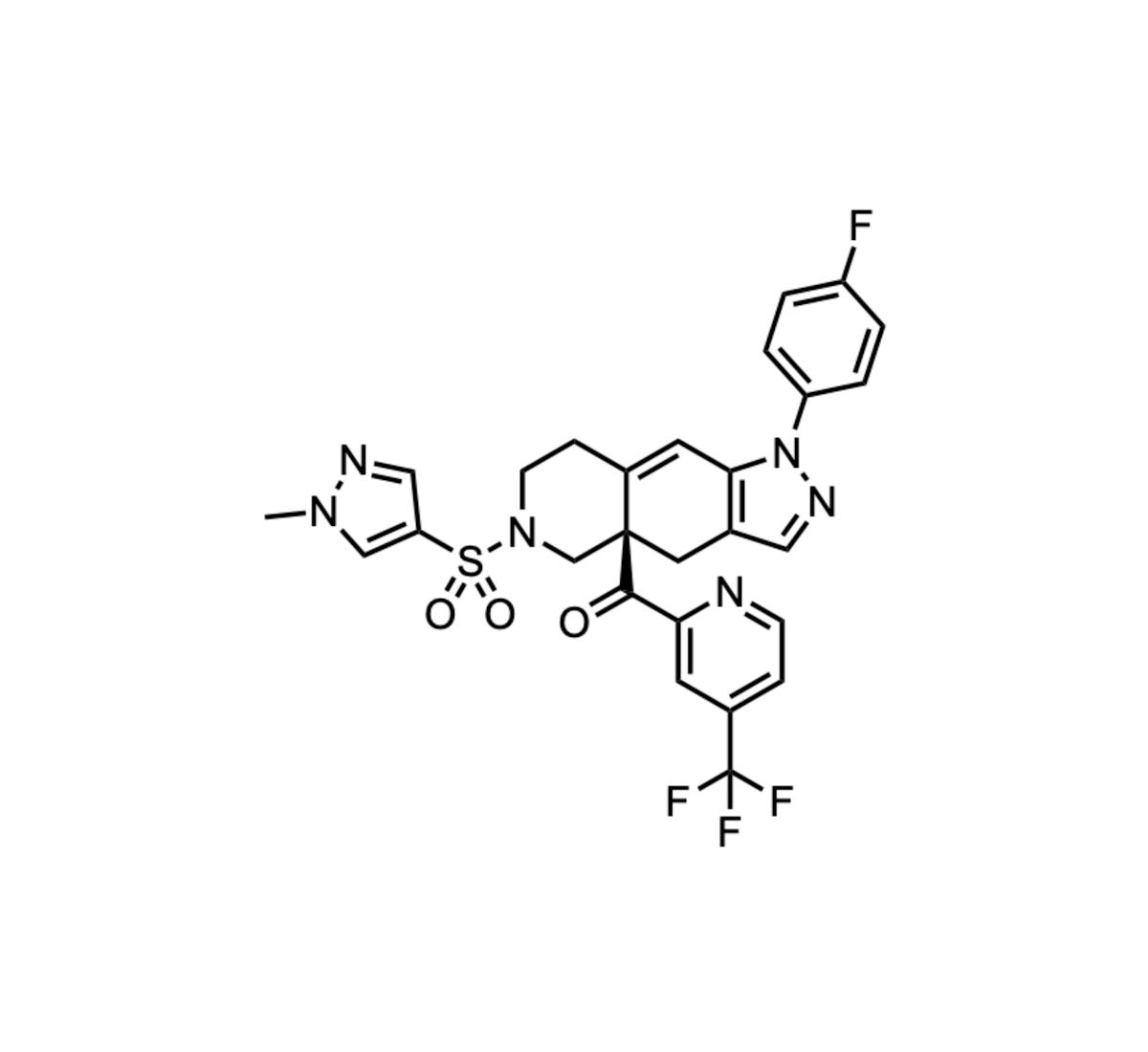
Relacorilant (CORT125134)
Corcept Therapeutics received FDA approval for relacorilant, branded Lifyorli, in combination with nab‑paclitaxel for platinum‑resistant ovarian cancer. The oral agent is a selective glucocorticoid‑receptor antagonist that blocks cortisol signaling without binding other steroid receptors, differentiating it from older cortisol‑pathway drugs....

TNO155
TNO155, also known as batoprotafib, is an oral, allosteric SHP2 inhibitor that stabilizes the phosphatase in its inactive conformation. Developed by Novartis in Cambridge, MA, it emerged from a 1.5 million‑compound high‑throughput screen combined with structure‑based drug design, becoming the first...

Quemliclustat
Quemliclustat (AB680) is a highly potent (5 pM) selective CD73 inhibitor that completed a Phase I trial in healthy volunteers, demonstrating a pharmacokinetic profile suitable for biweekly intravenous dosing. Early clinical data showed promising activity, prompting a successful Phase II study in pancreatic...

Module 3, Section 2: Quality Not Quantity
The article emphasizes a shift in high‑throughput screening toward curated, high‑quality compound libraries rather than sheer volume. It cites literature on global pharmacological mapping that shows enhanced hit relevance when nonspecific inhibitors are minimized. Phenotypic versus target‑based discovery is highlighted...

Soquelitinib
Corvus Pharmaceuticals announced soquelitinib (CPI‑818), an oral covalent inhibitor that irreversibly engages ITK at Cys442 while sparing the related kinase RLK. The selectivity addresses the broader off‑target activity seen with earlier covalent ITK agents such as ibrutinib. Soquelitinib is currently...

Asundexian
Bayer’s oral factor XIa inhibitor asundexian (BAY 2433334) has delivered positive Phase 3 data in the OCEANIC‑STROKE trial, positioning it as a potential first‑in‑class therapy for secondary stroke prevention. The drug aims to block pathological clot formation while minimizing the bleeding complications common...

A Deep Dive Into INN Proposed List 134
The World Health Organization released its International Nonproprietary Names (INN) Proposed List 134, introducing 124 new drug names slated for future approval. The list features a notable influx of antiviral and oncology agents, as well as the first biosimilar designations...

AZD5004
Elecoglipron (ECC5004/AZD5004), an oral small‑molecule GLP‑1 receptor agonist, completed Phase 2 trials in type 2 diabetes and obesity, meeting primary endpoints in the SOLSTICE and VISTA studies. AstraZeneca licensed global rights from Eccogene for an upfront payment of $185 million and potential milestones...