11th Annual Rare Disease Day | Advancing a Divalent siRNA for Prion Disease: An Investigator-Initiated Program
•February 25, 2026
0
Why It Matters
The divalent siRNA breakthrough could deliver the first disease‑modifying therapy for prion disorders, signaling a scalable approach for other protein‑misfolding diseases. It also demonstrates how nonprofit accelerators can fast‑track rare‑disease drug development, attracting investment and regulatory attention.
Key Takeaways
- •Rare diseases affect 1 in 10 North Americans
- •Only 500 of 8,000 genes have treatments
- •Event highlighted divalent siRNA for prion disease
- •Ladders to Cures applies functional genomics to rare diseases
- •Termeer Institute connects innovators to accelerate cure development
Pulse Analysis
Rare Disease Day, now in its 11th year, serves as a global platform to spotlight conditions that collectively affect one in ten people in North America. The 2026 event, co‑hosted by the Broad Institute’s Ladders to Cures Scientific Accelerator and the Termeer Institute, gathered leading researchers, clinicians, and patient advocates to discuss breakthroughs across neurodegenerative and genetic disorders. Speakers such as Anna Greka and Hilary Eaton underscored the widening gap between the 8,000 known disease‑causing genes and the fewer than 500 therapies currently approved, emphasizing the urgency for novel therapeutic strategies.
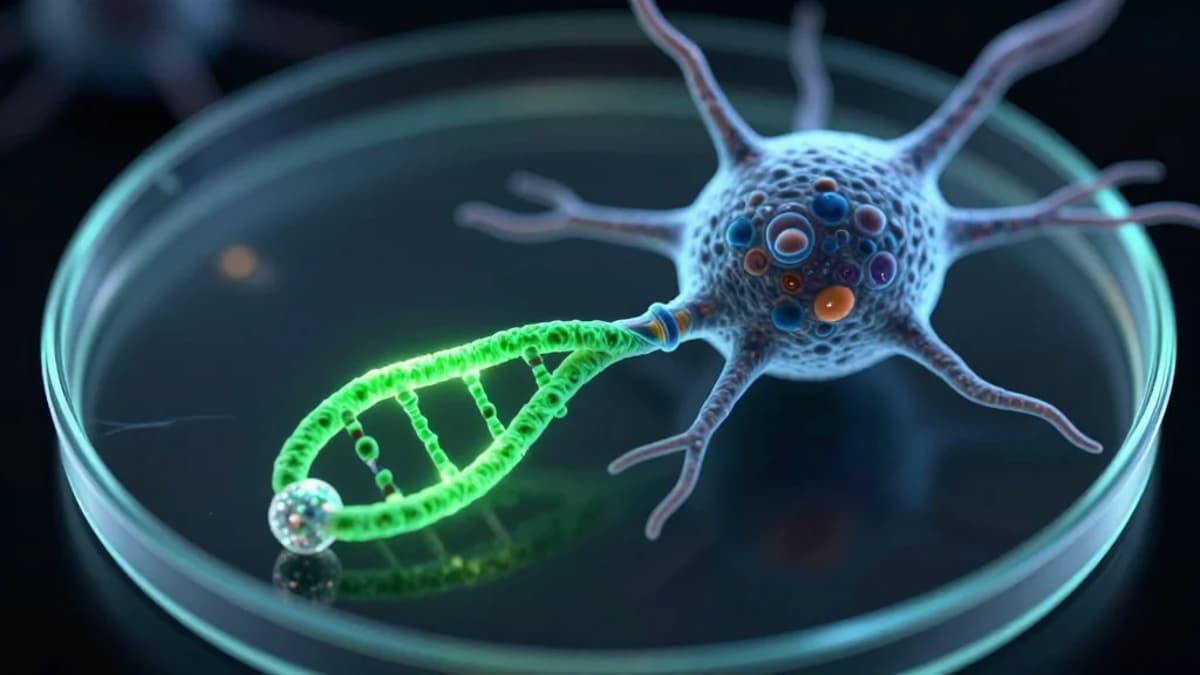
Among the announcements, an investigator‑initiated program unveiled a divalent small‑interfering RNA (siRNA) designed to silence the pathogenic prion protein. By targeting two adjacent sites on the messenger RNA, the divalent construct promises greater binding affinity and reduced off‑target effects compared with conventional monovalent siRNAs. Early preclinical data suggest the approach can halt prion replication in cellular models, offering a potential disease‑modifying therapy for a class of neurodegenerative disorders that has long eluded effective treatment. If translated to humans, this platform could be adapted to other protein‑misfolding diseases such as Alzheimer’s and Parkinson’s.
The collaboration between Ladders to Cures and the Termeer Institute illustrates a growing trend of nonprofit‑driven accelerators that blend functional genomics, computational biology, and chemical biology to de‑risk early‑stage projects. By providing shared infrastructure, data pipelines, and access to Broad Institute expertise, these programs accelerate candidate selection and de‑risk investment for biotech firms and venture capitalists. For the broader rare‑disease market, the successful development of a divalent siRNA could unlock a new therapeutic modality, attract sizable funding rounds, and stimulate regulatory pathways that prioritize precision medicines for underserved patient populations.
11th Annual Rare Disease Day | Advancing a Divalent siRNA for Prion Disease: An Investigator-Initiated Program
0
Comments
Want to join the conversation?
Loading comments...