Artificial Kinetochores Take the Pressure Off Aging Chromosomes During Meiosis
•March 11, 2026
0
Why It Matters
Age‑related cohesion loss drives infertility and miscarriage; a molecular tool that safeguards chromosome segregation could transform reproductive medicine.
Key Takeaways
- •Artificial kinetochores reduce microtubule pulling force on chromosomes
- •Weakened cohesin in aged oocytes leads to segregation errors
- •Protein assemblies expressed in mouse oocytes normalize chromosome distribution
- •Strategy could improve fertility outcomes for older parents
- •Clinical translation requires human‑relevant animal models
Pulse Analysis
Meiotic errors increase dramatically with maternal age as cohesin complexes that hold sister chromatids together deteriorate. This loss of cohesion compromises the fidelity of chromosome segregation, raising the risk of aneuploid embryos and infertility. Understanding the molecular underpinnings of this decline has been a priority for reproductive biologists seeking interventions that can preserve oocyte quality in older women.
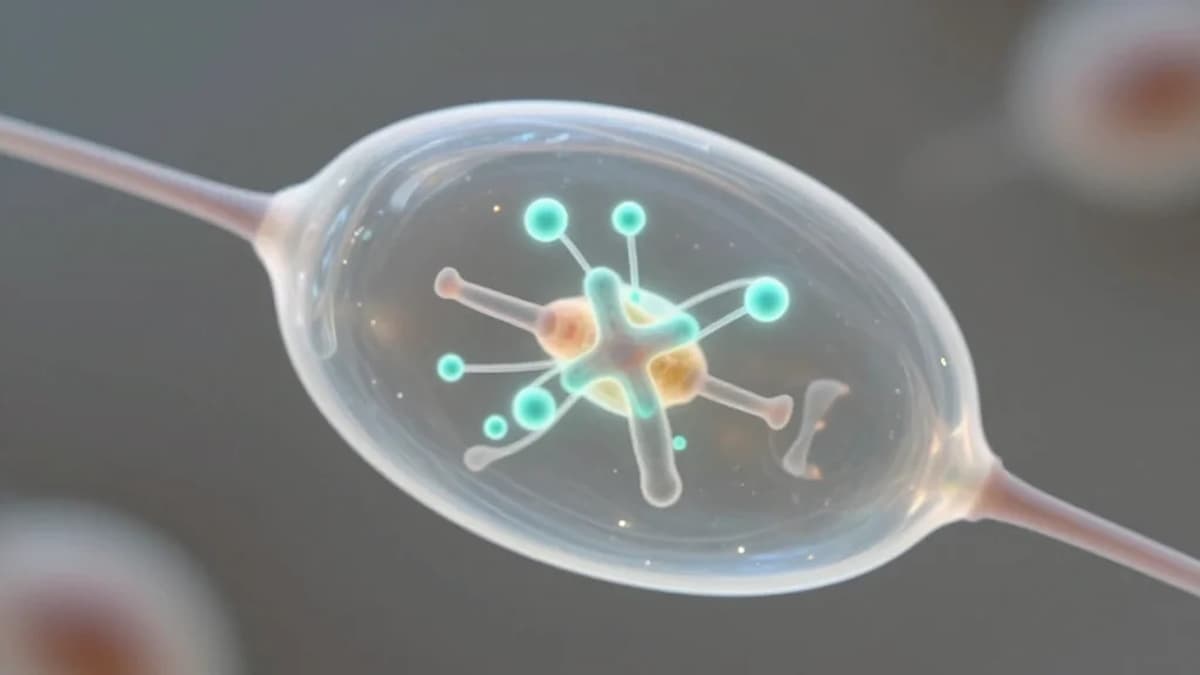
The RIKEN team’s artificial kinetochores are compact protein assemblies that can be genetically encoded and produced inside oocytes. By presenting additional binding sites for spindle microtubules, they dilute the pulling force exerted on each chromosome pair. In aged mouse oocytes, this reduction prevents premature chromatid separation, allowing even weakened sister chromatids to remain coupled through both meiotic divisions. The result is a marked improvement in the accuracy of DNA distribution without altering the native kinetochore machinery.
If translated to humans, this technology could become a cornerstone of fertility preservation for advanced‑age parents, complementing existing assisted‑reproductive techniques. However, significant hurdles remain, including the development of animal models that faithfully mimic human oocyte biology and rigorous safety assessments. Future research will need to address delivery mechanisms, long‑term effects, and regulatory pathways before artificial kinetochores can move from the laboratory to the clinic.
Artificial kinetochores take the pressure off aging chromosomes during meiosis
0
Comments
Want to join the conversation?
Loading comments...