
Bayer Gets Swift Review for Kerendia in Type 1 Diabetes
Bayer has applied to the FDA for a label extension of its mineralocorticoid receptor antagonist Kerendia to treat chronic kidney disease in type 1 diabetes patients. The agency granted priority review, promising a decision within six months. Phase 3 FINE‑ONE data showed a 34 % reduction in urinary albumin‑to‑creatinine ratio versus 12 % for placebo, alongside improved glomerular filtration. Kerendia’s sales jumped 84 % to $318 million in Q1, positioning it to cross the $1 billion blockbuster threshold this year.

Novo Nordisk Fight with KBP Bio Will Go to Arbitration
Novo Nordisk has taken its $830 million damages claim against Singapore biotech KBP Bio to arbitration after the Singapore Court of Appeal upheld an injunction freezing KBP and founder Dr Huang Zhenhua's assets. The dispute stems from a $1.3 billion licensing deal for...

Doctors Dismayed by Report of NHS AI Proposal
The NHS in England is revising its 2023 workforce plan, slashing annual staffing growth to between 1.1% and 2% and betting on artificial intelligence to offset doctor shortages. The proposal, drafted under former health secretary Wes Streeting and awaiting sign‑off...

Tuneable Peptide Biotech Parabilis Files IPO
Parabilis Medicines, the Cambridge‑based tuneable peptide biotech, filed a Nasdaq prospectus to raise roughly $100 million in an IPO under the PBLS ticker. The offering follows a $305 million private round and a multi‑billion‑dollar alliance with Regeneron, which contributed $50 million upfront and...
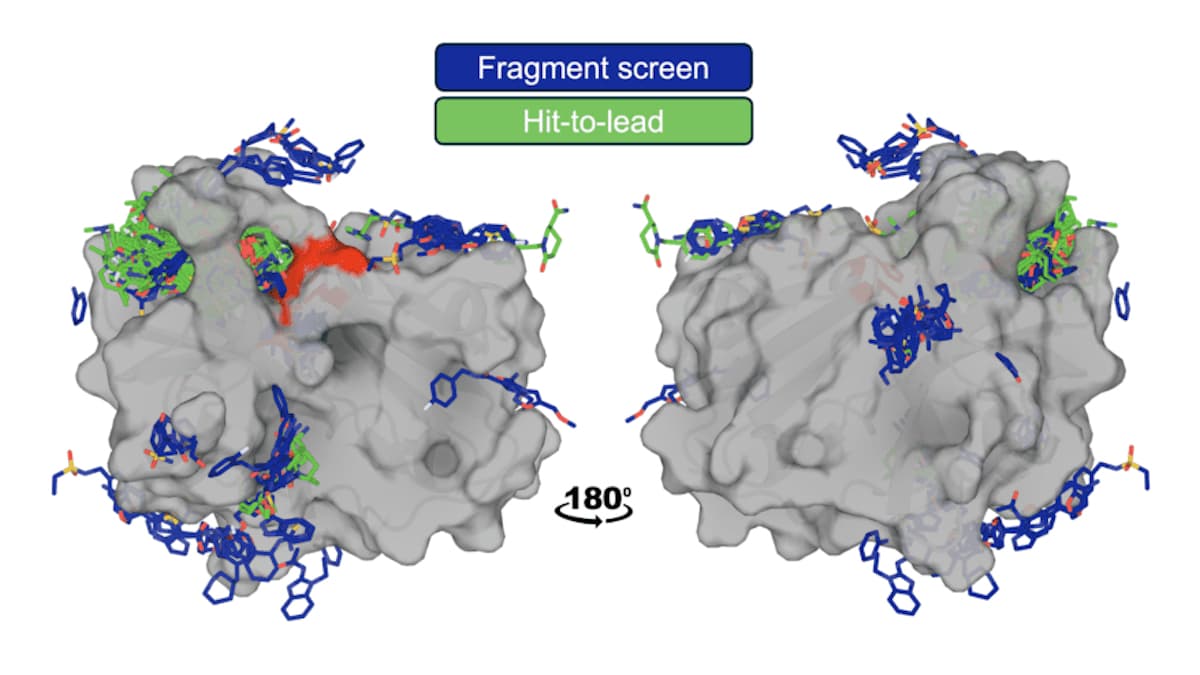
OpenBind Unveils Its First AI Model for Drug Discovery
OpenBind, a UK‑led consortium, has published its first AI‑ready dataset and a predictive model for the EV‑A71 2A protease, a target linked to hand, foot and mouth disease. The release includes 699 X‑ray structures and binding‑strength data for 601 compounds,...

MSD's Sac-TMT Delivers in First Phase 3 Readout
Merck (MSD) reported that its TROP2‑targeting antibody‑drug conjugate sacituzumab tirumotecan (sac‑TMT), licensed from China’s Kelun‑Biotech, improved progression‑free and overall survival in a phase 3 trial for advanced or recurrent endometrial cancer. The TroFuse‑005 readout marks the first pivotal data for sac‑TMT...

Novartis Reveals More Data Behind Pluvicto Expansion Bid
Novartas reported new PSMAddition trial data showing that adding its radioligand therapy Pluvicto to standard hormonal treatment cuts PSA progression by 58% in hormone‑sensitive metastatic prostate cancer. Deep PSA reductions below 0.2 ng/mL were achieved by 87.4% of patients versus 74.9%...

NICE Backs Stomach Cancer, PAH Drugs for NHS Use
The UK’s health technology assessment body NICE has endorsed AstraZeneca’s immunotherapy Imfinzi for peri‑operative treatment of aggressive stomach and gastro‑oesophageal junction cancers, and approved Merck & Co.’s Winrevair as an add‑on therapy for pulmonary arterial hypertension (PAH). More than 1,500 patients in...
FDA Hands BeOne, Taiho Approvals for Blood Cancers
BeOne Medicines’ BCL‑2 inhibitor Beqalzi (sonrotoclax) earned FDA accelerated approval as a third‑line treatment for mantle‑cell lymphoma, marking the first U.S. approval of a BCL‑2 drug for this indication. The approval is based on a phase 1/2 trial showing a 52%...
AZ Cues up Broader Use of Imfinzi in Bladder Cancer
AstraZeneca’s immunotherapy Imfinzi (durvalumab) demonstrated a significant survival benefit in the phase‑3 VOLGA trial for muscle‑invasive bladder cancer (MIBC) patients who cannot receive platinum chemotherapy. The study combined Imfinzi with Pfizer‑Astellas’ antibody‑drug conjugate Padcev before surgery, extending both event‑free and...

EU Agrees Plan to Tackle Essential Medicine Shortages
The European Union has reached a provisional agreement on the Critical Medicines Act, a legislative package aimed at strengthening the supply chain for essential drugs. The act requires member states to diversify procurement, prioritize EU‑based manufacturing, and launch strategic projects...
Novo Says High-Dose Wegovy Can Cause 28% Weight Loss
Novo Nordisk reported that its high‑dose 7.2 mg Wegovy formulation enabled early responders to lose nearly 28 % of body weight in the 72‑week STEP UP trial. Across the 1,400‑patient study, average weight loss reached 21 %, with 27 % of participants qualifying as early...
Novo Nordisk microRNA Drug Fluffs Its Lines in Heart Failure
Novo Nordisk’s microRNA‑targeting drug CDR132L failed to demonstrate a statistically significant improvement in left ventricular end‑systolic volume index in the phase 2 HF‑REVERT trial. The study enrolled 280 post‑myocardial‑infarction patients with an ejection fraction of 45% or lower and elevated NT‑proBNP...
GSK Enlists Local Aid for Chinese Rollout of Hep B Drug
GlaxoSmithKline (GSK) has signed a five‑and‑a‑half‑year agreement with Sino Biopharm’s subsidiary Chia Tai Tianqing Pharma (CTTQ) to import, distribute and promote its investigational hepatitis B antisense drug bepirovirsen across more than 5,000 Chinese medical centres, pending NMPA approval. The partnership gives...

Deal Finally Unlocks NHS Access to ITF's Duchenne Drug
ITF Pharma’s oral HDAC inhibitor Duvyzat has secured NICE endorsement for ambulatory Duchenne muscular dystrophy patients, unlocking access for more than 500 NHS England cases. The drug will be funded immediately through the Innovative Medicines Fund while NHS England arranges...

Capricor Sues Partner Nippon Shinyaku in DMD Therapy Spat
Capricor Therapeutics has filed a lawsuit against its Japanese partner Nippon Shinyaku and its U.S. subsidiary NS Pharma, alleging a fundamental pricing flaw in their distribution agreement for the Duchenne muscular dystrophy (DMD) cell therapy deramiocel. The biotech claims the...

Eisai, Biogen Face Delay to Subcutaneous Leqembi
The FDA has extended its review of Eisai and Biogen’s subcutaneous Leqembi Iqlik, moving the decision on its use as a starting dose to August 24. The agency requested a major amendment to the marketing application but has not raised safety...

J&J Seeks to Rewrite the Script on Depression Treatment
Johnson & Johnson’s new Generation Fine survey of 859 patients and 800 clinicians across eight countries found that nearly four‑in‑five people with major depressive disorder doubt oral antidepressants will fully resolve their symptoms. Residual symptoms linger for about 75% of...

Manufacturing Hangover Drives Daiichi Sankyo Into the Red
Daiichi Sankyo announced a fiscal‑year loss of ¥149.4 billion (about $950 million) despite a 12.5% revenue increase to ¥2,123 billion ($13.4 billion). The shortfall stems from costly third‑party manufacturing contracts for its antibody‑drug conjugates (ADCs) and an impairment of ¥19.3 billion ($123 million) after scrapping an...

Angelini Seeks US Beachhead with $4.1bn Catalyst Buy
Angelini Pharma agreed to acquire US‑based Catalyst Pharma for up to $4.1 billion, paying $31.50 per share—a 28% premium. The deal adds three FDA‑approved neuromuscular and epilepsy drugs that generated $589 million in 2023 sales and are forecast to reach $645 million by...

Lilly Inflates US Facility Investment Spending by $4.5bn
Eli Lilly has increased its U.S. capital‑investment program by $4.5 billion, bringing total spending in Indiana to $21 billion since 2020. The new funding supports the Lebanon Advanced Therapies plant, the company’s first dedicated genetic‑medicine manufacturing site, and future API and Foundry facilities....

Moderna's Q1 Revenues Rocket, Thanks to Ex-US Sales
Moderna reported first‑quarter revenue of $389 million, almost three times its 2025 level, with 80% generated outside the United States. U.S. sales rose to $80 million, up from $29 million a year earlier, reflecting deliveries under long‑term government agreements. The company now projects...

FDA's ODAC Delivers One Loss, One Win for AstraZeneca
The FDA’s Oncologic Drugs Advisory Committee (ODAC) voted 6‑3 against AstraZeneca’s oral SERD camizestrant in combination with CDK‑4/6 inhibitors for first‑line HR‑positive, HER2‑negative breast cancer with ESR1 mutations, citing a lack of overall survival benefit despite a 56% progression‑free survival...

Amgen, AZ Will Pilot FDA's Real-Time Clinical Trial Plan
The FDA unveiled a plan to receive clinical‑trial data in real time, aiming to accelerate drug development. AstraZeneca and Amgen have agreed to pilot the model with two studies that will stream endpoints and safety signals to regulators as they...

First-Quarter Sales Strength Buoys New GSK CEO Miels
GSK reported first‑quarter 2026 revenue of $10.3 billion, a 2% rise and 5% growth in constant currencies, surpassing analyst expectations. The boost came from specialty medicines and vaccines, positioning the company to meet its 2026 growth target of 3‑5% and its...

GSK Eyes October FDA Verdict on Chronic Hep B Drug
GlaxoSmithKline’s antisense drug bepirovirsen entered an accelerated FDA review, with a decision expected by October 26. The phase‑2 B‑WELL 1 and B‑WELL 2 trials showed a statistically higher functional cure rate versus placebo when added to standard hepatitis B therapy. If approved, bepirovirsen would...

Novartis Shares Fall as Entresto Sales Weaken
Novartis reported a 5% drop in first‑quarter net sales to $13.1 billion, driven by a 46% plunge in heart‑failure drug Entresto after U.S. patent expiry. Earnings per share fell 13% to $1.99 and the stock slipped nearly 3% as results missed...
GSK/Tesaro Claim Denied in Jemperli Lawsuit with AnaptysBio
A Delaware court rejected GSK and its Tesaro unit's attempt to terminate the 2014 licensing agreement with AnaptysBio, preserving the existing royalty rate for the PD‑1 cancer drug Jemperli. The ruling affirms that AnaptysBio has not breached the contract, while...

New NIHR-Funded TRC for Parkinson’s Disease
The National Institute for Health and Care Research (NIHR) has launched the Parkinson’s disease Translational Research Collaboration (PD‑TRC), the first of eight UK TRCs dedicated to Parkinson’s. Backed by NIHR and four major charities, the hub links 17 centres of...

Lilly Falls on Slower Start for Foundayo versus Wegovy
Eli Lilly’s oral GLP‑1 agonist Foundayo launched in early April with modest uptake, recording 1,390 prescriptions in week 1 and 3,707 in week 2, far below Novo Nordisk’s oral Wegovy, which logged over 3,000 fills in its first days and 18,410 in the second...

FDA Grants Three Priority Vouchers to Psychedelics
The FDA has granted three Commissioner's National Priority Vouchers to psychedelic‑focused firms—Compass Pathways, Transcend Therapeutics and the Usona Institute—following President Trump’s executive order to accelerate mental‑health treatments. All three companies already hold Breakthrough Therapy Designations, and the vouchers promise faster...
FDA Declines to Approve AbbVie's Botox Follow-Up
The FDA sent a complete response letter to AbbVie, rejecting its filing for TrenibotE, a rapid‑acting, shorter‑duration botulinum toxin intended as a follow‑up to Botox. The agency’s concerns focus on manufacturing data rather than safety or efficacy, and AbbVie says...
FDA Clears First Genetic Hearing Loss Gene Therapy
Regeneron became the first company to receive FDA clearance for a gene therapy targeting congenital hearing loss, approving its OTOF‑focused product Otarmeni. The therapy, an AAV‑delivered one‑shot infusion, will be offered free to U.S. patients, a rare move given typical...

Ray's Upscaled Series B, and Other Biofinancings
Ray Therapeutics closed a $125 million Series B to advance optogenetic therapies for retinitis pigmentosa, Stargardt disease and geographic atrophy, building on a $100 million Series A from 2023. Tortugas Neuroscience secured $106 million in seed and first‑round financing to push licensed schizophrenia and tinnitus...

Swiss Franc Strength Puts Roche's Results in a Poor Light
Roche reported a headline 5 % drop in first‑quarter revenue as the Swiss franc surged, but constant‑currency sales rose 6 % to CHF 14.77 billion (about $16 billion) and 9 % versus the weakened dollar. The pharma division’s reported revenue fell 4 % to CHF 11.47 billion (≈$12.5 billion) yet...

New Data Build Case for Roche's Oral BTK Drug for MS
Roche reported that its oral BTK inhibitor fenebrutinib dramatically reduced relapse rates and MRI lesions in two phase 3 FENhance trials for relapsing multiple sclerosis, outperforming Sanofi's Aubagio. The drug cut annualised relapse rates by 51.1% and 58.5% and lowered inflammation...

Medicare Obesity Drug Pilot Extended After Insurer Pushback
The Trump administration has pushed back the start of the Medicare GLP‑1 Bridge pilot by a year after insurers fell short of the 80% enrollment target. The federal government will continue to cover drugs such as Novo Nordisk’s Wegovy, Eli Lilly’s...

Update: Lilly Makes $7bn Bid for in Vivo CAR-T Firm Kelonia
Eli Lilly has confirmed a $3.25 billion upfront offer for Kelonia Therapeutics, with milestone payments that could lift the total value to $7 billion. Kelonia’s in vivo CAR‑T candidate KLN‑1010 demonstrated MRD‑negative responses in a four‑patient early‑stage study, highlighting the promise of off‑the‑shelf cell...

AZ Is Three for Three with COPD Hope Tozorakimab
AstraZeneca’s anti‑IL‑33 antibody tozorakimab has succeeded in all three pivotal Phase 3 COPD trials, showing a statistically significant reduction in moderate‑to‑severe exacerbations. The MIRANDA study confirmed benefit with biweekly dosing, while OBERON and TITANIA validated once‑monthly regimens. These results place AZ...

Multiple Myeloma Drug Blenrep Backed for Wider NHS Use
The UK’s health technology regulator NICE has broadened the approved use of GSK’s BCMA‑targeting drug Blenrep, allowing it to be combined with Takeda’s Velcade and dexamethasone for a larger second‑line multiple myeloma population. The new guidance lifts the restriction that...

UK Says It Has Hit Target on Commercial Trial Set-Up Times
The UK’s National Institute for Health and Care Research (NIHR) reported that average clinical‑trial set‑up time fell to 122 days in the six months to March, beating its 150‑day target set for March 2026. The reduction follows a suite of reforms,...

Beeline Raises $300m for BMS Drugs, and Other Biofinancings
Beeline Medicines launched with a $300 million Series A led by Bain Capital, acquiring Bristol Myers Squibb’s immunology pipeline that includes an oral TLR7/8 inhibitor for lupus and other candidates targeting IL‑2‑CD25, TYK2, IL‑18 and IL‑10 pathways. The week also saw sizable financings...

Novartis CEO Joins Board of "Responsible" AI Firm Anthropic
Novartis chief executive Vas Narasimhan has been appointed to the board of Anthropic, a leading responsible‑AI firm. The move follows Anthropic’s $400 million acquisition of Coefficient Bio, expanding its platform for AI‑driven target and drug discovery. Anthropic recently launched Claude for...
Alamar, Hemab, Kailera, and Seaport Advance IPOs
A wave of biotech IPOs is hitting Nasdaq in early 2026 as Alamar Biosciences, Hemab Therapeutics, Kailera Therapeutics and Seaport Therapeutics file prospectuses. Alamar plans to sell 9.4 million shares at $15‑$17, targeting up to $154 million to expand its proteomics biomarker...

IDEAYA/Servier PKC Drug Aces Uveal Melanoma Trial
IDEAYA’s PKC inhibitor darovasertib, combined with Pfizer’s crizotinib, achieved a statistically significant improvement in progression‑free survival in the phase 2/3 OptimUM‑02 trial for HLA‑A*02:01‑negative metastatic uveal melanoma. Median PFS extended to 6.9 months versus 3.1 months for investigator‑chosen immunotherapy, and the...

NICE Update Doubles Eligible Patients for AZ's Lokelma
NICE’s draft guidance doubles the number of patients in England and Wales who can receive AstraZeneca’s Lokelma for hyperkalaemia. The eligibility threshold was lowered from a serum potassium level of 6.0 mmol/L to 5.5 mmol/L, and the specialist‑only initiation rule was removed....

Senju Launches First-in-Class Dry Eye Disease Drug in Japan
Senju Pharma has launched Avarept, the first TRPV1 antagonist drug for dry eye disease (DED) in Japan, licensed from Mochida and distributed by Takeda. The ophthalmic suspension is priced at ¥577.50 (approximately $3.63) per 5 ml bottle. DED affects over 20 million...

Diagnostics Lag Is Holding Back New Therapies, Says Study
A new UCSF analysis published in Science warns that diagnostic development is lagging behind therapeutic breakthroughs because of regulatory and reimbursement gaps. The authors highlight that nearly half of the world’s population—about 47%—has limited or no access to essential tests,...

Chinese Trial Backs Base-Editing Drug for Thalassaemia
A Chinese investigator‑led trial of CorrectSequence Therapeutics' ex vivo base‑editing drug CS‑101 showed that all five patients with transfusion‑dependent beta‑thalassaemia became transfusion‑independent after a single infusion, with an average cessation time of 16 days and sustained hemoglobin gains over three months....

Syneron Raises $150m for Macrocyclics, and Other Financings
Syneron Bio, a Beijing‑based biotech, closed a $150 million Series B just four months after raising $100 million, underscoring strong investor appetite for macrocyclic peptide therapeutics. The company’s AI‑powered Synova platform claims to design drug candidates for protein targets that have eluded conventional...