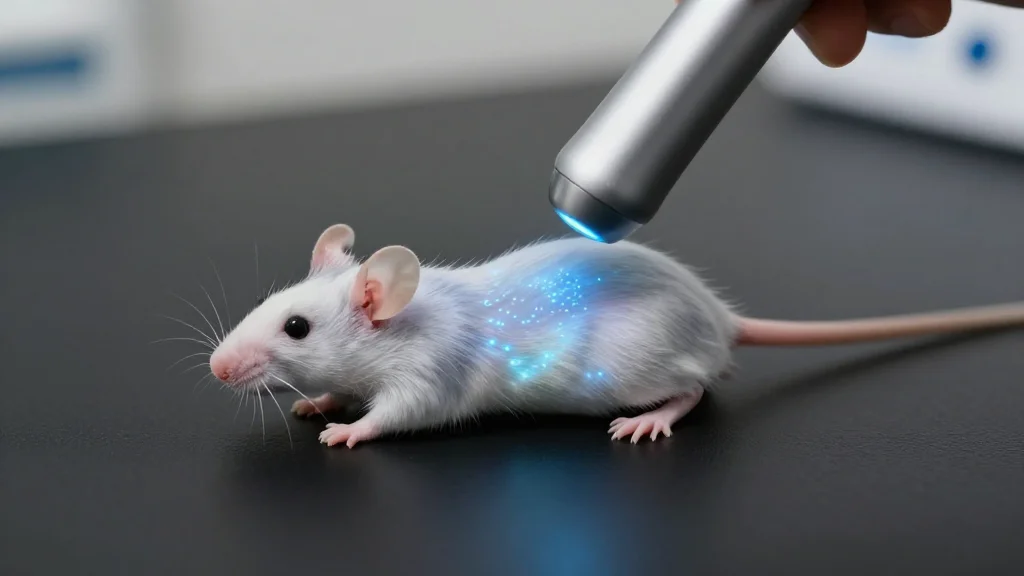
Stanford Team Demonstrates Ultrasound‑Powered Nanophosphor Light Source Inside Living Tissue
Why It Matters
The ability to generate light inside the body without incisions resolves a long‑standing bottleneck in photomedicine, where photon attenuation has forced clinicians to rely on invasive hardware. By leveraging ultrasound—a modality already ubiquitous in diagnostic imaging—the approach integrates seamlessly with existing clinical workflows, potentially accelerating adoption. Moreover, the platform’s modularity—allowing different nanoparticle compositions to emit various wavelengths—creates a versatile toolbox for applications ranging from neuromodulation to targeted antimicrobial treatment. Beyond therapeutic uses, the technology could transform basic research. Scientists studying deep‑brain circuits currently depend on head‑mounted fiber optics that constrain animal behavior. An ultrasound‑activated light source would enable freely moving studies, improving the ecological validity of neuroscientific findings and reducing animal stress. The ripple effects across drug development, diagnostics, and fundamental biology could be substantial, positioning nanophosphor‑based ultrasound activation as a foundational enabling technology for the next decade of biomedical innovation.
Key Takeaways
- •Mechanoluminescent nanoparticles emit 490 nm blue light when compressed by ultrasound.
- •Ultrasound penetrates centimeters of tissue, allowing activation of particles anywhere in the body.
- •In live mice, targeted ultrasound triggered neuronal firing and directional turning behavior.
- •Current ceramic core must be replaced with biodegradable material before human use.
- •Potential market impact includes non‑invasive optogenetics, photodynamic therapy, and gene editing.
Pulse Analysis
Historically, optogenetics has been limited by the need for invasive fiber‑optic delivery, a constraint that has slowed clinical translation despite robust preclinical data. The Stanford breakthrough sidesteps this limitation by converting a well‑established imaging modality—ultrasound—into a light‑generation trigger. This convergence of acoustics and nanophotonics could redefine the value proposition of phototherapy platforms, shifting the competitive landscape from hardware‑centric firms to hybrid nanomaterial companies.
From a market perspective, the technology aligns with a broader trend toward minimally invasive interventions. Companies that have built pipelines around light‑activated drugs or gene‑editing tools will likely view ultrasound‑driven nanophosphors as a plug‑and‑play upgrade, reducing the need for costly surgical procedures. Early‑stage investors may therefore prioritize startups that can demonstrate scalable synthesis of biocompatible mechanoluminescent particles and secure regulatory pathways.
Looking ahead, the critical milestones will be material safety validation and demonstration of efficacy in larger mammals. If those hurdles are cleared, the field could see a cascade of clinical trials targeting deep‑seated cancers, cardiac arrhythmias, and neuropsychiatric disorders. The convergence of nanotech, acoustics, and synthetic biology promises a new class of therapeutic devices that are both precise and patient‑friendly, potentially reshaping how clinicians think about light‑based medicine.
Stanford Team Demonstrates Ultrasound‑Powered Nanophosphor Light Source Inside Living Tissue
Comments
Want to join the conversation?
Loading comments...