WHO Recommends New Diagnostic Tools to Help End TB
Why It Matters
Accelerating low‑cost, rapid TB testing narrows diagnostic gaps, speeds treatment initiation, and strengthens global health security. The economic upside underscores TB control as a high‑return investment for governments and donors.
Key Takeaways
- •Point‑of‑care TB tests cost under half existing molecular diagnostics
- •Results delivered in under one hour, enabling quicker treatment
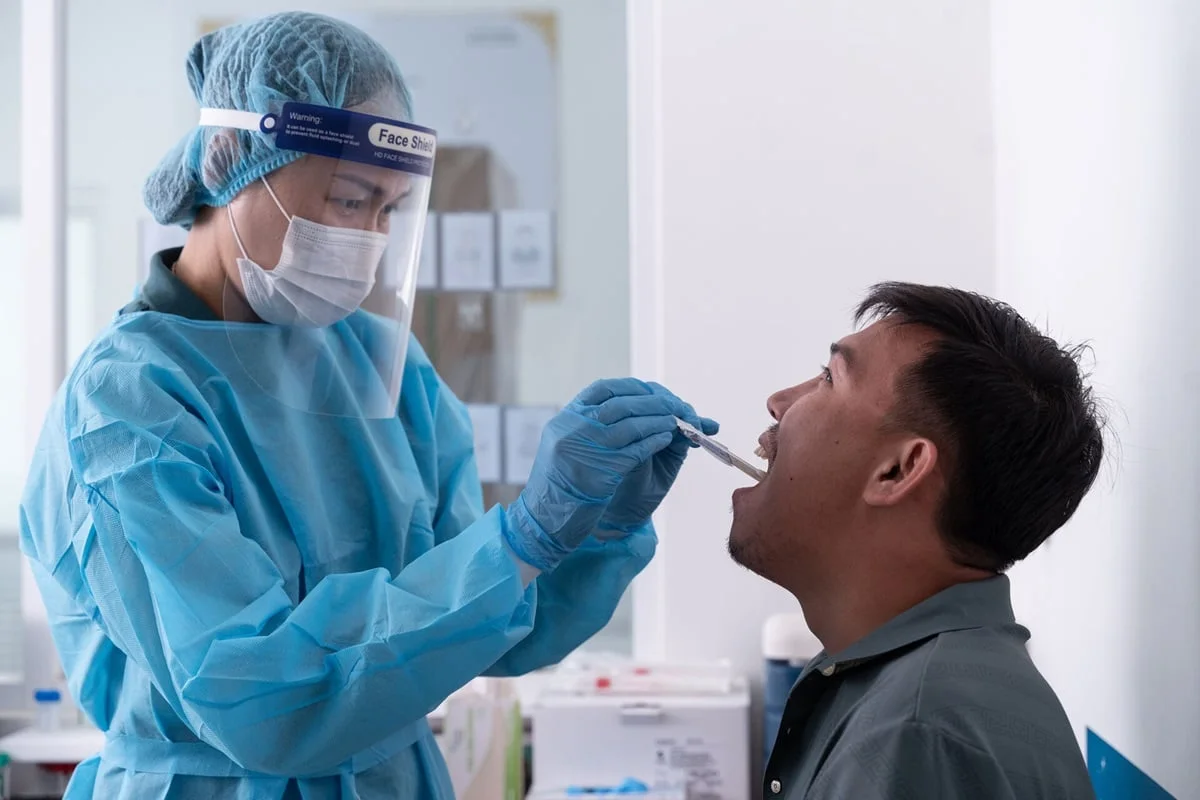
- •Tongue‑swab sampling reaches patients unable to produce sputum
- •Sputum pooling cuts commodity costs and machine time
- •WHO estimates $43 health/economic return per dollar invested
Pulse Analysis
Tuberculosis remains a leading cause of death, with more than 3,300 fatalities and 29,000 new infections each day. Traditional laboratory‑based diagnostics often require costly equipment, skilled personnel, and lengthy transport times, creating bottlenecks in low‑resource settings. WHO’s new point‑of‑care guidelines introduce battery‑operated, modular devices that slash costs to under 50% of conventional molecular tests and provide results in less than 60 minutes. By moving testing closer to patients, health systems can initiate therapy sooner, reducing transmission chains and improving outcomes.
Beyond speed and affordability, the guidelines champion innovative sample collection methods. Tongue‑swab specimens enable testing for individuals who cannot expectorate sputum, expanding reach to adolescents and people with advanced disease who are at higher mortality risk. The recommended sputum‑pooling strategy aggregates multiple samples for a single assay, dramatically lowering reagent use and machine time—an essential adaptation for resource‑constrained programs. These tools also have cross‑pathogen potential, offering a platform that could be repurposed for HIV, mpox or HPV, aligning with the growing demand for versatile, patient‑centred diagnostics.
Financially, the WHO underscores a compelling return on investment: every dollar spent on TB control can yield up to $43 in health and economic benefits. Yet global research funding remains far below the estimated $5 billion annual need, jeopardizing the development of next‑generation diagnostics, drugs, and vaccines. Initiatives like the TB Vaccine Accelerator Council aim to bridge this gap by aligning stakeholders and accelerating equitable access. For policymakers, scaling these point‑of‑care solutions and securing sustained financing are critical steps toward the 2030 goal of universal TB testing and treatment, reinforcing both public health and economic resilience.
WHO recommends new diagnostic tools to help end TB
Comments
Want to join the conversation?
Loading comments...