Why Do Falls Rise with Age? Cerebellar Neuron Firing Problems (and Potential Therapeutics)
Key Takeaways
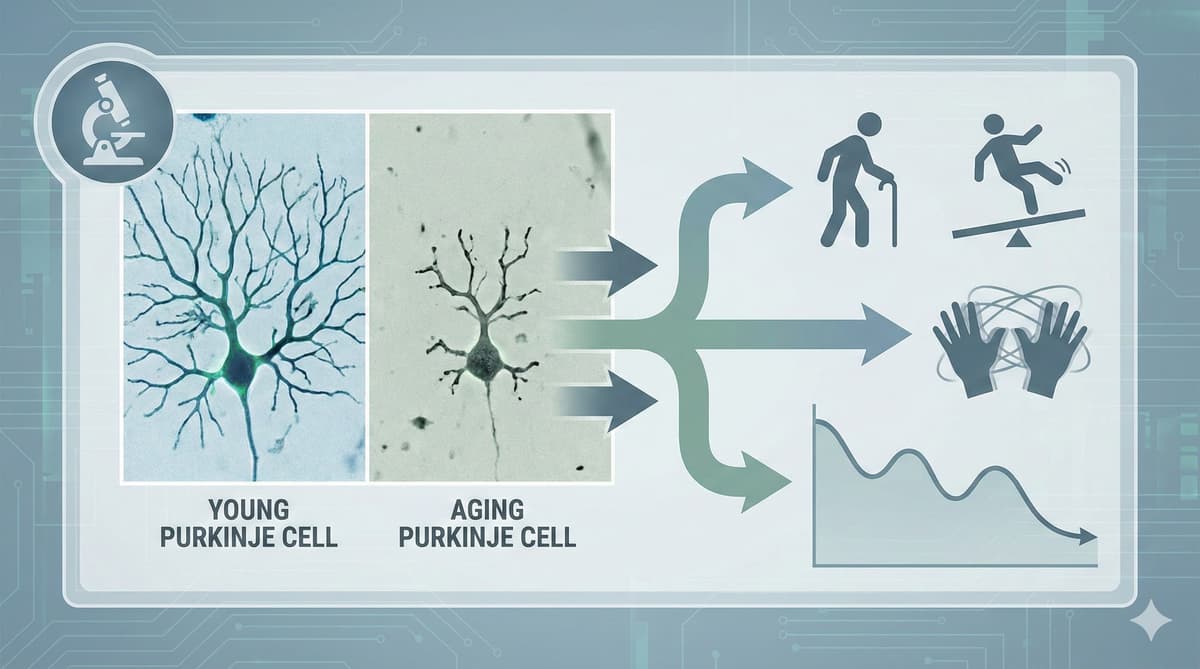
- •Purkinje cell count stays stable in aged mouse cerebellum
- •Intrinsic firing frequency declines with age, causing motor frailty
- •Chemogenetically increasing firing restores balance in 18‑month mice
- •Potential therapeutics include Kv blockers like 4‑Aminopyridine
- •Reversibility shifts focus from neurodegeneration to electrophysiology
Pulse Analysis
The cerebellum has long been viewed as a structural casualty of aging, with most research assuming that neuron loss drives the loss of balance and coordination. The McGill study overturns this paradigm by showing that, at least in mice, Purkinje cells—the sole output neurons of the cerebellar cortex—remain numerically intact while their pacemaker activity slows dramatically. This electrophysiological drift compromises the timing of motor commands, producing the hallmark clumsiness that precedes falls in seniors. Understanding that the deficit is functional rather than structural reframes the therapeutic agenda: instead of trying to replace dead cells, interventions can aim to restore the cells’ firing rhythm.
Translating these findings to humans will require agents that safely elevate Purkinje firing without inducing hyperexcitability. Existing potassium‑channel blockers such as 4‑Aminopyridine (Dalfampridine) already boost neuronal firing in multiple sclerosis and ataxia patients, offering a proof‑of‑concept that pharmacologic pacing is feasible. However, the narrow therapeutic window and seizure risk demand more selective modulators—perhaps compounds that target specific Nav/Kv channel subtypes or enhance calcium‑dependent pacemaker currents. Non‑pharmacologic approaches, including high‑intensity interval training, complex motor skill acquisition, and targeted wobble‑board perturbations, may also up‑regulate excitatory input and neuromodulatory tone, providing synergistic benefits.
The broader impact extends beyond fall prevention. If electrophysiological rejuvenation can restore motor function, it may also improve overall physical healthspan, reducing healthcare costs associated with injuries and long‑term care. Future research should prioritize biomarkers for in‑vivo Purkinje activity—such as advanced neuroimaging or wearable electrophysiology—to bridge the gap between rodent models and clinical trials. By focusing on the brain’s rhythm rather than its cell count, the field moves closer to interventions that keep older adults moving confidently and independently.
Why do falls rise with age? Cerebellar Neuron Firing Problems (and Potential Therapeutics)
Comments
Want to join the conversation?