
SAPT After TAVR Linked to Key Benefits over DAPT, New Data Confirm
New registry data from the TRITAVI cohort show that single antiplatelet therapy (SAPT) after transcatheter aortic valve replacement (TAVR) markedly improves survival compared with dual antiplatelet therapy (DAPT). Among more than 5,000 patients with a mean age of 81, six‑month all‑cause mortality was 2.4 % on SAPT versus 5.4 % on DAPT, and the gap persisted at 24 months (11.7 % vs 14.2 %). SAPT also reduced major bleeding without raising stroke risk, and its use has risen as DAPT prescriptions fell from 48 % to 38 % over the study period.
American Heart Association Takes on Cardiac Amyloidosis with New Initiative
The American Heart Association (AHA) has launched a national clinical initiative to raise awareness of cardiac amyloidosis and standardize early, non‑biopsy diagnosis. The program targets cardiologists, imaging specialists, hematologists and primary‑care providers, promoting a diagnostic algorithm that combines nuclear scintigraphy...
Have Clinicians Been Wrong About How They Diagnose PAD?
A new JACC study of 225,000 U.S. veterans shows that ankle‑brachial index values of 0.91‑1.00, previously called “borderline,” are linked to higher rates of major adverse limb events, amputations and revascularization than normal ABI. The risk elevation holds across gender...

Prasugrel After PCI Outperforms Other Common Antiplatelet Drugs
A new network meta‑analysis of 15 randomized trials involving nearly 49,000 PCI patients finds prasugrel delivers a superior balance of efficacy and safety compared with clopidogrel and ticagrelor. Prasugrel significantly reduced major adverse cardiovascular events, myocardial infarction, and stent thrombosis...

AI Tool Helps Remote Monitoring Clinics Explore Patient Data
Octagos introduced Ask Atlas, an AI-powered natural‑language interface for its electrophysiology remote‑monitoring platform. The tool lets clinicians pose simple questions and retrieve answers from device transmissions, electronic health records, workflow status, and billing data within seconds. By replacing manual chart...

Diabetes Drugs Improve Survival for Patients with ‘Broken Hearts’
Early administration of sodium‑glucose cotransporter‑2 (SGLT2) inhibitors was linked to a lower all‑cause mortality rate in patients with Takotsubo syndrome, a condition often dubbed “broken‑heart syndrome.” The real‑world analysis examined nearly 55,000 TTS cases from 2015‑2025, propensity‑matching 1,803 patients who...

Conduction System Pacing After TAVR Linked to Improved Outcomes in New Meta-Analysis
A new meta‑analysis of eight studies involving nearly 800 patients who required permanent pacemakers after transcatheter aortic valve replacement (TAVR) compares conduction system pacing (CSP) with conventional right ventricular pacing (RVP). While overall mortality rates were similar, CSP produced a...

FDA Confirms Class I Recall for Vascular Device Used by Cardiologists and Surgeons
The FDA has upgraded its warning on the Relay Pro Thoracic Stent Graft System to a Class I recall, the agency’s highest safety alert, after reports that the device can fail to detach from its delivery system. The recall targets non‑bare stent...
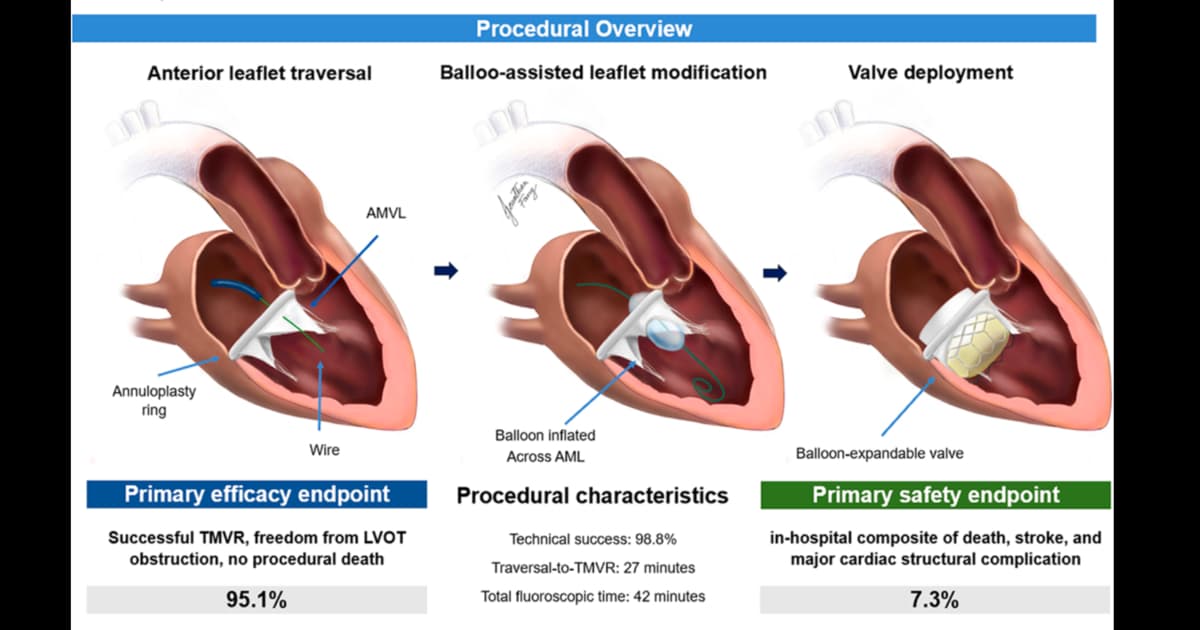
BATMAN to the Rescue: TMVR Technique Helps Cardiologists Reduce Risk of LVOT Obstruction
A new study in JACC: Cardiovascular Interventions shows that Balloon‑Assisted Translocation of the Mitral Anterior Leaflet (BATMAN) can dramatically lower left ventricular outflow tract (LVOT) obstruction risk during transcatheter mitral valve replacement (TMVR). The analysis of 83 high‑risk patients treated...
How TAVR Valve Implant Depth Impacts Clinical Outcomes
A large observational analysis published in JACC: Cardiovascular Interventions found that a higher transcatheter aortic valve replacement (TAVR) implantation position yields better early clinical outcomes. One‑year combined mortality and stroke was 9.7% across all depth groups, but permanent pacemaker implantation...
FDA Warns Public About Johnson & Johnson Heart Pump Controllers After Patient Death
The FDA issued an early‑alert warning that Johnson & Johnson MedTech’s Automated Impella Controllers (AICs) can unexpectedly restart after at least 80 minutes without residual pulsatility, causing a 35‑second pump pause. The software glitch has been linked to two serious...

Advocacy Group Wants TAVR Changes, Urging Trump Admin to ‘Stop Getting in Between Patients and Their Doctors’
The Alliance for Aging Research is pressuring the Trump administration’s CMS to broaden Medicare coverage of transcatheter aortic valve replacement (TAVR). A poll of 1,000 U.S. adults showed 77% favor parity with open‑heart surgery coverage and 67% would back candidates...

Medtronic Recalls Thousands of Surgical Devices Due to Contamination Risk
Medtronic announced a Class II recall of more than 18,000 retrograde cannulae used in open‑heart surgery after detecting a potential sterile‑barrier breach. The recall covers specific lots of Gundry and DLP silicone‑body cannula models ranging from 13FR to 15FR. Affected units...
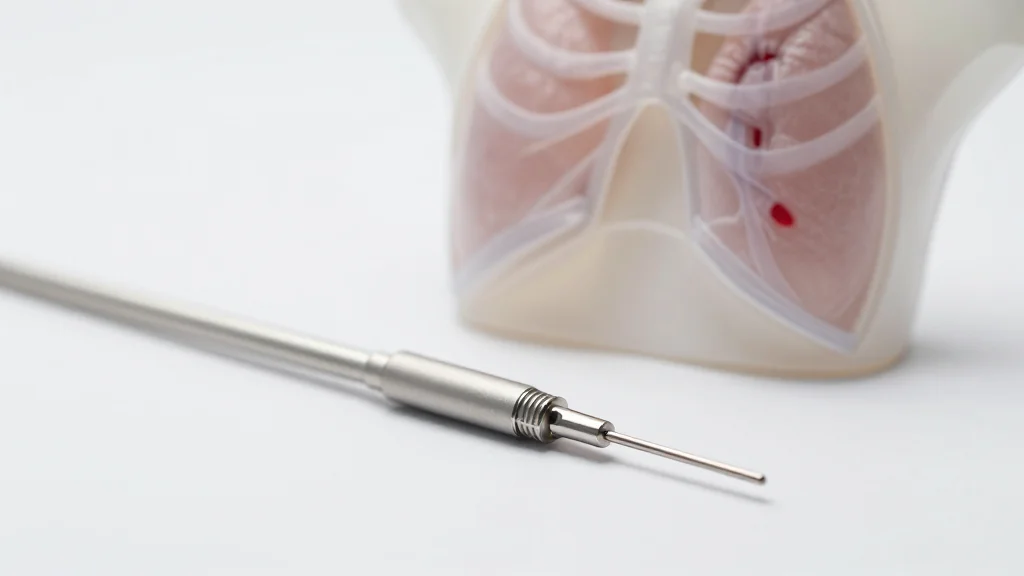
'Compelling' Study Shows Promise of New Pulmonary Embolectomy System
Jupiter Endovascular’s Vertex Pulmonary Embolectomy System cleared both primary efficacy and safety endpoints in the SPIRARE II pivotal trial, a prospective single‑arm study of 123 acute intermediate‑risk PE patients across 19 sites. The device delivered a mean 0.39 reduction in RV/LV...

Shockwave Medical's New IVL Catheter Overcomes Previous Limitations
Shockwave Medical unveiled an upgraded intravascular lithotripsy (IVL) catheter that adds a hydrophilic coating, a more flexible shaft, and a tapered tip, enabling easier navigation through tight, calcified lesions. The redesign also repositions lithotripsy emitters and cuts the pulse‑to‑pulse delay...