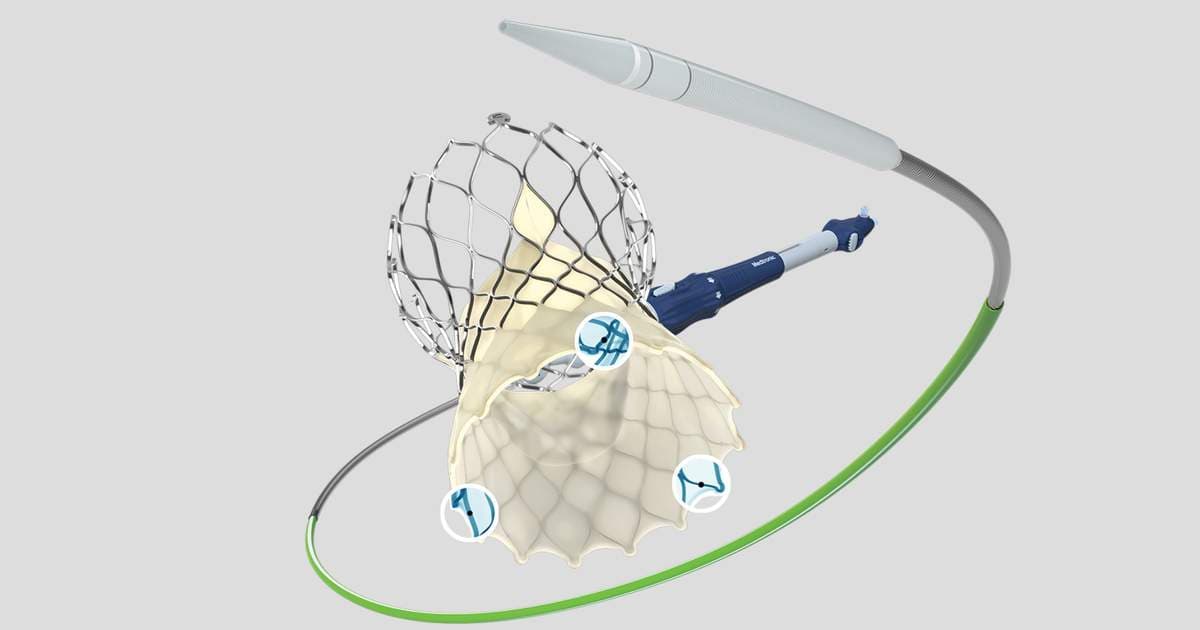
SMART Update: Self-Expanding TAVR Valves Still Linked to Superior Durability After 3 Years
New three‑year data from the SMART trial show that self‑expanding Evolut transcatheter aortic valve replacement (TAVR) valves outperform balloon‑expandable Sapien 3 devices in patients with small aortic annuli. The Evolut group experienced a bioprosthetic valve dysfunction rate of 16.3 % versus 54.4 % for Sapien 3, and demonstrated significantly lower valve thrombosis rates. Safety outcomes remained comparable between the two platforms. The study cohort was 90 % female, underscoring the relevance for women with small annuli.

More Good News for Boston Scientific’s Coronary IVL Tech
Boston Scientific reported strong early results from its single‑arm FRACTURE trial of the Seismiq 4CE coronary intravascular lithotripsy (IVL) catheter. Freedom from major adverse cardiac events at 30 days was 93.3%, well above the 86.2% target, and procedural success hit 93.7%...
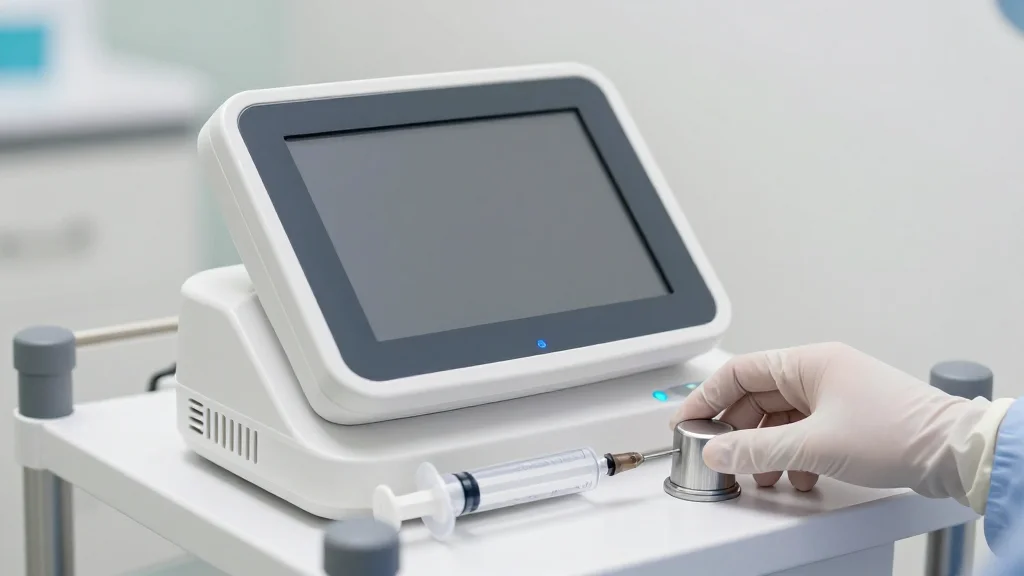
FDA Clears Next-Gen Contrast Management System for the Cath Lab
Acist Medical Systems, a Bracco subsidiary, received FDA clearance for its Acist Pro Diagnostic System, a next‑generation contrast management platform for cardiac catheterization labs. The system, already launched in Japan and Europe, offers real‑time contrast tracking, customizable presets, and on‑screen...

Heartflow Reports Significant Revenue Growth as CCTA Sees Wider Adoption
Heartflow announced a 41% year‑over‑year jump in first‑quarter 2026 total revenue to $52.6 million, with U.S. sales climbing 42% to $48.3 million. The surge is driven by expanding use of its AI‑powered FFR‑CT platform and the newly launched Plaque Analysis software. Inclusion...

Trump Administration Exempts Doctors From Work Visa Freeze
The U.S. Department of Homeland Security has lifted the freeze on processing existing H‑1B visas for foreign‑born physicians, allowing them to begin or continue work in American hospitals. The exemption comes after pressure from medical societies such as the ACC...
Thousands of Reprocessed EP Catheters Recalled
Stryker Sustainability Solutions, the reprocessing arm of Stryker, announced a Class II recall of more than 8,000 reprocessed electrophysiology (EP) catheters after discovering incomplete seals caused by a process‑control lapse. The recall spans multiple device models from manufacturers such as BARD,...
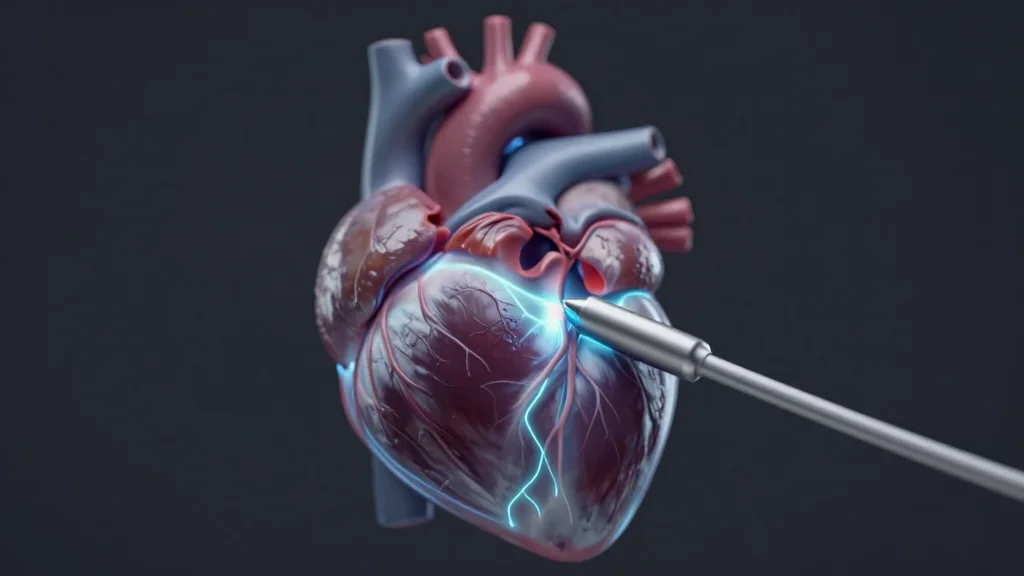
PFA Has Been a Game-Changer for Heart Patients—But There Are Still Risks
Pulsed field ablation (PFA) is reshaping atrial fibrillation treatment by using electrical fields instead of heat, delivering a markedly safer profile than radiofrequency or cryoablation. Clinicians report near‑zero phrenic nerve and esophageal injury, accelerating adoption of multiple FDA‑cleared catheters such...

Blood Pressure Drug Recalled After Failed Test
The FDA announced a Class II recall of 675 bottles of 20‑mg Enalapril Maleate tablets after a failed organic impurities test. The affected product, manufactured by Unique Pharmaceutical Laboratories (J.B. Chemicals & Pharmaceuticals) in India, was distributed across the U.S. by...
Cardiologists Are First in World to Use New Leaflet-Splitting Technique During TAVR
Interventional cardiologists performed the first‑in‑human transcatheter aortic root tricuspidization (ART) during transcatheter aortic valve replacement (TAVR) to treat bicuspid aortic stenosis. Seven symptomatic patients, average age 64.6, underwent ART‑assisted TAVR via transfemoral access with no 30‑day deaths or strokes. The...
AI Uses 12-Lead ECGs to Predict Long-Term Stroke Risk
Researchers at Brigham and Women’s Hospital and its Boston affiliates have unveiled ECG2Stroke, an AI model that reads standard 12‑lead electrocardiograms to forecast long‑term ischemic stroke risk. Trained on data from more than 100,000 patients, the convolutional neural network achieved...

Fewer Biopsies, Happier Patients: Why Molecular Blood Testing Is a Game-Changer for Managing Heart Transplant Recipients
Heart‑transplant patients have long depended on frequent endomyocardial biopsies, a process that can involve up to 14 invasive procedures in the first year and carries risks of perforation, bleeding, and sampling error. CareDx’s HeartCare platform introduces two complementary molecular blood...
Is TAVR Too Common Among Younger Patients? New Data Prompt a ‘Call to Action’
A new real‑world analysis of more than 100,000 aortic valve replacements shows that transcatheter aortic valve replacement (TAVR) is being used in 36% of U.S. patients under 65, far above guideline recommendations that favor surgical aortic valve replacement (SAVR) for...
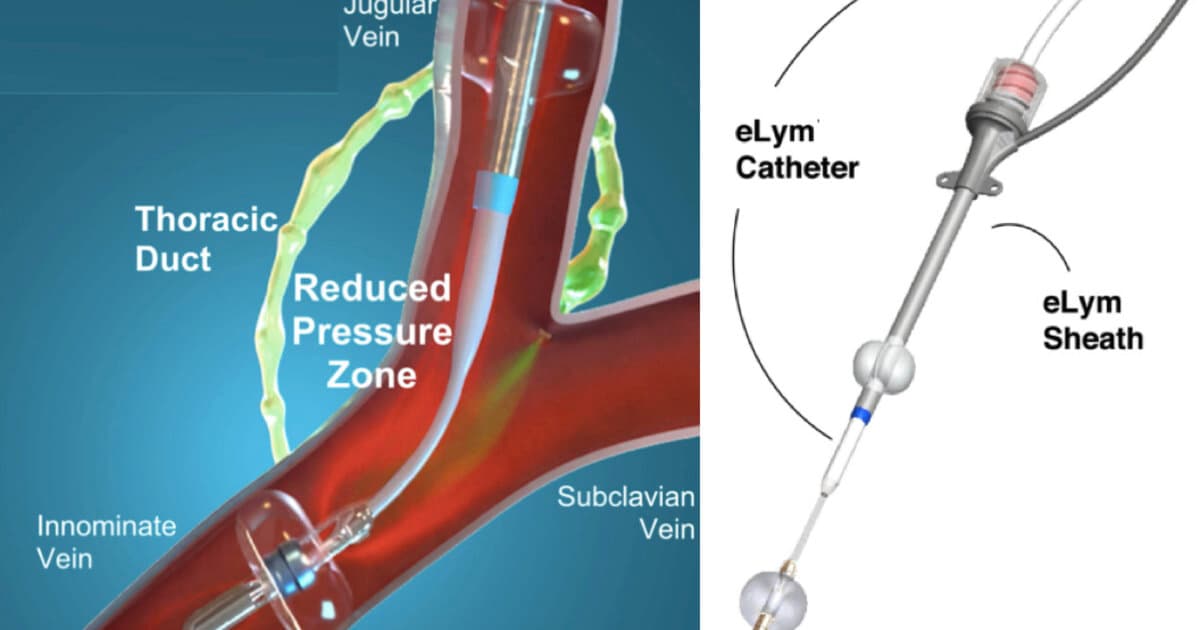
First-of-Its-Kind Drainage Device Helps Limit Heart Failure Rehospitalizations
The investigational WhiteSwell eLym catheter‑based system, designed for lymphatic drainage, was implanted in 40 acute decompensated heart‑failure patients in the DELTA‑HF trial. Ninety‑eight percent experienced lowered thoracic duct pressure, leading to an average 15‑lb weight loss and stable kidney function....
FDA Clears High-Pressure Balloon Catheter for BAV Procedures
Corvention, an Arizona‑based medtech startup, received FDA clearance for its KardiaPSI high‑pressure balloon catheter used in balloon aortic valvuloplasty (BAV). The device is engineered to retain its diameter under extreme pressures, delivering precise focal force in heavily calcified aortic valves....
New Coronary IVL Tech From Boston Scientific Impresses in First-in-Human Study
Boston Scientific’s Seismiq 4CE Coronary IVL Catheter, a laser‑driven intravascular lithotripsy system, completed a first‑in‑human study of 41 patients with severely calcified coronary lesions. The device achieved a 97.4% device‑success rate and a 90.2% freedom‑from‑major‑adverse‑cardiovascular‑events at 30 days, with procedural success...

ASNC Speaks to Policymakers About Prior Authorization, Medicare Cuts and More
During its second annual Hill Day, the American Society of Nuclear Cardiology (ASNC) convened 19 advocates who met with 66 congressional offices to discuss key policy issues. The society backed the bipartisan Improving Seniors’ Timely Access to Care Act to...
AATS Helps Heart Surgeons Diagnose and Manage Heparin-Induced Thrombocytopenia
A new expert consensus from the American Association for Thoracic Surgery (AATS) tackles the persistent challenges of diagnosing and managing heparin‑induced thrombocytopenia (HIT) in cardiac‑surgery patients. The 17‑member panel recommends delaying antibody testing until a biphasic platelet‑count pattern emerges, reducing...
How Expert Guidelines Have Influenced TAVR Vs. SAVR Decisions
New analysis presented at the AATS 2026 meeting shows that the 2020 ACC/AHA valvular disease guidelines halted the decline of surgical aortic valve replacement (SAVR) and stabilized its volume after years of erosion by transcatheter aortic valve replacement (TAVR). The...

Upfront Costs of Robotic Heart Surgery Are High—But It May Be a Smart Investment
New research presented at the American Association for Thoracic Surgery meeting examined over 8,000 mitral valve repairs, including 780 robotic procedures, and found that robotic surgery costs about $6,000 more per case than traditional methods. Despite higher operating‑room expenses, patients...

Cannabis Use Increases Risk of Death, Heart Attack for ED Patients
Researchers at the University of Texas Medical Branch examined over 1.7 million emergency department visits from 2005‑2022, comparing nearly 300,000 patients with recent cannabis‑use disorder to matched controls. The study found a 2.9% three‑year mortality rate for cannabis users versus 1.7%...

Resilia Tissue Developed by Edwards Lifesciences Associated with Long-Term Durability
Edwards Lifesciences presented 10‑year results from the COMMENCE surgical aortic valve trial, showing its Resilia tissue leaflets achieve 97.9 % freedom from structural valve deterioration. The technology combines calcium‑blocking chemistry with dry storage to limit calcification, delivering stable hemodynamics and low...
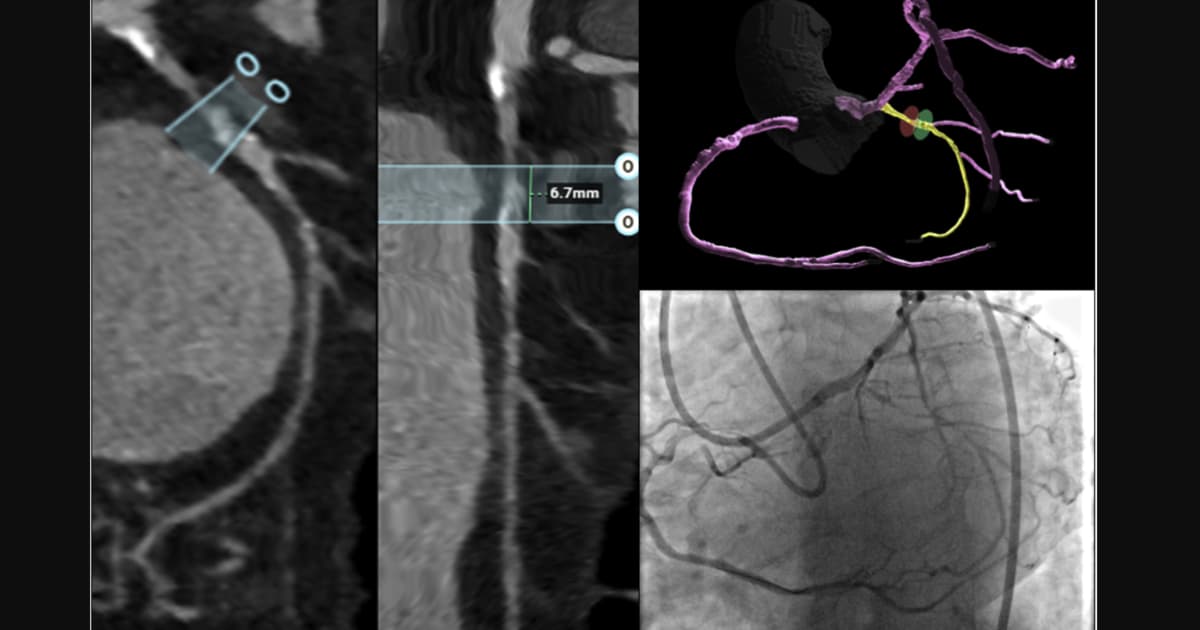
CCTA Brings Clarity, Better Outcomes to CTO PCI
A systematic review of 56 studies shows that coronary CT angiography (CCTA) performed before chronic total occlusion (CTO) percutaneous coronary intervention (PCI) enhances procedural planning and improves patient outcomes. The authors identified three concrete benefits: better case selection, more precise...

Physicians Chime In: Coronary Plaque Staging Offers More Precise Cardiac Patient Management
Heartflow’s Plaque Staging introduces a plaque‑volume based risk model that outperforms traditional stenosis and clinical scores. The framework is backed by data from more than 23,000 patients with up to 16 years of follow‑up, showing total plaque volume (TPV) predicts events...
SCCT President: Key Trends in Cardiac CT
SCCT President Chinnaiyan outlined how cardiac CT is evolving from a diagnostic tool to an AI‑driven platform for structural heart planning, PCI guidance, and preventive cardiology. New reimbursement codes for AI plaque assessment and FFR‑CT are accelerating adoption, while CCTA‑based...
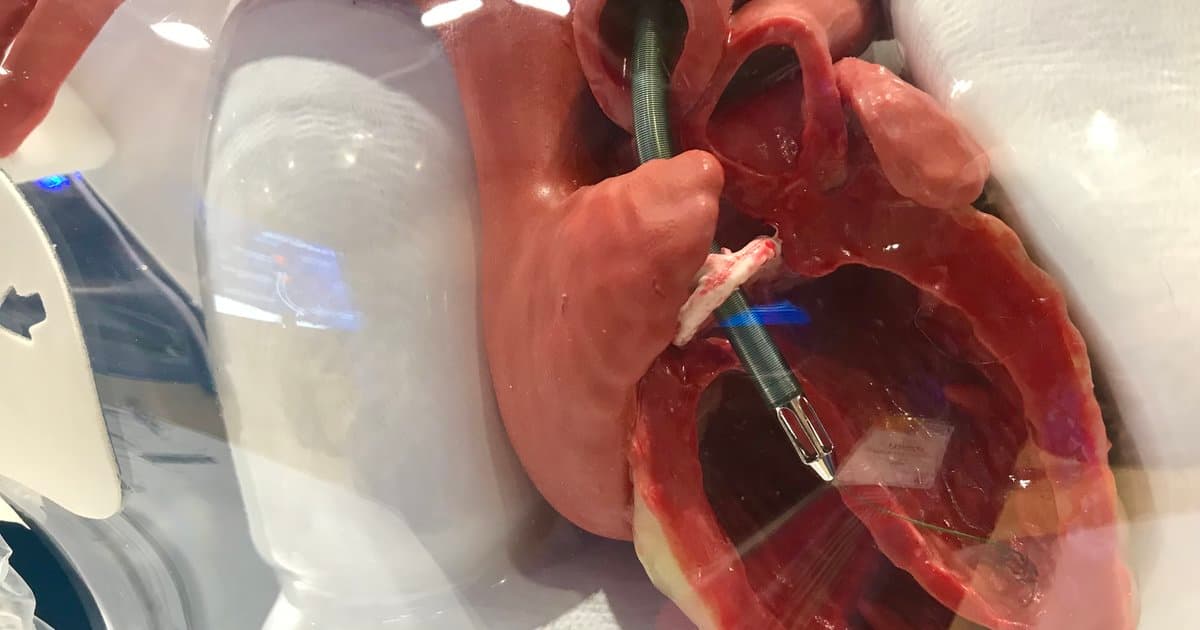
Impella 5.5 Shows Promise in Certain Non-Shock, High-Risk Cardiac Surgery Patients
The IMPACT trial presented at the 106th AATS meeting evaluated prophylactic Impella 5.5 support before weaning 100 non‑shock cardiac‑surgery patients (LVEF ≤35%) off cardiopulmonary bypass. Researchers found the strategy safe, feasible, and associated with favorable postoperative outcomes, hinting at a new...
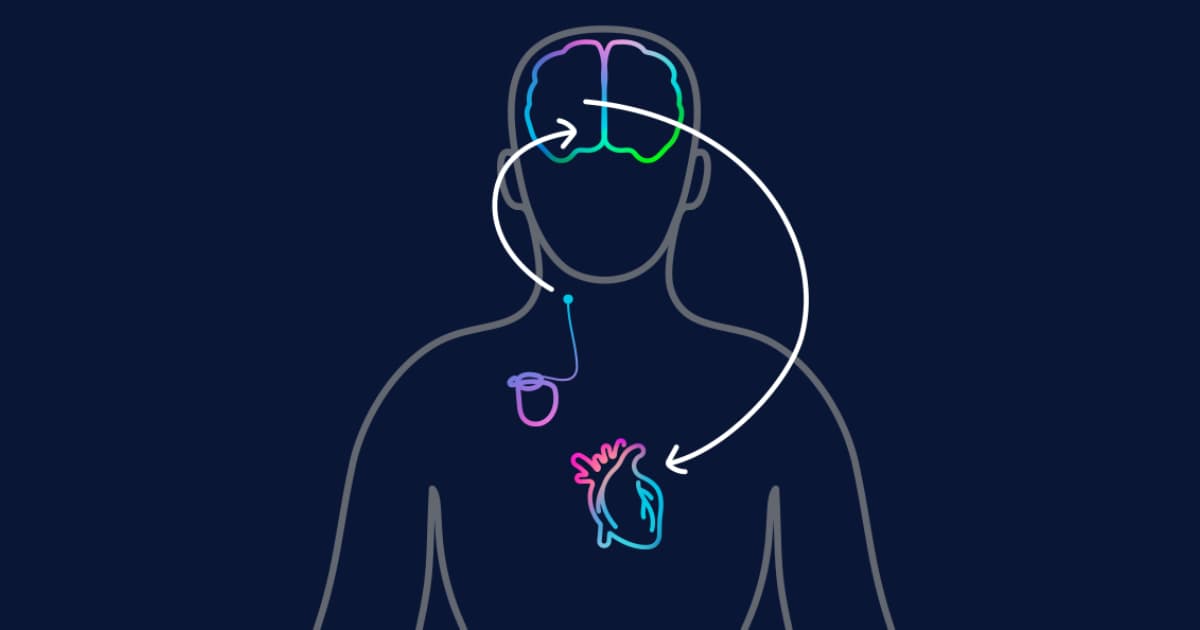
First Patient Enrolled in Massive Heart Failure Trial
CVRx has begun enrolling patients in BENEFIT‑HF, a pivotal trial of its Barostim implant for heart failure. The study plans to enroll roughly 2,500 NYHA Class II‑III patients across the United States and Germany and will run through 2032. Participants must...
TAVR or SAVR? Cardiologists and Heart Surgeons Highlight the Care that Goes Into Each Decision
Three leading U.S. cardiovascular societies—SCAI, ACC and STS—issued a joint statement reaffirming that multidisciplinary heart teams guide every decision for patients with severe aortic stenosis. The statement responds to recent Wall Street Journal coverage highlighting complications after transcatheter aortic valve...
FDA Details Class I Catheter Recall over Safety Risk
The FDA has finalized a Class I recall for Cook Medical’s Centimeter, Aurous and Beacon Tip Sizing Catheters after an April early‑alert flagged a risk of cracking or breaking during vascular procedures. A Class I recall signals a reasonable probability of serious...

Advanced AI Helps 3D Imaging Labs Evolve with the Times
Advanced AI and visualization software are reshaping cardiac 3D imaging labs, slashing processing times from three hours to about 25 minutes for TAVR planning. At Banner Health, a five‑person team now handles roughly 400 exams weekly, a workload that previously...

New Ways to Predict TAVR Outcomes for Individual Heart Patients
Two recent studies offer fresh tools for forecasting outcomes after transcatheter aortic valve replacement (TAVR). Researchers in Turkey validated the C‑reactive protein‑albumin‑lymphocyte (CALLY) index, a low‑cost biomarker that blends inflammation, immune activity and nutrition, as an independent predictor of all‑cause...

Stroke Prevention Devices for TAVR Fail to Make an Impact
A new meta‑analysis of eight randomized trials involving more than 11,000 transcatheter aortic valve replacement (TAVR) patients found that cerebral embolic protection devices (CEPDs) did not significantly lower overall, disabling, or non‑disabling stroke rates. Roughly half of the participants received...

Surgery Still Outperforms GLP-1 Drugs in Terms of Heart Health
A Mayo Clinic study of more than 800 patients compared metabolic and bariatric surgery (MBS) with GLP‑1 drugs such as semaglutide and tirzepatide. Surgery produced an average 28% weight loss versus 11% for medication and cut lifetime cardiovascular risk by...

Redo TAVR: Supra-Annular, Intra-Annular Valves Linked to Comparable Outcomes
A study of 172 redo transcatheter aortic valve replacement (TAVR‑in‑TAVR) procedures from the international PANDORA registry shows comparable one‑year outcomes regardless of whether the initial and second valves are supra‑annular or intra‑annular. The median interval between the index and redo...

Heart Rhythm 2026: Electrophysiologist Mina Chung Discusses CPR, PFA and Much More
At Heart Rhythm 2026, HRS President Mina Chung outlined a multi‑pronged agenda that includes a new task force to expand CPR and AED training in high schools, leveraging digital‑health apps for continuous skill retention. The society also launched a real‑world...
Doctors Agree: There’s a Lot to Love About Cardiology’s Future
A Medscape survey of nearly 6,000 U.S. physicians shows 59% of cardiologists are optimistic about the short‑term outlook and 48% about the long‑term, placing cardiology in the top ten specialties for confidence. Only 10% express short‑term pessimism and 24% long‑term...

Women Live Longer After TAVR than Men
New research published in Heart, Lung and Circulation shows that women have superior long‑term survival after transcatheter aortic valve replacement (TAVR) compared with men. The analysis of 600 patients treated at a high‑volume Norwegian center between 2012 and 2019 found...

PFA's Dominance and Other Key Electrophysiology Trends on Display at Heart Rhythm 2026
Pulse‑field ablation (PFA) is emerging as a durable, safer alternative for atrial fibrillation, delivering higher first‑pass isolation rates and fewer repeat procedures. Experts predict a dual‑energy future where PFA and radiofrequency ablation (RFA) are combined to treat thicker tissue and...
Virtual Nursing Staff May Help Save Cardiology Departments From Burnout
Cardiology departments are facing rising physician burnout as administrative duties eclipse patient care, prompting health systems to seek innovative staffing models. MedStar Health and Abundant Venture Partners launched Auxira Health, a startup that supplies remote clinical pods of advanced practice...

IV Fluids Recalled over Possible Contamination—Patients Face Risk of Vascular Complications
B. Braun Medical is recalling two lots of its 1‑liter Lactated Ringer’s IV electrolyte solution (lots J4P756 and J4S843) after detecting particulate matter that could trigger pulmonary emboli, vessel occlusions, or phlebitis. The affected bags were distributed across the United States...

FDA Warns Public About Vascular Device After 3 Deaths
The FDA issued an early‑alert notice on Terumo Aortic’s Relay Pro Thoracic Stent Graft system after three deaths were linked to a failure of the device to unclasp from its delivery system. The warning applies to the non‑bare stent configuration sized...
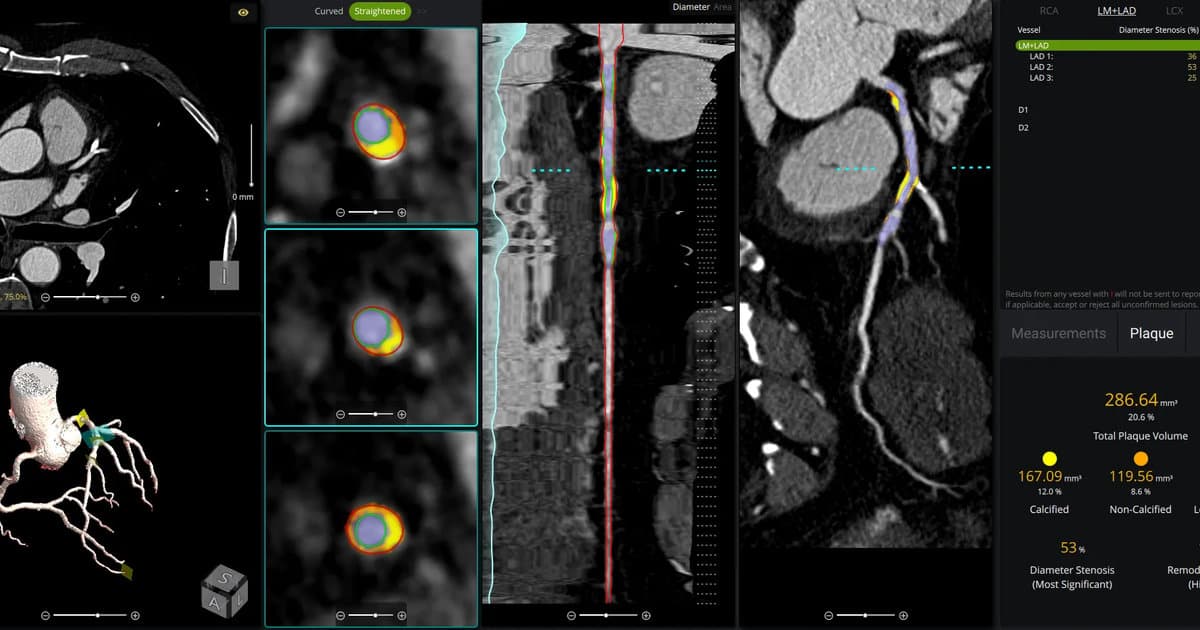
How to Implement AI-Powered Coronary Plaque Analysis Software—And Ensure You Get Paid
Artificial intelligence is now being used to analyze coronary plaque in CT angiography, offering detailed, non‑invasive assessments of coronary artery disease. Medicare expanded coverage and introduced Category 1 CPT codes in 2024, allowing providers to be reimbursed for AI‑driven plaque analysis....
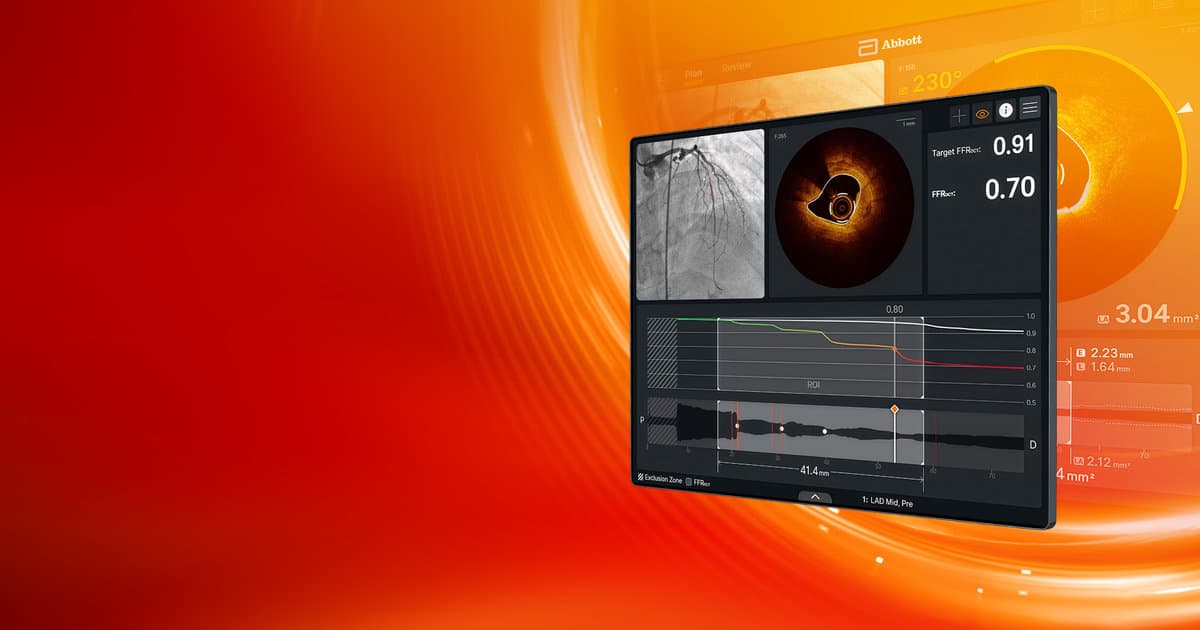
Abbott Gains FDA Clearance, CE Mark Approval for AI-Powered Imaging Platform
Abbott announced that its next‑generation coronary imaging platform, Ultreon 3.0, has received both FDA clearance and CE mark approval. The AI‑enhanced system captures optical coherence tomography (OCT) images and delivers real‑time guidance for percutaneous coronary interventions (PCI). By integrating advanced algorithms,...

New SCAI President Dawn Abbott Outlines Workforce, Training and Inclusion Priorities
Dawn Abbott, MD, has taken the helm of the Society for Cardiovascular Angiography and Interventions (SCAI) and is prioritizing workforce sustainability, expanded training pathways, and greater inclusion in interventional cardiology. She highlighted a mismatch between the growing number of fellowship...

Boston Scientific Pacemakers Recalled Again—Software Update Now Available
The FDA issued a Class I recall affecting more than 1.4 million Boston Scientific pacemakers after reports that a battery‑related flaw could force devices into a permanent safety mode, resulting in over 800 injuries and two deaths. Boston Scientific has released a...
Multiple GLP-1 Drugs Linked to Lower AFib Risk
A retrospective analysis of 13,034 patients who started GLP‑1 receptor agonist therapy between 2020 and 2024 found a significant reduction in atrial fibrillation (AFib) incidence compared with a propensity‑matched cohort of over 385,000 untreated individuals. The benefit persisted regardless of...

Many High-Risk Pregnant Patients Still Miss Out on Guideline-Recommended Care
New research from the Mass General Brigham system reveals that only about a quarter of high‑risk pregnant patients receive the guideline‑recommended low‑dose aspirin to prevent preeclampsia. The study, covering more than 60,000 pregnancies from 2013 to 2023, shows aspirin use...

First Bedside Procedure of Its Kind Performed by Traveling Clinicians on Premature Infant
Cardiologists in Florida performed the first traveling bedside transcatheter patent ductus arteriosus (PDA) closure on a 22‑week‑old premature infant. Led by Dr. Shyam Sathanandam at Nicklaus Children’s Hospital, the minimally invasive procedure was completed in the NICU isolette, eliminating the...
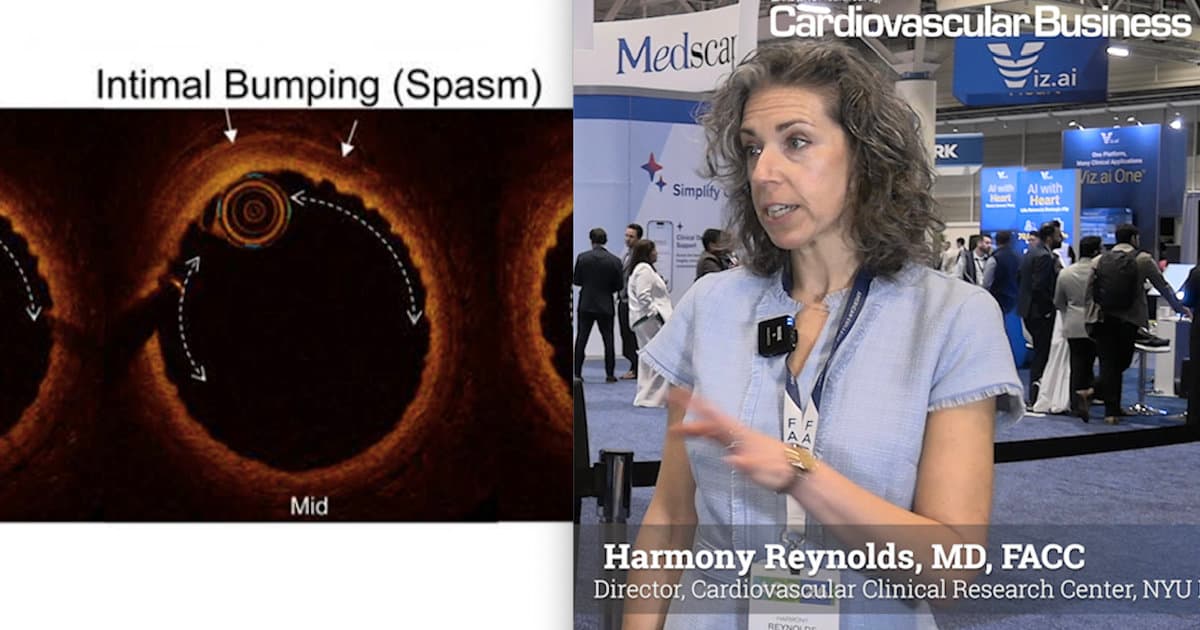
Researchers Use Multi-Modality Imaging to Learn More About MINOCA
Researchers at NYU Langone Health used combined optical coherence tomography and cardiac MRI to uncover the underlying causes of myocardial infarction with nonobstructive coronary arteries (MINOCA) in a large mixed‑sex cohort. The multi‑modality approach identified a definitive cause in 79%...

FDA Approves 2 Implantable Heart Devices From Biotronik
The FDA has cleared Biotronik’s Acticor Sky and Rivacor Sky implantable cardioverter‑defibrillator (ICD) and cardiac resynchronization therapy defibrillator (CRT‑D) systems, set to launch at the Heart Rhythm Society 2026 meeting. The devices introduce left bundle branch area pacing, single‑lead DX atrial sensing,...

Trump Administration Moves to Speed up Medicare Coverage for Breakthrough Cardiology Devices
U.S. CMS and FDA have unveiled the Regulatory Alignment for Predictable and Immediate Device (RAPID) pathway, a joint proposal to accelerate Medicare coverage for FDA‑designated breakthrough devices. Under RAPID, a device’s full FDA approval would trigger a 30‑day public comment...