
IL-15 Super‑Agonist ANKTIVA: Longevity’s Hidden Cancer Breakthrough
In 2024 the FDA approved a drug for bladder cancer. It is now the most interesting molecule in longevity medicine that no one is talking about. The drug is ANKTIVA - an IL-15 super-agonist. IL-15 is the body's signal for keeping killer immune cells (NK cells, CD8+ memory T cells) young. It is also released by your skeletal muscle every time you lift weights. (1/5)

Meta Biohub Unveils Protein‑Design AI Tackling Cancer
Mark Zuckerberg and Priscilla Chan’s Biohub just released new Evolutionary Scale Models, to map, predict, and design proteins, with system showing results against cancer and immune disease targets https://t.co/GcRGpatDrL #ArtificialIntelligence #Innovation #Technology #Tech #TechNews
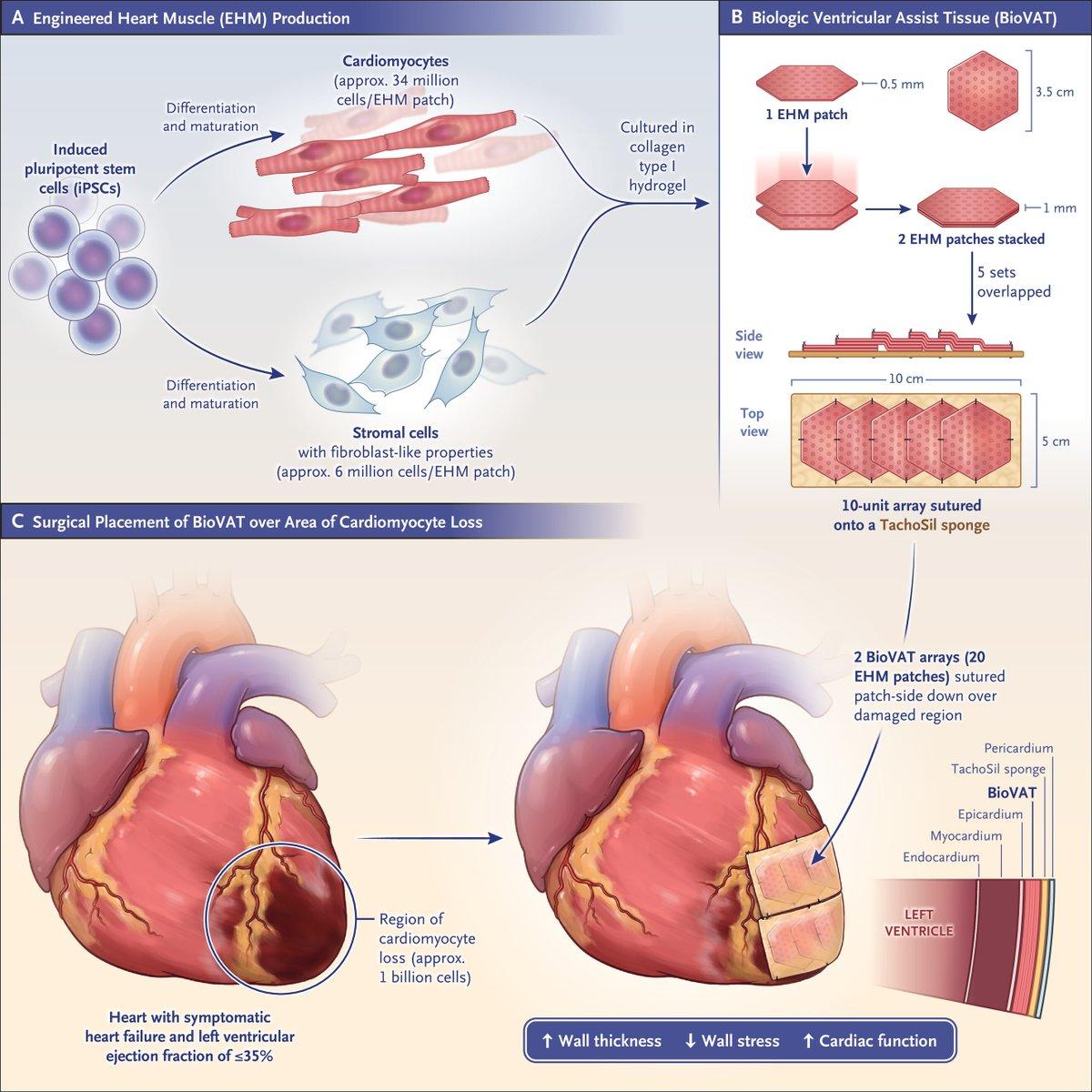
Stem‑cell Heart Patches Improve Outcomes in 12 of 20 Patients
Cardiac "remuscularization" for treating severe heart failure with patched heart muscle derived from stem cells (a biological ventricular assist device) successful in 12 of 20 patients @NEJM https://t.co/mAwgj2rAwE https://t.co/3ogxaJFDE0 https://t.co/J1ozMsS4Oh
Perioperative Ap
#ASCO26 LBA1 - Taplin - Perioperative APA + ADT - prostate CA [May 31, 2026] https://t.co/oWEeVo2oyq #NCT03767244 #pcsm
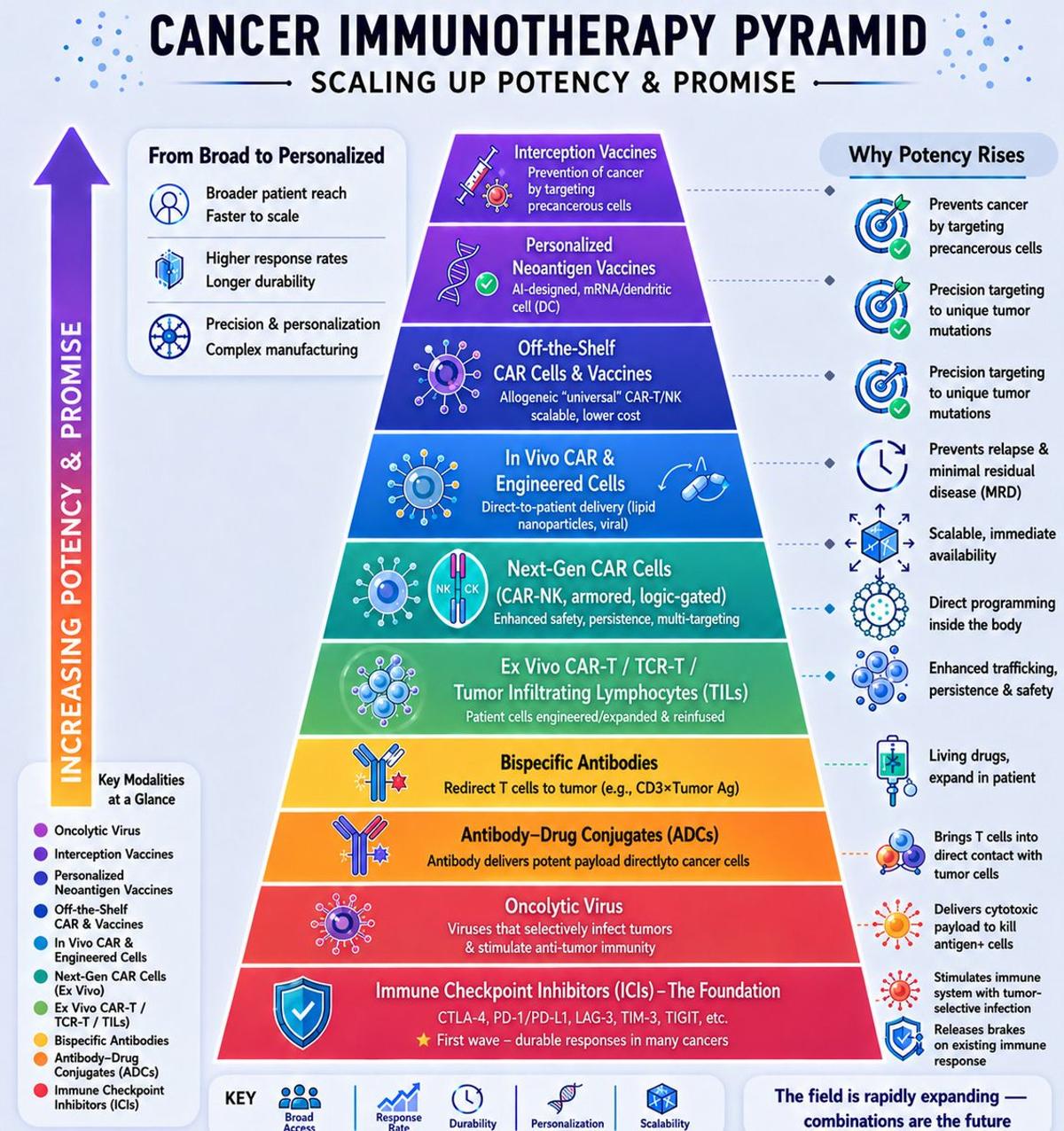
Vaccines Deliver Remissions in Hardest Cancer Types
The ramp up of cancer immunotherapy is remarkable. Now we're seeing vaccines achieve some cures or remissions in the most refractory cancers: pancreatic, melanoma, glioblastoma, renal, triple-negative breast cancer. ✓ out the new Ground Truths (link in profile) https://t.co/8PE5nOMfj1

Lilly's Verve Gene Editor Cuts Cholesterol, but Delays Loom
Eli Lilly $LLY says Verve’s gene editor lowers cholesterol levels in early, but development timelines have slipped. https://t.co/fuJOPyDwQb via @Jasonmmast

Tri‑complex RAS Inhibitors: 40k Compounds,
Came across this great talk from $RVMD's Adrian Gill on @drughunter_com about the development of probably one of the most important oncology drug classes in history : Tri-complex RAS inhibitors. It's not too technical so non-chemists can follow as well. Synthesized...
Money, Patience, Intent—And Animal Models Drive Drug Discovery
After we showed that we can discover a drug in the UAE from scratch, many countries and their R&D organizations started reaching out with the offers "set up a site in our country and gain access to our wonderful population...
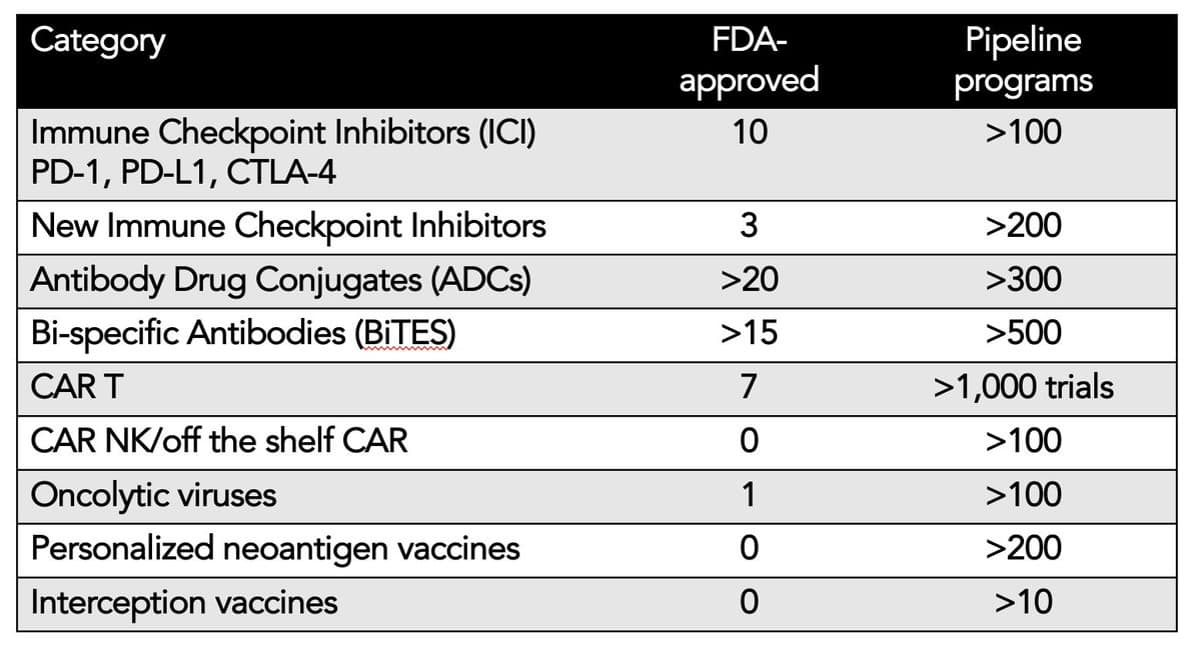
Cancer Immunotherapy Surge: Emerging Treatments and Future Outlook
I wrote about the remarkable proliferation of cancer immunotherapies, and what's in store https://t.co/od4MeFMGik https://t.co/5jI10LLbLq

Leukemia Drug HHT Delays Aging, Extends Mouse Lifespan
Homoharringtonine exhibits senotherapeutic activity that mitigates diet- and age-associated obesity and insulin resistance and extends lifespan in mice [an FDA-approved anti-leukemic drug; authors found that "HHT treatment delays aging and extends the lifespan in progeroid and aged mice"] https://t.co/LEegM8XUtR

GLP‑1 Cancer Link Unclear, Dedicated Trials Needed
Whether there is a real impact of GLP-1 drugs on cancer is unresolved. If confirmed, it could simply reflect weight loss or, as seen for other conditions (e.g. heart, kidney), weight loss independent effects. We need dedicated trials to resolve...

20 Phase‑3 Trials Poised to Redefine Cancer Care
In the wake of the #ASCO26 data drop, we reflect on 20 key phase 3 trials which could make a significant impact. Some of these may make their competitors irrelevant if they change the standard of care: https://t.co/EGtw35EsSk https://t.co/CG0Oe4QxDg

Genomics Predict Venetoclax Response in T(
Genomic Determinants of Clinical Outcomes in Multiple Myeloma with t(11;14)(CCND1;IGH) Treated with Venetoclax [Dec 7, 2024] @MKaddouraMD et al. @lbaughn #ASH24 Abst 249 https://t.co/TLon5bCQ3i #mmsm #PrecisionMedicine

New Trial Compares Dara‑Bor‑Dex vs Cy‑Bor‑Dex for Myeloma‑Induced Kidney
EAA241 - Ph 2 RCT Dara-Bor-Dex vs Cy-Bor-Dex in the treatment of Newly Diagnosed Multiple Myeloma with Light Chain Cast Nephropathy (LCCN) [Study activated 8/11/25] @keruakous https://t.co/1NgvVZ3fTA #NCT07085728 #mmsm @eaonc

Incyte Leverages AI Partnerships to Accelerate Future R&D
35-year-old Incyte is turning to AI to power through the coming decades, with new R&D deals with Edison Scientific & Genesis Molecular AI: https://t.co/5rinB9m8m4

Retatrutide Delivers Record 28% Weight Loss, Raises Safety Concerns
Retatrutide, a triple receptor drug for GLP-1, GIP, and glucagon, is the most powerful weight loss drug yet. A significant issue is too much weight loss among the trial participants. New randomized trial results announced today with 28% body weight...

Retatrutide Delivers up to 30% Weight Loss in Phase 3 Trials
Retatrutide just delivered some very impressive phase3 results for $LLY at 4mg, 9mg and 12mg -- https://t.co/7sKbaDoInq Retatrutide is their GLP-3 ie triple agonist which hits the GLP, GIP and Glucagon receptors. At the primary endpoint of 80 weeks... patients on 4mg,...

IL-6 Blocker Shows Promise for Treating Depression
Would depression respond to an anti-inflammation drug (interleukin-6 blocker)? A small pilot, placebo-controlled randomized trial suggests this might be possible https://t.co/zKO76yvRAo https://t.co/e8K9YLEAQj
Longevity Biotech Matures: Precise Indication‑Focused Drug Development
The longevity biotechnology field is coming of age in the best possible way. A recent analysis captures how the sector has learned to speak pharma’s language fluently—moving from broad claims about “slowing aging” to precise, indication-focused drug development with conventional endpoints,...

BARDA Ships Experimental Ebola Antibody to Protect High‑risk Americans
BARDA is coordinating shipment of an experimental antibody treatment for potential use in high-risk Americans exposed to #ebola, HHS tells CNN While they didn't name the treatment, Mapp's MBP134 has shown activity in animal studies against Bundibugyo strain

AI‑Powered Consulting Cuts FDA Approval Time and Cost
Getting FDA approval is slow and very expensive. @Panacea_Bio hires the best FDA regulatory consultants in the industry, and pairs them with their AI platform, to deliver the fastest and lowest-cost pathway to FDA approval for Biotechs and MedTechs. https://t.co/OvR0zEYr7Y https://t.co/eIaiDbwmSx

Kazia’s Paxalisib Shows >50% CTC Drop, Early Responses
Kazia Therapeutics ($KZIA) is a catalyst-driven setup centered on upcoming ASCO data from their Paxalisib drug development platform. The update is following earlier disclosures from their ongoing Phase 1b ABC-Pax study evaluating paxalisib in metastatic triple-negative breast cancer (TNBC) showing...

Merck's China-Licensed ADC Succeeds in First Global Trial
Merck ADC, licensed from China, hits mark in first big global trial https://t.co/tAvLkw6pYm @ByJonGardner $MRK $GILD $AZN

Once‑night Pill Proves Effective for Severe Sleep Apnea
A once a night pill for severe obstructive sleep apnea effective in a Phase 3 randomized trial https://t.co/zq2Ew65WHK https://t.co/ovQEh7taeN https://t.co/N8vXzYincy
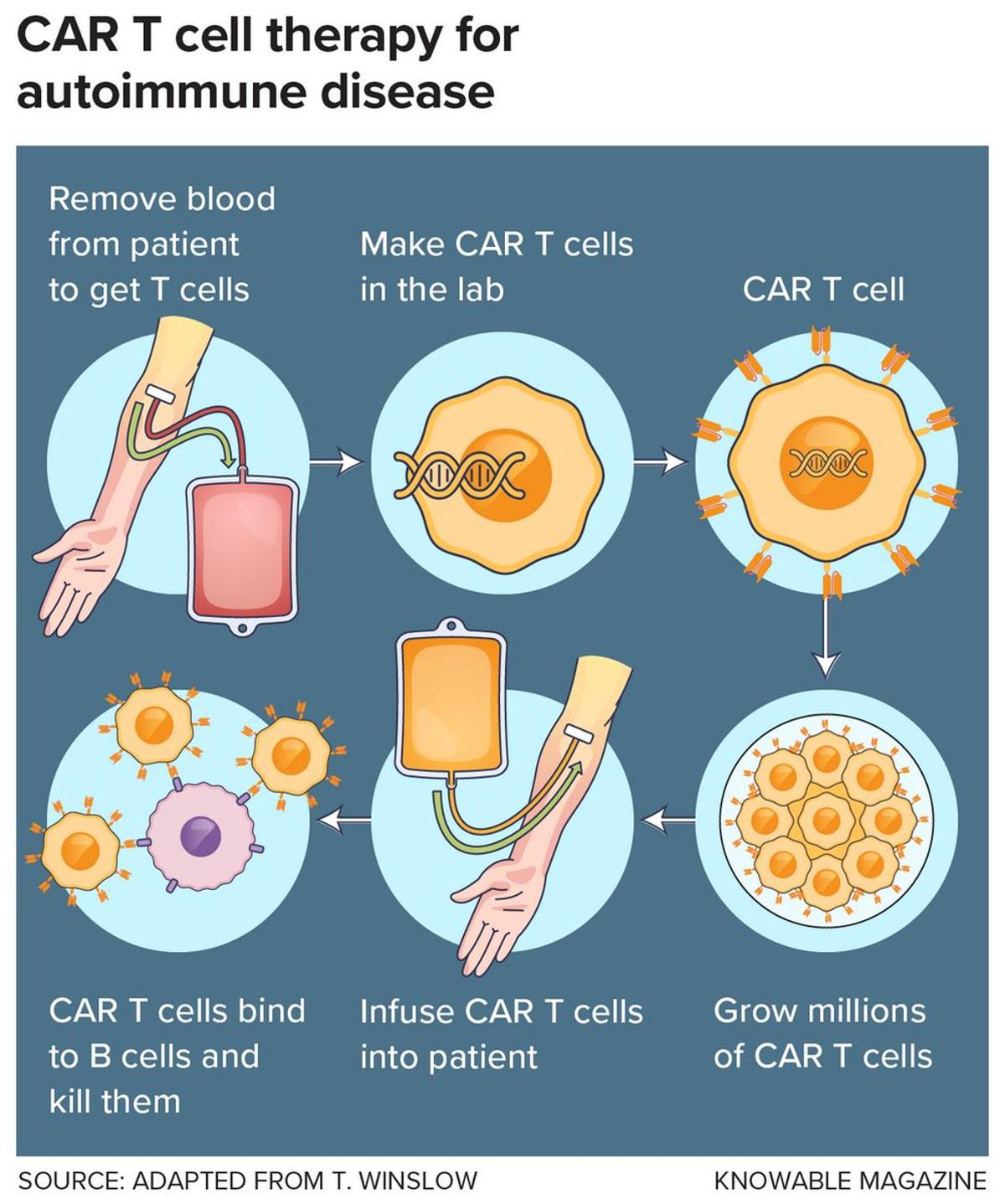
CAR‑T Therapy Shows Promise for Autoimmune Diseases
CAR T therapy, originally developed to fight blood cancers, is now being tested as a potential way to “reset” the immune system in autoimmune diseases like MS, lupus, Graves’ disease and stiff person syndrome. Early results are promising, but risks remain,...
ABBV's BLA for Pivekimab Sunirine Remains Pending
$ABBV announced BLA submission for pivekimab sunirine back in Sep 2025, but no news since on whether this was accepted/Pdufa date set.
FDA Greenlights Enhertu for Early HER2‑positive Breast Cancer
#AZN Enhertu approved by US FDA both before and after surgery for treating patients with HER2-positive early breast cancer.

Repeated Psilocybin Doses Show Safety, Promise for OCD
A randomized clinical trial of repeated doses of psilocybin for the treatment of obsessive–compulsive disorder 🍥"Administration of up to eight doses of psilocybin in a clinical research setting appears to be safe and potentially effective for patients with OCD. Larger trials...

FDA CDER Acting Director Tracy Hoeg Set to Leave
FDA Center for Drug Evaluation & Research (CDER) Acting Director Tracy Beth Hoeg expected to depart from agency - Reuters
Ensitrelvir Outperforms Paxlovid yet Faces Approval Hurdles
Ensitrelvir is pretty clearly also a better acute treatment than Paxlovid. More potent and many fewer drug-drug interactions. Just proved too hard to get approval for acute treatment.

Chemo-Free Padcev‑Keytruda Shows Promise for Cis‑eligible Patients
$AZN Volga might have matched $MRK $PFE KN-905 in cis-ineligible patients, but here comes chemo-free Padcev + Keytruda in the cis-eligible setting. Via @ApexOnco -> https://t.co/KmwCfgZLBm

RGNX Shows 71% Microdystrophin, Eyes 2027 Approval
$RGNX Duchenne gene therapy study results are out: microdystrophin expression reached an average of 71% of normal across all 31 boys at 12 wks. Functional improvements in a subset of boys followed for one year also seen. The big question...

Therapeutic Peptides: The Next Pillar of Modern Medicine
I just finished reading one of the most comprehensive research reviews ever published on therapeutic peptides, and it explains why peptides are becoming one of the most important categories in modern medicine. Here's what you need to know:
Adenine Base Editing Reverses Dravet Syndrome in Mice
Today in @ScienceTM, we report the use of in vivo adenine base editing to correct a variant causing Dravet syndrome, a severe childhood epilepsy and neurodevelopmental disorder, substantially ameliorating disease symptoms and extending lifespan in an animal model. 1/13 https://t.co/uQEwWvj94y

New mRNA‑nanoparticle Vaccine Boosts Cancer Potency, Durability
An innovation taking mRNA-nanoparticle cancer vaccines to the next level for potency and durability @NatureBiotech https://t.co/6wBToMGgEA
Oral Pill
Two new randomized studies just published @NatureMedicine for weaning from injectable GLP-1 drugs to maintain weight loss —Pill, orforglipron https://t.co/r9Nx9J9vJg —Bacterial supplement for gut microbiome https://t.co/km2V3NkIOX

Marty Makary: The Worst FDA Commissioner in 25 Years
And here's @matthewherper on why Marty Makary was the worst FDA commissioner in 25 years https://t.co/a44yjB5sZn

LSD Triggers Region‑Specific White Matter Plastic
Curious. Opposite DTI/FA findings to those we recently published on. Region specific? Neuroplastic white matter changes in patients with major depression following lysergic acid diethylamide treatment: Cell Reports Medicine https://t.co/sIXRAZumie
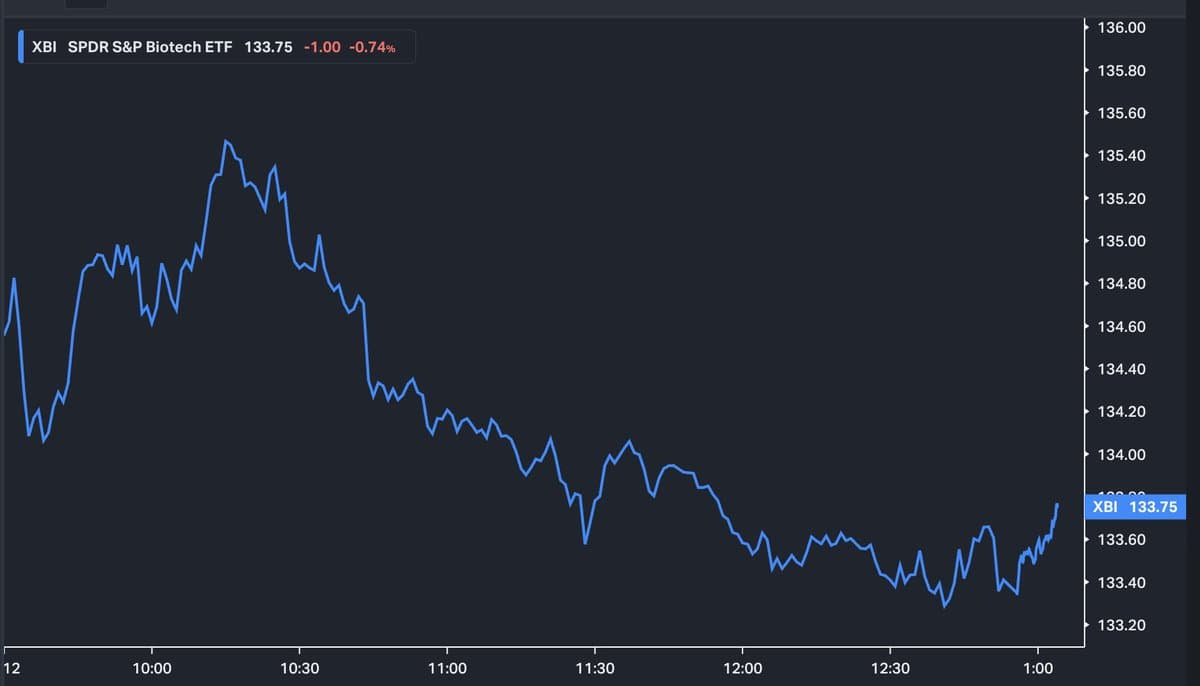
XBI Sees Modest Boost After Anticlimactic FDA Decision
$XBI getting small lift (so far) from the Un-Martying of the FDA. It's all very anticlimactic. https://t.co/q4ta0bkJh4

Arvinas, Pfizer Secure New Partner for PROTAC Breast Cancer Therapy
Arvinas, Pfizer find new partner for ‘Protac’ breast cancer drug https://t.co/IKMKNP1Zfk by @Lilah_Alvarado $ARVN $PFE $RIGL + 7%

Programmable Drug Targets Cancer Cells via DNA Recognition
FinalDose is building the first programmable drug platform - a single smart drug molecule that finds diseased cells by their DNA and destroys them. They're starting with all cancers. Congrats on the launch, @Jeffliu6068Liu, @sklin_lite, and @liyaohuang2! https://t.co/uKJgl7lpmR https://t.co/l4b1hS2mn7
Personalized Neoantigen Vaccine Shows Promise Against Glioblastoma
Personalized neoantigen vaccines vs cancer: we've seen benefits for pancreatic, renal and melanoma. Today initial, encouraging results vs glioblastoma in 9 patients with this deadly brain cancer @NatureCancer https://t.co/ACdCjiXnjJ
RIGL to Sell Approved Veppanu for ARVN, PFE
$RIGL will sell the newly approved Veppanu for $ARVN and $PFE. Modest financial terms https://t.co/9RsbSMzxBf
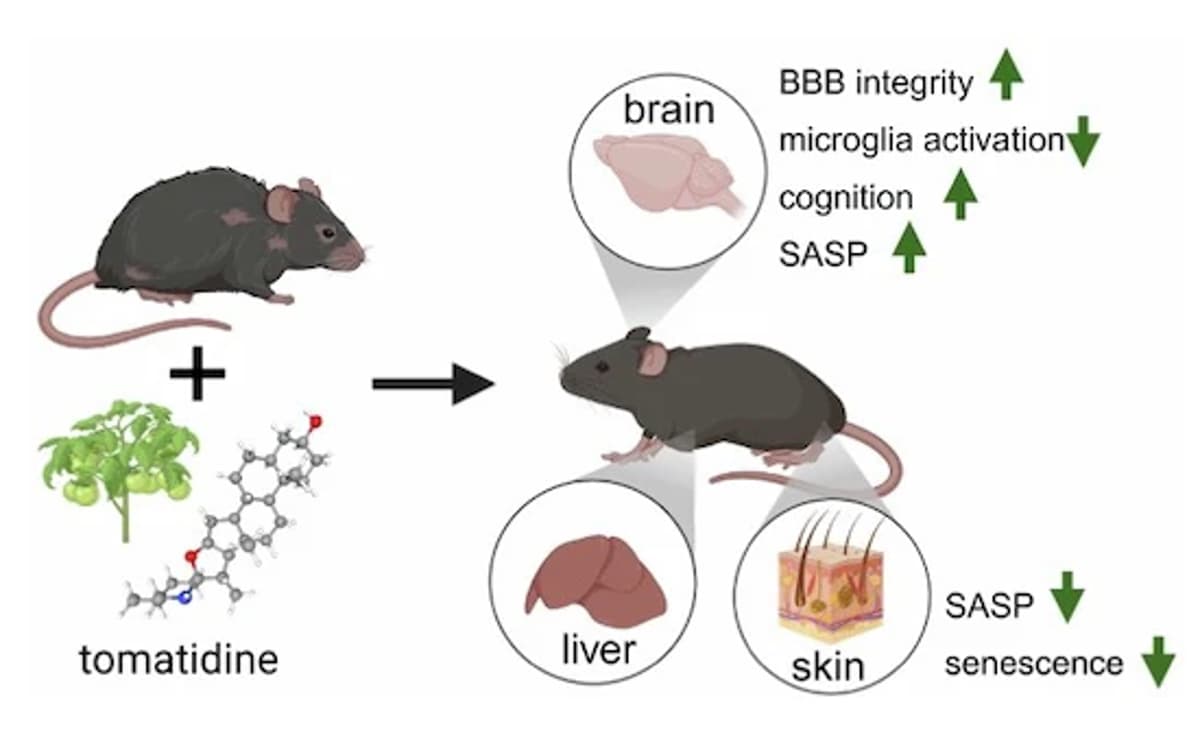
Tomatidine Boosts Memory and Cuts Cellular Aging in Mice
Tomatidine is a senotherapeutic compound that improves cognitive function and reduces cellular senescence in aged mice https://t.co/jVfshXgzxQ https://t.co/6l86CBdoBC
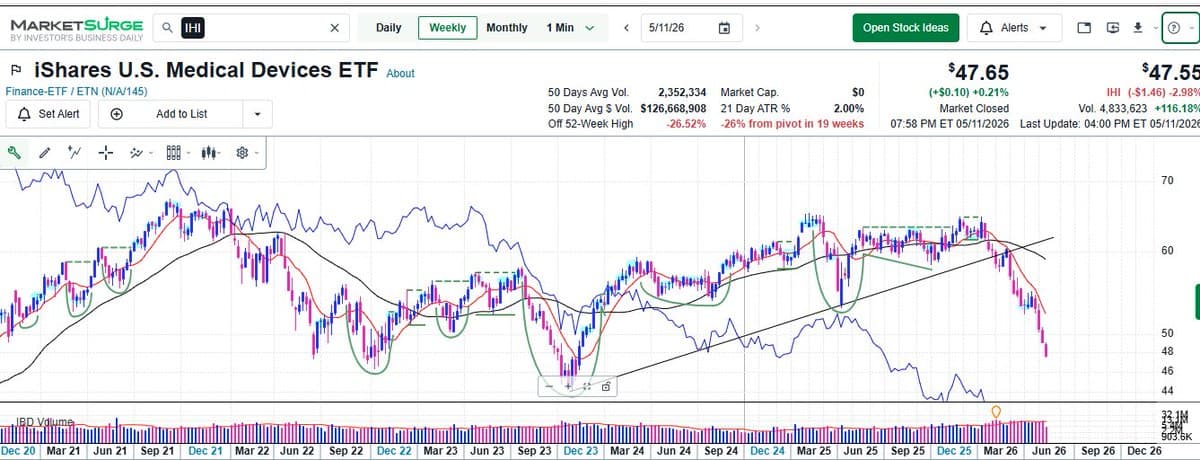
GLP‑1 Disruption Drives Healthcare to 13‑year Low
GLP-1 disruption has caused the concentrated Med-Tech ETF $IHI to weigh sharply on Healthcare's performance this yr, resulting in what used to be the 2nd largest sector in SPX slipping to just 8% @IBDinvestors @marketsurge Selectivity crucial until this Sub-industry...

EnGene's Phase II Data Triggers 83% Sales Forecast Plunge
11May: How much have peak sales forecasts nosedived after $ENGN reported updated Phase II data for detalimogene? Read about enGene Therapeutics' 83% plunge plus an IPO roundup and updates on $ATRA, $TRDA, $MRNA, & $VRDN in my latest StockWatch for...
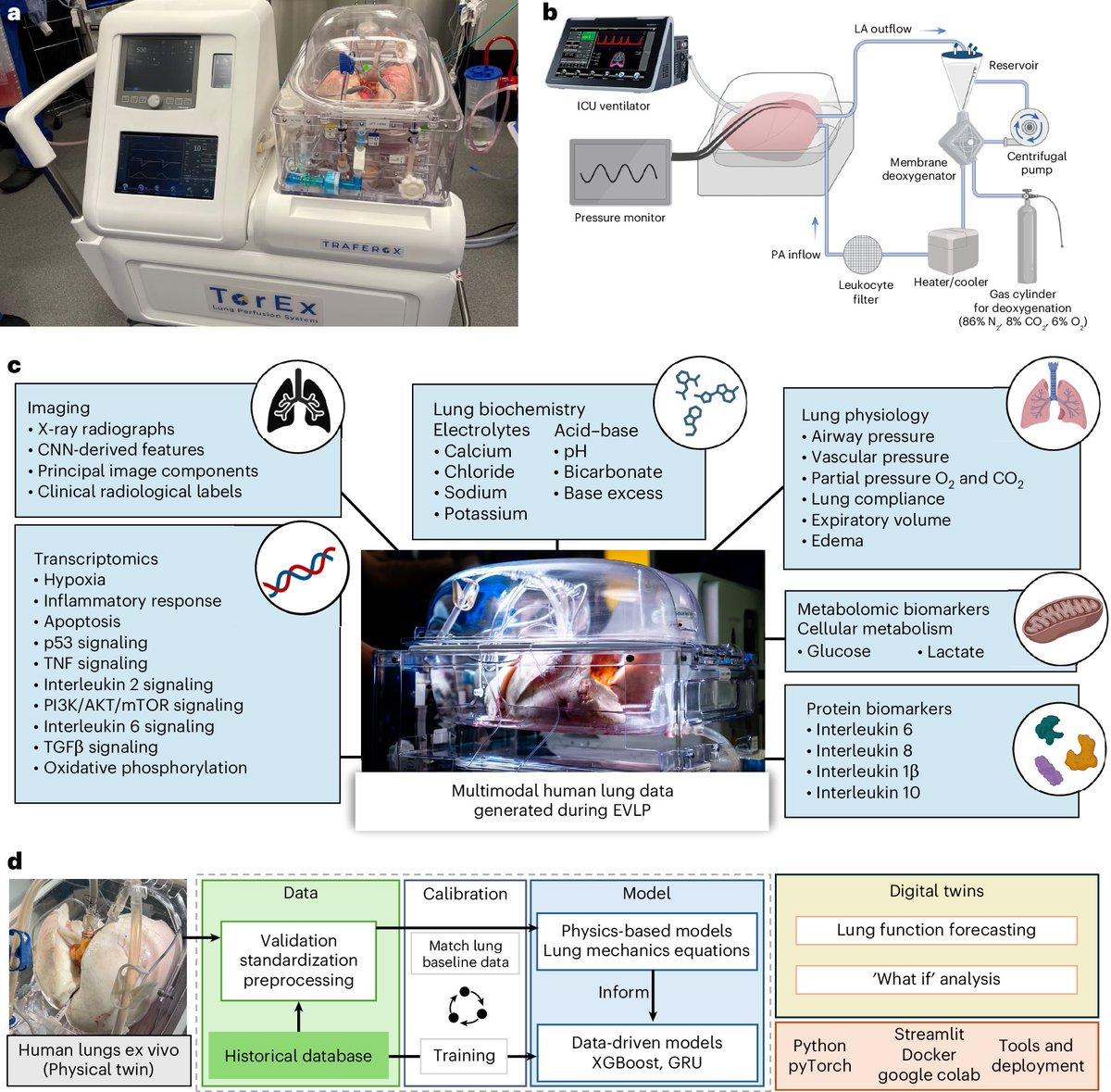
Digital Twins of Human Lungs Personalize Therapy Assessment
Digital twins of ex vivo human lungs enable accurate and personalized evaluation of therapeutic efficacy https://t.co/0FMBqmiMyM https://t.co/3MrdTqK82w

DOSE Trial Reveals Best Heart Failure Infusion Strategy
Bolus or drip? What the DOSE trial actually showed about heart failure [PODCAST] http://dlvr.it/TSV5l4 Podcast #Cardiology

New AI Target Discovery Paper Sets Benchmark
New Paper Alert 🚨: A collaboration with the leading target ID experts in Nature Reviews Drug Discovery. If you are looking to teach your AI target discovery - this is a good place to start. Thanks Peter Kirkpatrick for giving...

Supreme Court Allows Abortion Pill Access for Three Days
JUST IN: The Supreme Court grants a widely accessed abortion pill temporary availability for at least three days.