Today's Healthcare Pulse

FDA greenlights durvalumab combo for high‑risk bladder cancer
The FDA approved durvalumab (Imfinzi) combined with Bacillus Calmette‑Guerin for BCG‑naïve, high‑risk non‑muscle invasive bladder cancer. The POTOMAC trial enrolled 1,018 patients and showed a 32% reduction in disease recurrence risk (hazard ratio 0.68, p=0.015). Durvalumab is given at 1,500 mg IV every four weeks for up to 13 cycles.
Also developing:
By the numbers: Apogee Therapeutics raises $1.3B royalty financing

Regeneron Will Offer Rare Disease Therapy Free As Part Of TrumpRx Deal
Regeneron announced it will provide its newly approved gene therapy for a rare pediatric hearing loss at no cost to U.S. patients under the TrumpRx program. The therapy, cleared by the FDA earlier this year, treats an X‑linked condition affecting fewer than 1,000 children. The White House revealed the partnership on April 23, 2026, positioning the deal as a flagship example of drug‑price reform. By offering the treatment for free, Regeneron aims to improve access while navigating a high‑price biotech market.

Specialty Pharmacy's Critical Role as Advanced Therapies Move Into the Community: Fran Gregory, PharmD, MBA
Advanced therapies are set to expand dramatically, rising from 61 approved products today to roughly 180 by 2030. The growth includes cell and gene therapies, bispecific antibodies, and next‑generation monoclonal antibodies, many targeting high‑volume diseases such as cardiovascular and metabolic...
First Bispecific-ADC Combo Shows Strong Results in R/R Mantle Cell Lymphoma
A phase‑2 study published in Blood shows that combining the bispecific antibody mosunetuzumab with the antibody‑drug conjugate polatuzumab vedotin yields an 88.1% overall response rate in relapsed or refractory mantle‑cell lymphoma (MCL). The regimen produced a 78.6% complete response rate,...

Why Experiential Consent Is Replacing Traditional Medical Consent Forms
Traditional medical consent forms are written at a 15th‑grade level, creating a comprehension gap that courts increasingly deem ineffective. Recent audits in ophthalmology show over 500 of 3,400 post‑operative reviews cite misunderstanding of visual outcomes. The legal shift to the...

Pan-RAS Inhibitor ADCs Emerge at AACR 2026
At the 2026 American Association for Cancer Research meeting, four biotech companies unveiled antibody‑drug conjugates that deliver pan‑RAS inhibitors directly to tumor cells. Pan‑RAS inhibition has recently delivered unprecedented survival improvements in pancreatic and other RAS‑mutant cancers, but its clinical...

Breast Cancer on the Rise in Women Under 50: Rani Bansal, MD
The American Cancer Society’s 2025 report shows women under 50 now face an 82% higher breast‑cancer risk than men, up from 51% in 2002. Incidence of estrogen‑receptor‑positive tumors is climbing, driven by later child‑bearing, fewer births, reduced breastfeeding, rising obesity...

Digital Health Technologies (DHTs) for Drug Development
The FDA has issued a Request for Information and Comments on the use of Digital Health Technologies (DHTs) in drug and biologic development, with a comment deadline of June 1, 2026. Under PDUFA VII, the agency will publish a DHT framework, establish a...

Harvard Study Finds 12‑Minute Daily Meditation Cuts Stress Hormones in Two Weeks
Harvard Medical School researchers demonstrated that a 12‑minute guided mindfulness session each day for two weeks significantly reduced perceived stress and salivary cortisol in a 210‑person trial. The rapid physiological shift underscores meditation’s potential as a scalable, non‑pharmacologic intervention.
The FDA Gives the Green Light to the First Gene Therapy for Deafness
The FDA has approved the first gene therapy designed to restore hearing in patients born with the rare OTOF‑related form of deafness. Developed by Regeneron, the treatment delivers a functional OTOF gene via adeno‑associated virus directly into the inner ear....

How to Promote an Unproven Cancer Cure
A Florida‑based Wellness Company is financing sponsored posts on conservative websites and X accounts that promote a self‑reported survey claiming its ivermectin‑mebendazole pill treats cancer. The study, which surveyed 197 customers, reported 84.4% clinical benefit and 48.4% tumor reduction, but...

Insurance Denials Meet Their Match in AI-Powered Appeals
AI startups are reshaping the healthcare appeals landscape by automating denied‑claim letters and filing processes. Claimable, founded in 2023, uses a curated large‑language model to generate appeal letters for 28 conditions and 90 treatments, charging $50 per case. The company...

Grace Therapeutics Hits FDA Roadblock on GTx-104, Plans Resubmission
Grace Therapeutics announced that the FDA issued a Complete Response Letter for its GTx-104 NDA, flagging chemistry, manufacturing, and non‑clinical concerns. The company will address the items and seek a Type A meeting before resubmitting, keeping hopes alive for the first...

SELUTION DeNovo: Sirolimus DCB Safe and Effective in ACS Patients
A subanalysis of the SELUTION DeNovo trial presented at SCAI 2026 shows that the sirolimus‑eluting Selution drug‑coated balloon (DCB) is non‑inferior to drug‑eluting stents (DES) in acute coronary syndrome (ACS) patients, with 1‑year target vessel failure (TVF) rates of 5.3%...

CMS and FDA Unveil RAPID Pathway to Cut Medicare Device Coverage Time to Two Months
The Centers for Medicare & Medicaid Services and the Food and Drug Administration announced the Regulatory Alignment for Predictable and Immediate Device (RAPID) coverage pathway, a joint program that could deliver Medicare coverage for eligible breakthrough devices within two months...

How to Help Nurses Avoid Technology Over-Reliance
Healthcare leaders are urged to balance digital fluency with core clinical skills to prevent nurses from becoming overly dependent on AI tools, according to NYU informatics professor MaryAnn Connor and FANA CEO Olga Kagan. The experts stress that while AI...

Trump Administration Moves State‑Licensed Medical Marijuana to Schedule III, Offering Tax Breaks
Acting Attorney General Todd Blanche signed an order shifting state‑licensed medical marijuana from Schedule I to Schedule III, unlocking federal tax deductions and easing research rules. The move, hailed by industry groups as the biggest federal cannabis shift in decades, draws sharp...

Lunsotogene Parvec Becomes First FDA-Approved Gene Therapy for OTOF-Related Hearing Loss
Regeneron’s lunsotogene parvec‑cwha (Otarmeni) received accelerated FDA approval as the first gene therapy for OTOF‑related hearing loss. The single‑dose AAV vector is indicated for patients with severe‑to‑profound sensorineural loss who have confirmed biallelic OTOF variants and no prior cochlear implant....

Lumira Ventures Secures First $200 Million Close of Fifth Healthcare Fund
Lumira Ventures announced the first closing of its fifth healthcare-focused fund, Lumira Ventures V, LP, at $200 million. The close includes a $52 million Series B investment in a pre‑commercial medical device company and marks a leadership transition with co‑founder Gerry Brunk becoming...

LeonaBio to Host Virtual Key Opinion Leader Event Highlighting Potential of Lasofoxifene in Treatment-Resistant ER+/HER2-, ESR1-Mutated Metastatic Breast Cancer
LeonaBio announced a virtual Key Opinion Leader webcast on April 29, 2026 to discuss lasofoxifene, its novel selective estrogen receptor modulator, for treatment‑resistant ER+/HER2‑, ESR1‑mutated metastatic breast cancer. The company expects to complete enrollment in the Phase 3 ELAINE‑3 trial, which pairs...

Early ALPHA3 Data Could Signal Shift to Frontline Use of CAR T in LBCL
Allogene Therapeutics reported that its off‑the‑shelf CAR‑T product cemacabtagene ansegedleucel (cema‑cel) achieved 58.3% minimal residual disease (MRD) negativity at day 45 versus 16.7% with observation in the interim futility analysis of the phase 2 ALPHA3 trial. The study enrolled LBCL patients who...

AHA Blog Highlights How Paragon Report Gets Hospitals Wrong
The American Hospital Association (AHA) posted a blog on April 23 criticizing a Paragon Health Institute report released the day before. The AHA contends the think‑tank’s analysis rests on distorted, debunked data and recommends cuts that would shrink or shut hospital...

CDC Blocks Release of COVID‑19 Vaccine Effectiveness Study, Halting Key Data
The Centers for Disease Control and Prevention halted the publication of a study that found COVID‑19 vaccines cut emergency‑room visits and hospitalizations by roughly 50% during the last winter. The decision, made by acting CDC Director Dr. Jay Bhattacharya, has...

MedPAC April 2026 Public Meeting Summary
The Medicare Payment Advisory Commission (MedPAC) convened its April 2026 public meeting on April 9‑10, 2026. HFMA released a concise summary of the session, noting the Commission’s forward‑looking statements about potential payment reforms. The summary emphasizes that these projections reflect MedPAC’s likely...

OpenAI Unveils Free ‘ChatGPT For Clinicians’ To Aid In Clinical Tasks
OpenAI announced on April 22, 2026 a new version of its large‑language model called ChatGPT for Clinicians. The service is offered at no cost to any U.S.-verified physician, nurse practitioner, physician assistant, or pharmacist. It is designed to help clinicians...
Freeze-Dried Platelets Combat TBI Brain Swelling and Bleeding
Researchers at UCSF have shown that Thrombosomes, a freeze‑dried platelet‑derived product, dramatically reduces bleeding and cerebral edema in a mouse model of traumatic brain injury (TBI). The biologic, originally created for battlefield hemorrhage, can be stored at room temperature for...

Why Paragon Gets Hospitals Wrong: Report Ignores Reality of Care Delivery
Paragon Health Institute released a report that reduces hospitals to abstract cost and pricing models, ignoring the day‑to‑day realities of emergency care, labor‑intensive services, and community needs. The critique argues the institute misidentifies cost drivers, labeling Medicare and Medicaid payments...
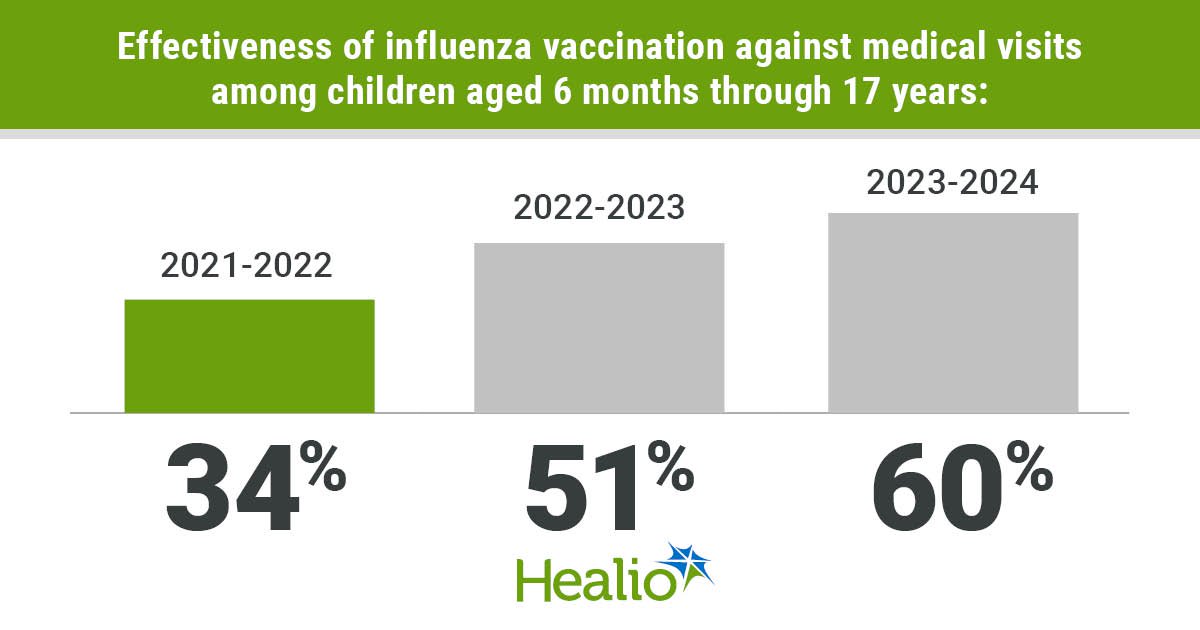
Flu Vaccines Reduced Medical Visits in Children in Recent Seasons
New research published in Pediatrics confirms that seasonal influenza vaccines cut pediatric hospitalizations and outpatient visits between 2021 and 2024. Analyzing data from nearly 20,000 children across seven medical centers, the study found overall vaccine effectiveness of 55%, ranging from...

Trump Administration Reclassifies some Medical Marijuana Products as Less Dangerous
The Justice Department, via Acting Attorney General Todd Blanche, issued an order moving FDA‑approved and state‑licensed medical marijuana products from Schedule I to Schedule III. The change reclassifies these cannabinoids as having a moderate to low potential for dependence, aligning them more...

List of Active Radioactive Drug Research Committee (RDRC) Sites
The Department of Health and Human Services has published an updated roster of active Radioactive Drug Research Committee (RDRC) sites, encompassing more than 40 institutions nationwide. The list includes premier academic medical centers such as Mayo Clinic, Stanford, and the...
TRIDENT: Triple Antihypertensive Pill Cuts Recurrent Stroke in ICH
The TRIDENT trial showed that a single low‑dose triple‑antihypertensive pill (telmisartan, amlodipine, indapamide) added to standard care reduced recurrent stroke in patients with prior intracerebral hemorrhage from 7.4% to 4.6% (HR 0.61). Mean systolic blood pressure during follow‑up was 127 mm Hg in...
Cardiometabolic Intervention: Evaluation of PCSK9 Inhibitors as the Successor to the GLP-1 Phenomenon
The 2026 analysis pits GLP‑1 receptor agonists against PCSK9 inhibitors, showing that the latter now deliver comparable or superior reductions in major adverse cardiovascular events and are expanding into oral formulations. Clinical trials such as VESALIUS‑CV demonstrate primary‑prevention benefits for...

RAS Cracked… yet the Hard Part Starts Now
A new RAS‑targeted therapy delivered a 58% overall response rate and a hazard ratio of 0.40 in previously treated pancreatic cancer, data unveiled at AACR in San Diego and slated for full presentation at ASCO. These outcomes, once thought impossible,...

ESCMID Global 2026: Adibelivir Emerges as Potential Disease-Modifying Therapy for HSV
Innovative Molecules presented Phase I/Ib data on adibelivir (IM‑250), a novel helicase‑primase inhibitor, at ESCMID Global 2026. The drug demonstrated nanomolar potency against clinical and acyclovir‑resistant HSV‑1/2 isolates and showed a favorable safety profile up to 200 mg with no dose‑limiting toxicities....

OK, Kennedy and Hegseth Can Take a Victory Lap for This One—The Mandate to Take a Flu Shot in the...
The U.S. Department of Defense announced the cancellation of the flu‑shot mandate for active‑duty, reserve, and civilian personnel. Children’s Health Defense (CHD) claims credit for the policy shift, framing it as a win for individual liberty. The blog’s author predicts...
The Skinny on Skinny Labels: The Active Inducement Problem That Patent Practitioners Should Know
The U.S. Supreme Court will hear Hikma Pharmaceuticals v. Amarin Pharma, a case that tests whether a generic maker can be liable for induced patent infringement despite using the FDA’s skinny‑label pathway. The dispute centers on Hikma’s generic icosapent ethyl, which omitted...

Watch: Acknowledging Health Care’s Great Divide
In a recent KFF Health News interview, former Obama adviser and health‑policy scholar David Blumenthal explained why fixing America’s health‑care system is so difficult. He highlighted the president’s outsized, often overlooked, authority to shape health policy and the entrenched partisan...

How Dermatologists Are Helping People Who’ve Been Sex Trafficked
Dermatologists across the United States are increasingly offering free tattoo‑removal services to survivors of sex trafficking, turning a visible mark of abuse into a pathway toward healing. The New York Times highlighted survivors like Kathy Givens and Melody Montemayor, who underwent multiple laser...

CMS Tells Govs To ‘Swiftly’ Revalidate Providers As Medicaid Programs Craft Broader Strategy
CMS Administrator Mehmet Oz sent letters to all 50 state governors on April 23, 2026, urging a swift revalidation of Medicaid providers operating in high‑risk areas. The directive gives states 30 days to complete the revalidation and to outline a broader, long‑term strategy for...

New Waves of Hospice Executives Take the Lead
A series of senior‑level appointments are reshaping hospice leadership across the United States. Becky Tooker becomes president of Hosparus Health, bringing 25 years of hospice and home‑health expertise. Bristol Hospice adds three regional executives—Valerie Meyer, Marriza Negrete and Jason Hill—to manage...
Fertility Clinics Sell IVF; Seek Holistic Care Elsewhere
Every week I talk to couples who are struggling to conceive. They come to us because they feel completely lost and in the dark about their own fertility. And almost all of them have the same story. They went to...

Trump’s Drug Pricing Deals Underdeliver on Promised Cuts
Trump celebrated closing the first round of 17 drug pricing deals, now looking ahead to many more. But the reality of the deals, so far, have fallen short of grand promises the president made. https://t.co/QmhvZNgNyI

Key Takeaways: How Regulatory Exclusivity, PTA, PTE, and Double Patenting Shape Pharmaceutical Lifecycle Value
The recent Sterne Kessler webinar dissected how FDA regulatory exclusivities, patent‑term adjustment (PTA), patent‑term extension (PTE) and obviousness‑type double patenting (ODP) intersect to shape a drug’s lifecycle value. Regulators can grant exclusivity periods that outlast patent terms, while PTA can add...

All RFK Jr. Hearings Summarized, Plus Fix‑It Discussion
On this week's #WTHealth podcast: we watched ALL the RFKjr hearings so you didn't have to! with @SherylNYT, @AliceOllstein, and @victoriaregisk. Plus "How Would You Fix it?" with David Blumenthal. https://t.co/YpCpbgcSVC
FDA Approves First Free Gene Therapy Restoring Deaf Children's Hearing
JUST IN: FDA approves first ever gene therapy that restores hearing in children born deaf, with treatment available at no cost

First Bedside Procedure of Its Kind Performed by Traveling Clinicians on Premature Infant
Cardiologists in Florida performed the first traveling bedside transcatheter patent ductus arteriosus (PDA) closure on a 22‑week‑old premature infant. Led by Dr. Shyam Sathanandam at Nicklaus Children’s Hospital, the minimally invasive procedure was completed in the NICU isolette, eliminating the...

Healthcare Software Shifts From Workflows to Intent
Healthcare software is moving from workflows… to intent. Srinivas Velamoor talks about systems that understand what you want, not just what you click ↓ https://t.co/iGlc6onYkI @NextGen #AmbulatoryEHR #HITSM https://t.co/0h8tr7GxkZ
The BioPharm Brief: CAR-T Advances, Pediatric Biologic Approval, and Oral GLP-1 Progress
A2 Biotherapeutics will unveil early data on its A2B694 CAR‑T therapy, which targets HLA‑A*02 loss of heterozygosity in solid tumors, at ASCO 2026. The FDA approved dupilumab for children ages 2‑11 with chronic spontaneous urticaria, marking the first biologic for this pediatric...
SurGenTec Adds Navigation Option to FDA-Cleared SI Joint Fusion System
SurGenTec received FDA 510(k) clearance for its TiLink navigation instruments, which integrate with Medtronic’s StealthStation platform to provide real‑time guidance during minimally invasive sacroiliac (SI) joint fusion. The new tools are designed to help surgeons locate, access, and prepare the...

Profit Gap: Preventing Strokes Beats Treating Them
Is there a window in American healthcare where preventing strokes is more profitable than treating them? @Farzad_MD left his dream job at the HHS to find out. New Lifers this week: Spotify: https://t.co/sl8a6nuUlC Apple: https://t.co/yDarYoJcDz Youtube: https://t.co/UDbEjHA3cD

Is Ozempic Conversion Therapy?
The author provocatively equates Ozempic, a GLP‑1 weight‑loss drug, with conversion therapy, arguing that both aim to change a person's physical or identity traits. The piece references the Human Rights Campaign’s condemnation of conversion therapy while questioning whether weight‑loss interventions...