Today's Healthcare Pulse

FDA greenlights durvalumab combo for high‑risk bladder cancer
The FDA approved durvalumab (Imfinzi) combined with Bacillus Calmette‑Guerin for BCG‑naïve, high‑risk non‑muscle invasive bladder cancer. The POTOMAC trial enrolled 1,018 patients and showed a 32% reduction in disease recurrence risk (hazard ratio 0.68, p=0.015). Durvalumab is given at 1,500 mg IV every four weeks for up to 13 cycles.
Also developing:
By the numbers: Apogee Therapeutics raises $1.3B royalty financing

What You Should Know About National Prescription Drug Take Back Day
The DEA’s 30th National Prescription Drug Take Back Day is set for Saturday, April 25, 2026, with roughly 4,200 drop‑off sites across the United States. The event offers a free, anonymous way for the public to discard expired or unused pills, patches, and solid‑form prescriptions. Since its inception in 2010, the program has collected over 20 million lb (10,196 tons) of medication, including 571,054 lb at the October 2025 event alone. Health officials stress that removing household drug stockpiles curbs misuse, accidental poisonings, and opioid‑related overdoses.

Substance Use Disorder Biotech Tempero to Close After Earlier 'Serious' Safety Event
Tempero Bio, a biotech focused on novel treatments for substance‑use disorders, announced it will wind down operations following a serious adverse event in its late‑stage clinical trial. The company had raised more than $200 million to advance a kappa‑opioid receptor antagonist...

Tricuspid Training Series: Echocardiographic Evaluation of Patients with Tricuspid Regurgitation
In the latest Heart Valve Matters podcast, cardiologists Rick Nishimura and Paul Grayburn dissect how echocardiography is used to evaluate tricuspid regurgitation (TR). They outline the imaging techniques—2‑D, Doppler, and emerging 3‑D modalities—required to grade severity and assess right‑ventricular function....

Pharmaceutical Executive Daily: FDA Expands Approvals for Tzield and Dupixent
The FDA broadened Tzield’s approval to treat children as young as one year with stage 2 type 1 diabetes, aiming to delay progression to stage 3 disease. It also expanded Dupixent’s label to cover young patients with uncontrolled chronic spontaneous urticaria, adding another...

CDC Data Show Weekly ER Visits for Tick Bites Higher than Usual
The CDC’s Tick Bite Tracker shows weekly emergency‑room visits for tick bites are at their highest levels since 2017 in every U.S. region except the South‑Central states. The surge comes as the nation heads into Lyme Disease Awareness Month, prompting...
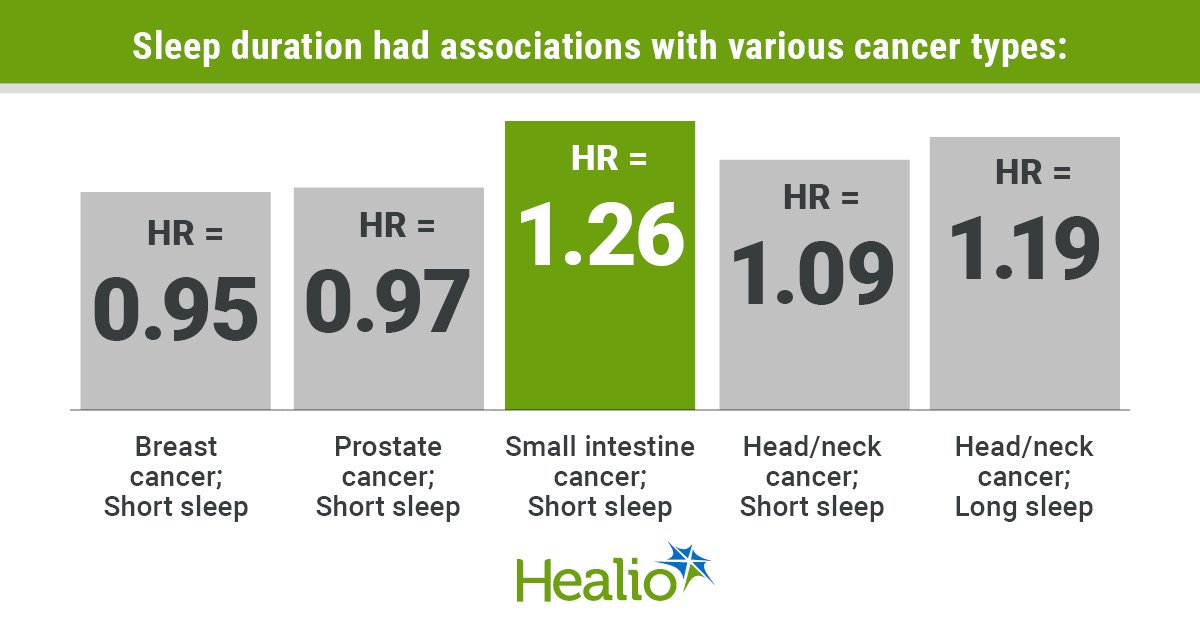
Sleep Duration Has ‘Complex’ Association with Cancer
Researchers presented a pooled meta‑analysis of seven prospective cohorts involving 918,000 adults, finding that sleeping less than 7 hours per night is linked to a slight overall reduction in cancer incidence but raises risk for specific malignancies such as small‑intestine cancer,...

6 High-Resolution Additive Manufacturing Tips for Faster Medtech Development
The article presents six actionable tips for using high‑resolution additive manufacturing (AM) to speed up medical‑device development. It urges teams to adopt an iteration‑first mindset, exploit sub‑10 µm layer precision to answer targeted engineering questions, and revisit designs once deemed impractical....

Find Information About a Drug
The FDA and NIH provide a suite of online tools that let consumers, clinicians, and researchers locate comprehensive drug information. Resources such as Drugs@FDA, DailyMed, and MedlinePlus deliver FDA‑approved labeling, safety data, and side‑effect details for both prescription and over‑the‑counter...

Self-Inflicted Wounds
CMS has proposed CJR‑X, a mandatory, nationwide bundled‑payment model for all Medicare joint replacements effective October 2027. The original CJR program saved roughly $112 million over two years, mainly by reducing post‑acute care, but CJR‑X projects only $725 million in savings over five...

Novartis' Radioligand Therapy Lutathera Could Soon Face Generic Competition
Novartis’s Lutathera, the first FDA‑approved radioligand therapy for neuroendocrine tumors, recorded $1.5 billion in 2023 sales and dominates a market projected to exceed $3 billion by 2028. A generic version filed by Sandoz aims for a 2025 launch, marking the first non‑brand...

Mass General Brigham Secures Nearly $866M Financing Package For Ongoing Expansion
Mass General Brigham secured a $865.5 million tax‑exempt bond package, led by J.P. Morgan Securities, to fund its ongoing West End campus expansion. The financing will support the 482‑bed Ragon Building, which will boost oncology and cardiovascular services, and add five stories...

Really Need Long Pedicle Screws in Good Bone? Ever?
A cadaveric biomechanical study compared 35 mm “short” pedicle screws with the longest possible screws in lumbar vertebrae under cyclic fatigue loading. In vertebrae with normal bone density, both screw lengths endured similar fatigue loads (~315 N), indicating the pedicle alone provides...

House Panel Advances FDA Spending Hike In Party-Line Vote
The House Appropriations Committee’s FDA subcommittee voted along party lines to advance a bill that adds roughly $200 million to the agency’s budget. The measure cleared the subcommittee with little debate and will move to a full committee markup next week....
FDA Approves Dupilumab for Young Children With Uncontrolled CSU
The FDA has approved dupilumab (Dupixent) for children ages 2‑11 with uncontrolled chronic spontaneous urticaria (CSU), marking the first biologic therapy for this pediatric group. The decision is based on the LIBERTY‑CUPID phase 3 program, which demonstrated significant reductions in itch...

Drug Safety Information
The FDA’s Drug Safety Information hub aggregates a suite of resources—Drug Safety Communications, MedWatch reporting, alerts, post‑market monitoring, and searchable databases such as FDALabel—to help clinicians, patients, and industry stay informed about medication risks and benefits. The portal also offers...

Stroke Impact Determines Future Dementia Risk
A national cohort of over 42,000 adults tracked for up to 30 years shows a clear dose‑response link between stroke severity and later dementia. Survivors of severe ischemic strokes face roughly five times the odds of developing dementia, while even...
Publisher’s Platform: Hepatitis A and the Food Service Industry: A Case for Universal Vaccination
Bill Marler’s op‑ed urges universal Hepatitis A vaccination for food‑service workers, noting that the CDC removed the vaccine from the routine childhood schedule. He stresses that the virus spreads during the two weeks before symptoms appear, turning a single unvaccinated employee...

Vitamin D May Prevent Diabetes in People with Certain Genes
A new analysis of the D2d trial shows that a daily 4,000 IU vitamin D supplement reduces the risk of developing type 2 diabetes by 19 % in prediabetic adults who carry the AC or CC variants of the vitamin D receptor gene, while those...
Google Search Trends Reflect a Shift Toward Minimally Invasive Heart Care
New research presented at the SCAI 2026 Scientific Sessions shows public interest in transcatheter aortic valve replacement (TAVR) surged 340% from 2015 to 2025, while searches for surgical aortic valve replacement (SAVR) fell 42%. The spike aligns with clinicians doubling...
FDA Approval of Regeneron’s Hearing Loss Gene Therapy Breaks Barriers
Regeneron Pharmaceuticals received FDA approval for Otarmeni, the first gene therapy targeting congenital deafness caused by otoferlin deficiency. The treatment, approved under the Commissioner’s National Priority Voucher program, demonstrated clinically meaningful hearing gains in 11 of 12 patients in the...
Drug-Coated Balloons Reduce the Need for Permanent Heart Stents
A sub‑study of the SELUTION DeNovo trial presented at the SCAI 2026 meeting shows that a sirolimus‑eluting balloon (SEB) can treat NSTEMI and unstable angina with outcomes comparable to drug‑eluting stents (DES). The analysis of 1,089 patients found one‑year target‑vessel...

Early Heart Pump Use Improves Survival in Patients Experiencing Cardiogenic Shock
The CERAMICS registry, a single‑arm study of 124 cardiogenic shock patients across 20 U.S. hospitals with on‑site mechanical circulatory support (MCS), showed that rapid Impella placement and PCI within roughly 75 minutes led to a 71% overall survival to discharge....

Treatment Goals Guide Cardiogenic Shock Care More Often in Women
The Northwell‑Shock Registry analysis of 1,374 AMI‑related cardiogenic shock patients revealed that women are less likely to undergo invasive coronary angiography (78% vs 86% in men). When angiography is performed, subsequent PCI rates are virtually identical between sexes. Deferral of...

MedCity Pivot Podcast: Modernizing Prior Auth
The MedCity Pivot podcast featured Abarca Health’s Javier Gonzalez and Amazon Pharmacy’s Tanvi Patel discussing how to modernize prior authorization. They highlighted three pillars—policy complexity, data quality, and operational risk—and explained that electronic prior authorizations (ePA) could cut 60‑70% of...
Specific Intestinal Fungi Play Role in the Pathogenesis of MASLD and Cardiovascular Disease
The study examined 103 patients with metabolic dysfunction‑associated steatotic liver disease (MASLD) and found that higher fecal Candida albicans levels were linked to increased coronary artery calcification, especially among those with cirrhosis. Liver stiffness measured by magnetic resonance elastography correlated...
Microplastics in the Liver May Drive Global Liver Disease Rates
Researchers at the University of Plymouth’s Centre of Environmental Hepatology have published a review in Nature Reviews Gastroenterology & Hepatology linking micro‑ and nanoplastic accumulation in the liver to oxidative stress, inflammation and fibrosis. The paper introduces the concept of...

Investigation of SARS-CoV-2 Variants at Primer Binding Sites in Diagnostic Platforms and the Effect on Laboratory Diagnostic Samples
Researchers examined ~26,000 SARS‑CoV‑2 genomes to assess how mutations in primer and probe binding sites affect RT‑PCR diagnostic accuracy. They evaluated twelve primer sets across time, geography, and variant categories, finding mismatch rates from 0.15% up to 77.15% and linking...

426. The Case for Universal Vaginal Estrogen Use After Menopause
The episode explains why universal vaginal estrogen use after menopause is essential, detailing how systemic HRT fails to restore the vaginal and lower urinary tract tissues that suffer from genitourinary syndrome of menopause (GSM). Low‑dose vaginal estradiol—whether in compounded cream,...

Charge Masters Are Fake Prices Exploiting Patients for Profit
Gotta make charge masters real prices or get rid of them. The CM is like WAC in pharma. It's a made up number that abuses the uninsured , those in their deductible phase, and those who need...

Adrian Owen & Faraz Shafaghi, Creyos
Creyos, a neurological testing firm co‑founded by neuroscientist Adrian Owen and product leader Faraz Shafaghi, offers a cognitive assessment platform that delivers objective baseline data at the point of care. The tool is now incorporated into annual wellness visits and...
GLP-1 Drugs Target the Roots of Dementia
A systematic review of 30 preclinical studies finds that GLP‑1 receptor agonists—particularly liraglutide, semaglutide, dulaglutide and exenatide—consistently reduce amyloid‑beta plaques and tau tangles, the hallmarks of Alzheimer’s disease. The drugs also appear to curb neuroinflammation and improve brain insulin signaling,...

Regenerative Medicine: Promise, Hype, and What Actually Works
Regenerative medicine spans stem cells, platelet‑rich plasma (PRP) and autologous conditioned serum (ACS), but not all modalities live up to hype. Dr. Thomas Buchheit emphasizes that stem‑cell injections rarely persist in tissue and mainly trigger immune‑mediated repair, while PRP and...

AI-Driven Synthetic Evolution Accelerates Biological Design
Evolution is the most powerful optimization algorithm ever run. It just takes billions of years. Researchers are now rewriting the code. #SynBioBeta2026 is May 4-7th in San Jose, California, you can learn more about the conference and get your tickets here:...

CDC Announces Salmonella Outbreak in 13 States Linked to Backyard Poultry.
The CDC has confirmed a multistate Salmonella outbreak linked to backyard poultry, affecting 34 people in 13 states, with 13 hospitalizations and no deaths reported. Healthy‑looking chickens and ducks can carry the bacteria, exposing owners and their families through direct...
Regeneron Approves Free Gene Therapy, Sparks Industry Precedent Worries
$REGN wins FDA approval for its gene therapy to treat a very rare genetic form of hearing loss. The company is giving away the therapy for free. OTOF-related hearing loss affects approx. 50 newborns per year, so really rare, but......

Tirzepatide Significantly Reduces Cardiovascular Risk in High-Risk Patients
Two recent real‑world studies demonstrate that tirzepatide, a dual GIP/GLP‑1 receptor agonist, markedly lowers cardiovascular risk in high‑risk patients. In a propensity‑matched cohort of 1,281 type‑2 diabetics undergoing percutaneous coronary intervention, tirzepatide reduced mortality by 62% and cut major adverse...

DEBATE Today: Trump’s Psychedelic Order
Rav Arora announced on X that two leading drug‑policy experts, Kevin Sabet and Matthew Johnson, will join his Illusionist podcast to debate President Trump’s newly issued executive order on psychedelics. The order, released in early April, directs federal agencies to accelerate...
STAT+: Legislatures in Colorado and Virginia Resist Moves to Constrain Drug Affordability Boards
Legislators in Virginia and Colorado pushed back against attempts to limit the authority of state drug‑affordability boards. In Virginia, the General Assembly voted unanimously to keep the original bills that would create a board with power to set price caps...

What Is the UK Biobank Project and What Are the Privacy Concerns Around It?
The UK Biobank, launched in 2003, has amassed genetic, clinical and lifestyle data from 500,000 volunteers, fueling thousands of research papers and AI tools that predict disease risk. In April 2026, de‑identified health records from the biobank were listed for...
Why Clinical Care Resilience Is a Top Priority in Healthcare
Healthcare leaders warn that cyber‑threats and system failures can cripple clinical operations, making care‑resilience a top priority. Recent ransomware incidents at Michigan Medicine, University of Vermont Health Network, and Children’s National illustrate the need for cross‑departmental planning and frequent security...
Medical Training Must Evolve Faster than Technology
The future of medicine is arriving faster than our training models are evolving. A student starting medical school in 2026 won’t earn their M.D. until 2030, and likely won’t finish residency or practice independently until 2033 or beyond. By then, the...

Obese HFpEF Patients Show Sarcom
In people with HFpEF (heart failure with preserved ejection fraction) and severe obesity, there is a heart muscle cell defect with sarcomere hyper-phosphorylation. Besides weight loss, sarcomere enhancers (not yet studied) may help. @ScienceMagazine https://t.co/or9VaPjD8J https://t.co/FG98KIzIfN

Labels, Language and Other Strategies to Improve Communication About Lower Grade Ductal Carcinoma in Situ: Integration of Findings From Theoretical...
A mixed‑methods study combined a theoretical review with patient interviews to pinpoint language that best supports women diagnosed with low‑grade ductal carcinoma in situ (DCIS). Using Communication Accommodation Theory, researchers mapped clinician‑patient interactions across five communication domains and uncovered a...

Affordable Housing Serves as Unrecognized Care Coordination Hub
This is a really important reframing. Affordable housing operators are already acting as de facto care coordinators, but without the infrastructure, funding, or recognition that #healthcare systems receive. https://t.co/NIe4JUGjLe

Hiltzik: A Judge Labels RFK Jr.'s Attack on Transgender Care 'Unlawful' And an Act of 'Cruelty'
Federal Judge Mustafa T. Kasubhai in Oregon struck down Robert F. Kennedy Jr.'s December 18 declaration that threatened to cut Medicaid and Medicare funding for hospitals offering gender‑affirming care to minors. The decision, part of a lawsuit filed by 19...
Roche Launches New Elevidys Trial to Address EU Rejection in Duchenne Therapy Bid
Roche has launched a new global phase 3 trial of its Duchenne muscular dystrophy gene therapy Elevidys, targeting ambulatory boys after the European Medicines Agency rejected its earlier submission. The study will enroll about 100 patients and compare Elevidys to...
Roche Bets on Cancer and Obesity Drugs Amid Biosimilar Pressure
Roche, facing biosimilar threats, puts faith in new cancer and obesity drugs https://t.co/IIt0INA9sI @ByJonGardner $RHHBY

25% of Chronic Pain Patients Show ADHD Traits
A University of Tokyo study of 958 Japanese adults with chronic pain found that roughly 25% exhibited significant ADHD traits, a rate 2.4 times higher than in the general population. The research shows that ADHD does not directly cause pain...

New ADC Yields Encouraging Clinical Benefit in Platinum-Resistant Ovarian Cancer
In a Phase I trial presented at AACR 2026, the investigational antibody‑drug conjugate QLS5132 demonstrated notable antitumor activity in patients with advanced platinum‑resistant ovarian cancer. Among 28 heavily pretreated participants, the overall objective response rate was 50% and disease control...

STAT+: Sanofi Research Priorities in Flux as New CEO Logs In
Sanofi’s new chief executive, Belén Garijo, assumes leadership amid questions about the French drugmaker’s research direction. The company recently intensified its immunology portfolio, but recent trial disappointments have dampened expectations. Garijo is expected to reassess R&D allocations, potentially shifting focus...