Today's Healthcare Pulse

Pharma Shifts Ad Spend to Spot TV Amid Heightened FDA Scrutiny
Pharmaceutical advertisers are redirecting dollars toward spot television placements as the FDA intensifies its oversight of drug marketing. The trend reflects a strategic move to capture audiences with shorter, targeted ads while navigating tighter regulatory expectations.
Also developing:
By the numbers: Apogee Therapeutics secures $1.3B royalty financing

Brightspring Health Services Inc (BTSG) Q1 2026 Earnings Call Transcript
BrightSpring Health Services reported $12.9 billion in total revenue for 2025, a 28% year‑over‑year increase driven by 31% growth in Pharmacy Solutions and 11% growth in Provider Services. Adjusted EBITDA rose 34% to $618 million, with margins expanding thanks to procurement efficiencies and a favorable revenue mix. The company is set to close the Community Living divestiture, generating roughly $715 million to further reduce leverage to about 2.6x, while the Amedisys/LHC acquisition is expected to contribute $30 million of EBITDA in 2026. Guidance for 2026 projects revenue of $14.45‑$15.0 billion and adjusted EBITDA of $760‑$790 million, excluding pending deals, despite $775 million of headwinds from the Inflation Reduction Act and brand‑to‑generic conversions.

Targeted and Timely: Curating Aesthetic Care for GLP-1 Patients
The surge in GLP‑1 agonist use for weight loss is prompting a wave of elective aesthetic procedures. A recent survey of 127 U.S. aesthetics providers shows 20% of GLP‑1 patients opting for surgical lifts and 39% of nonsurgical users considering...

Moderna Inc (MRNA) Q1 2026 Earnings Call Transcript
Moderna reported Q1 2026 revenue of $108 million, a 35% year‑over‑year decline, while posting a $1 billion net loss. Cost of sales rose to 104% of product sales, reflecting lower volume and an adverse mix, but combined R&D, SG&A and cost‑of‑sales fell...

The Women’s Health Playbook
The Women’s Health Playbook podcast, hosted by Priya Bathija, breaks down the complexities of navigating U.S. healthcare for women, offering actionable strategies for every stage of a doctor’s visit—from choosing the right clinician to preparing for appointments and deciphering medical...

Trump Picks Nicole Saphier for Surgeon General After Withdrawing Casey Means Nomination
President Donald Trump withdrew Dr. Casey Means’ stalled surgeon‑general nomination and announced Dr. Nicole B. Saphier as his new pick. Saphier, a radiologist and breast‑imaging director at Memorial Sloan Kettering’s Monmouth campus, is known for a wellness podcast and frequent...

We Tried to Get Trump to Back Psychedelic Therapy — Here’s What Happened
The authors drafted a bipartisan policy blueprint to accelerate psychedelic‑based treatments, targeting veterans with PTSD, addiction and depression. After Joe Rogan shared the proposal, it amassed over 1.5 million views on X, prompting meetings with health‑policy influencer Calley Means and a...

The Trump Administration Casts Out the ‘Soul’ of MAHA
President Trump announced on Truth Social that he is withdrawing Casey Means’ nomination for surgeon general, ending the Make America Healthy Again (MAHA) push to install a wellness influencer with a controversial health agenda. Means, a former surgical resident turned...

Rep. Langworthy Continues Push for Dietary Supplement Regulatory Uniformity Act
The Dietary Supplement Regulatory Uniformity Act (H.R. 7366) would amend the Federal Food, Drug, and Cosmetic Act to prevent states from imposing supplement requirements that conflict with federal law, allowing exemptions only when a state rule is stricter and does not...

Health Canada Publishes Prebiotics Monograph
Health Canada has released a prebiotic monograph that adopts the International Scientific Association for Probiotics and Prebiotics (ISAPP) 2017 definition. The document enumerates approved prebiotic ingredients—including inulin, acacia gum, FOS, GOS, XOS, and the human‑milk oligosaccharides 2′‑FL and LNnT—and draws...

Professional Misconduct in Malawi’s Nursing and Midwifery Professions: A Descriptive Study of Trends, Causes and Implications
A descriptive analysis of 102 professional misconduct cases recorded by Malawi’s Nurses and Midwives Council between 2014 and 2024 reveals that negligence dominates, representing 82% of confirmed violations. The infractions are concentrated among nurses and midwives aged 20‑39 with less...
![2026 Cholesterol Guidelines: LDL Goals, Lp(a), and Coronary Calcium Scoring [PODCAST]](/cdn-cgi/image/width=1200,quality=75,format=auto,fit=cover/https://kevinmd.com/wp-content/uploads/baum_thumbnail.png)
2026 Cholesterol Guidelines: LDL Goals, Lp(a), and Coronary Calcium Scoring [PODCAST]
The American College of Cardiology and American Heart Association released the 2026 cholesterol guideline, re‑introducing explicit LDL‑C targets for the first time in eight years. The update adds universal lipoprotein (a) screening, designates a coronary calcium score > 300 as a high‑risk marker,...

The Right to Choose to Die
Alvin Roth, Nobel‑winning economist, discusses assisted dying on VoxTalks Economics, framing it as a “repugnant transaction” that markets can regulate. He outlines the legal landscape—12 U.S. states and D.C., Canada, and Switzerland—where medical aid‑in‑dying (MAID) is permitted under varying criteria....

PAMA Laboratory Data Reporting Requirements for Hospitals Are Set to Impact Medicare Lab Payments
Hospitals that run clinical diagnostic labs must report commercial final‑paid claim rates for January‑June 2025 during a May 1‑July 31 window, a requirement under the Protecting Access to Medicare Act (PAMA). Roughly 2,600 hospitals are subject to the mandate, and the data...

Blue Cross Blue Shield Customers May Soon See Settlement Payments
Blue Cross Blue Shield’s $2.67 b antitrust settlement has moved to the final distribution phase, with the net $1.9 b fund slated for payouts beginning May 2026. The settlement resolves claims that the insurer’s network limited competition, allegedly inflating premiums for millions of...

GLP-1s Reduce Heavy Drinking Days in Patients With Obesity, Alcohol Use Disorder
A randomized, double‑blind trial in Copenhagen found that once‑weekly semaglutide 2.4 mg significantly reduced heavy‑drinking days in patients with alcohol use disorder and obesity. Over 26 weeks, the semaglutide group saw a 41.1‑percentage‑point drop in heavy‑drinking days versus 26.4 points for...

Sabra CEO: Skilled Nursing Deal Flow Tight Amid Fierce Competition From OpCo and PropCo Private Buyers
Sabra Health Care REIT is pivoting away from skilled‑nursing assets toward its Seniors Housing Operating Portfolio (SHOP), with private‑pay revenue now representing over 50% of the portfolio for the first time. The CEO said skilled‑nursing deal flow remains tight as...

From Prototype to Production: Building a Validation Strategy That Scales with Manufacturing Volume
Medical device manufacturers must redesign validation strategies as prototypes transition to full‑scale production. Early validation plans often ignore equipment wear, multi‑shift operation, and material lot diversity that emerge at higher volumes. Regulatory bodies like the FDA and ISO 13485 only require...

Ivermectin Can't Cure Cancer
A new preprint circulating online alleges that a combination of ivermectin and mebendazole can cure cancer, a claim promoted by the unregulated Wellness Company. The paper is a survey of patients at the company’s clinics and offers no clinical trial...

Trump Withdraws Surgeon General Pick: Why & What’s Next For MAHA?
President Donald Trump withdrew Dr. Casey Means’ nomination for U.S. Surgeon General after a stalled Senate HELP Committee hearing and announced board‑certified radiologist Dr. Nicole Saphier as his replacement. Means, a functional‑medicine advocate with an inactive medical license, faced bipartisan...

Trump Nominates Fox News Doctor to Be the Next Surgeon General
President Donald Trump withdrew Casey Means’ surgeon‑general nomination after Senate resistance and announced Fox News radiologist Nicole B. Saphier as his replacement. Means, a wellness influencer with an incomplete residency and anti‑vaccine reputation, faced objections from Republican chair Bill Cassidy....

Daily Bulletin...
President Donald Trump formally withdrew Dr. Casey Means’ nomination for U.S. Surgeon General after the Senate stalled her confirmation. In her place, the White House tapped Fox News health commentator Nicole Saphier as the new nominee. Means, a former COVID‑19...
:max_bytes(150000):strip_icc():format(jpeg)/affordable-care-act.asp-final-43c18941b07c4e49b29738891503d38d.png)
Affordable Care Act (ACA): What It Is, Key Features, and Updates
The Affordable Care Act (ACA), signed in March 2010, created a nationwide health‑insurance marketplace, expanded Medicaid eligibility, and barred insurers from denying coverage for pre‑existing conditions. It mandates essential health benefits and offers premium tax credits that lower costs for low‑...
Morning Headlines 5/1/26
Larry Ellison is reportedly financing Texas Attorney General Ken Paxton to pursue legal actions against business rivals, a strategy critics say leverages state power for private gain. The lawsuit filed by Paxton’s office has been described by legal analysts as...

Managing AI in Medical Technology: From Innovation to Compliance
AI is rapidly reshaping medical technology, prompting heavy investment in algorithm‑driven devices and software. At the same time, regulators worldwide are tightening oversight, creating a need for structured AI governance. The newly released ISO/IEC 42001 standard offers a lifecycle framework that...

Politicians Enable Fentanyl Crisis; Demand Zero Tolerance
San Francisco politicians and their constituents enable this suffering. They are the cause of it, because they can stop it instantly. Today, tomorrow and every one of the last 1,000 days this has happened. Fentanyl is a super drug and...
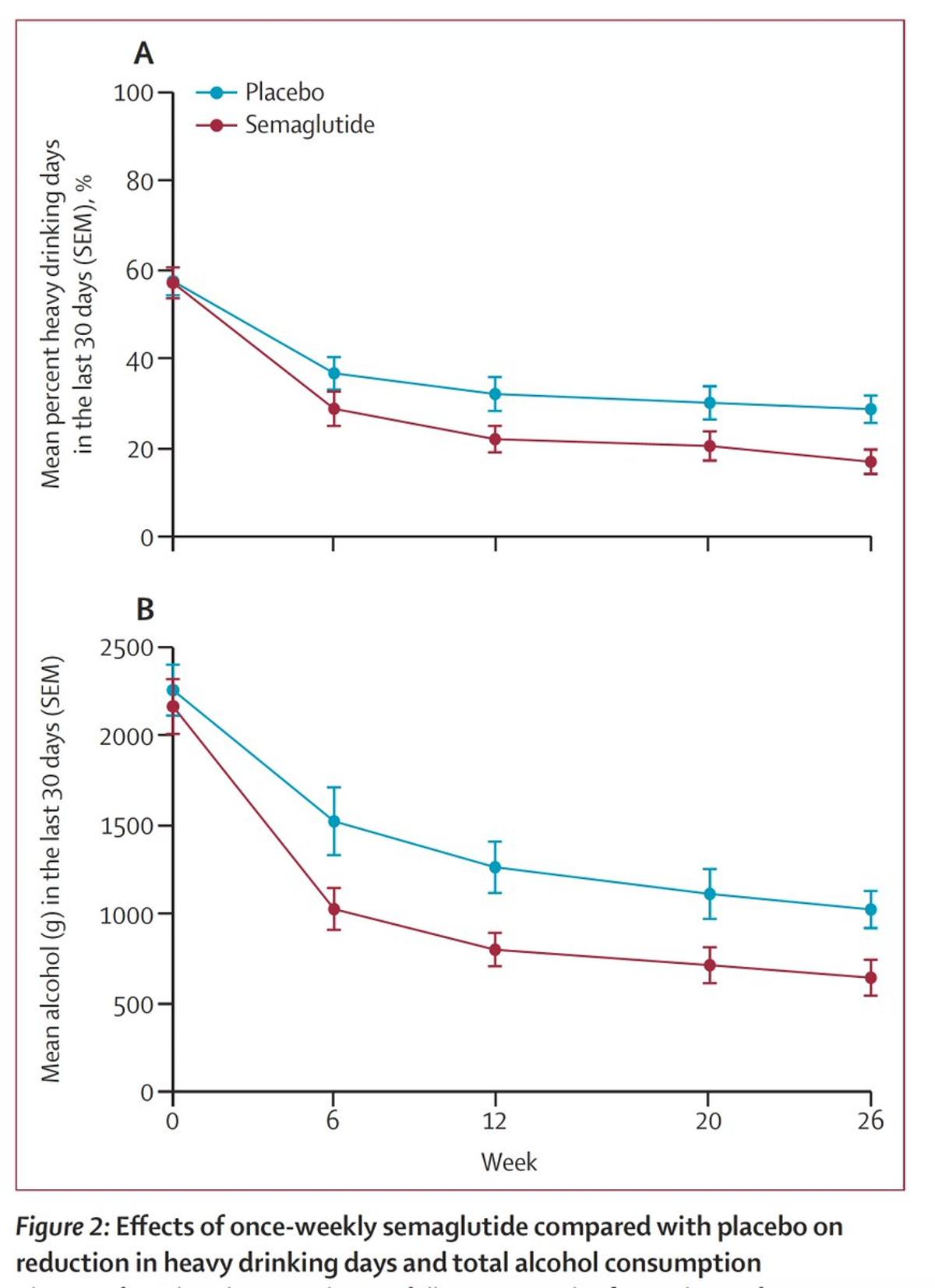
Ozempic Cuts Drinking in Obese AUD Patients
First randomized trial to show Ozempic reduces alcohol consumption in people seeking treatment for alcohol use disorder. Placebo-controlled, double-blind. Participants with BMI >30 kg/m2. https://t.co/M9Pk2gzKHD @TheLancet https://t.co/OT660QIn6N

Why Digital Health Initiatives Fail: 35 Healthcare Leaders Weigh In
A survey of 35 health‑care executives reveals that many digital health projects—patient portals, EHR interoperability, AI tools, remote monitoring, and staffing algorithms—have fallen short of expectations. Leaders cite insufficient change management, lack of clinician trust, poor workflow integration, and inadequate...
GLP‑1 Drugs Cause Less Muscle Loss than Assumed
Do GLP-1 drugs really cause muscle loss? Turns out perhaps less than what we thought… here’s what you need to know, from: https://t.co/VwrRxnoblW https://t.co/M2RaE1f3EG
AI Beats Doctors in Emergency and Complex Diagnosis Reasoning
An AI program performed better than human doctors on reasoning tasks such as making emergency room decisions and diagnosing complex cases, according to a new study published Thursday in @ScienceMagazine https://t.co/zAeOYW32I4 https://t.co/SGgO3KNfqM

The Future of Medicaid Pediatric Care and Value-Based Partnerships: Chris Johnson, MBA
Bluebird Kids Health is rolling out retail‑based pediatric clinics that sit inside grocery‑anchored shopping plazas, targeting Medicaid families in underserved areas. The model pairs convenient, bright spaces with a suite of digital tools for scheduling, 24/7 mobile access, and automated...

Dr. Wayne Tasker, Director of Behavioral Health Services, TeamHealth
Dr. Wayne Tasker, TeamHealth’s director of behavioral health, discusses his path from adolescent substance‑abuse work to geriatric behavioral health and the rise of person‑centered dementia care in skilled nursing facilities. He outlines simple communication rules—30‑second pause, single‑step commands, and eye...
Navigating Uncertainty: How to Scale Intelligent Care and Make It Stick
Health systems are grappling with a flood of digital tools, but the real hurdle is building disciplined, organization‑wide intelligent care models. At Becker’s 16th Annual Meeting, Huron and Tampa General leaders outlined a three‑pillar framework—operations hub, virtual care, and smart...

Nicole Saphier Nominated Surgeon General Despite Divergent Views
Casey Means is out and Fox contributor Nicole Saphier is the new nominee for surgeon general. While MAHA folks seem to be embracing her, Saphier's views — on vaccines, on the use of Tylenol during pregnancy — aren't entirely in...

‘Depth over Breadth’: Health Systems Eye Quality of AI Applications, Not Number
Large U.S. health systems are shifting from counting AI projects to measuring their clinical and operational value. Leaders at Universal Health Services, Advocate Health, UPMC, Mayo Clinic and NYU Langone describe multi‑stage governance that vets pilots for safety, workflow fit...

Volunteer Compliance Supports Hospices’ Sustainable Growth
Hospice providers must meet Medicare’s 5% volunteer‑care requirement, prompting a focus on recruitment, retention, and precise hour tracking. Leaders like Jennifer Scurry and Wade Udelhoven stress that strong volunteer coordinators and community partnerships are essential to compliance. Emerging volunteer roles, such...

The BioPharm Brief: Metabolic Phase III Progress, HER2 Oncology Momentum, and US Manufacturing Expansion Drive Industry Scale
Zealand Pharma and Roche are moving petrelintide, an amylin analog, into Phase 3 trials for chronic weight management, with enrollment slated for the second half of 2026 after earlier studies showed double‑digit weight loss and tolerability comparable to placebo. The FDA...
Mayo Clinic: Remote Patient Monitoring Can Detect Transplant Complications
Mayo Clinic researchers tracked 116 lung‑transplant recipients for a year using home‑based remote monitoring kits that captured lung function, vital signs and weight. The program generated roughly 470 alerts, and about one‑quarter of those prompted care changes such as earlier...

Medical School Rankings Reshape What They Measure
U.S. News & World Report’s medical‑school rankings have long guided applicants, but recent withdrawals by elite institutions such as Harvard, Columbia, Stanford, Penn and Mount Sinai expose deep flaws. Critics argue the current tiered model over‑weights research funding, MCAT scores...

States Scramble to Submit Medicaid ‘Provider Revalidation’ Plans to CMS
CMS Administrator Mehmet Oz has ordered all 50 states to develop and submit two‑year Medicaid provider revalidation plans by May 23, targeting high‑risk, non‑hospital providers such as home‑based services and durable medical equipment. States must first notify CMS within 10 days...

Drug Trials Snapshots: BIMZELX
UCB’s BIMZELX, a dual IL‑17A/F monoclonal antibody, received FDA approval on Oct 17 2023 for adults with moderate‑to‑severe plaque psoriasis. The approval was based on two phase‑3 placebo‑controlled trials that enrolled 839 patients across 13 countries, with a demographic profile of 72%...

Drug Trials Snapshots: AGAMREE
Santhera Pharmaceuticals received FDA approval on October 26, 2023 for AGAMREE (vamorolone), a corticosteroid designed to treat Duchenne muscular dystrophy (DMD) in patients aged two years and older. The approval rests on a multinational, double‑blind, 24‑week trial of 121 male patients that...

University of Texas Medical Branch Names CIO
The University of Texas Medical Branch (UTMB) in Galveston has appointed Jayson Laban as its vice president and chief information officer, effective May 1. Laban, who has been serving as interim CIO since Jan. 1, will now lead the institution’s technology strategy,...

MetroHealth Primary Care Providers Seek Unionization: 5 Things to Know
Cleveland-based MetroHealth’s primary‑care clinicians are organizing a union after a rally on April 28, demanding relief from what they describe as unsustainable workloads. The proposed Primary Care Providers Union of MetroHealth would cover physicians, physician assistants, APRNs and nurse practitioners,...

Drug Trials Snapshot: TALVEY
TALVEY (talquetamab‑tgvs), a bispecific antibody from Janssen Biotech, received FDA approval on August 10 2023 for adult patients with relapsed or refractory multiple myeloma who have exhausted at least four prior therapies. The pivotal trial enrolled 339 participants across 47 sites, with...

Drug Trials Snapshots: OJJAARA
OJJAARA (momelotinib) received FDA approval on September 15 2023 for adults with myelofibrosis‑related anemia. The approval rests on two pivotal trials—MOMENTUM, which enrolled JAK‑inhibitor‑experienced patients, and SIMPLIFY‑1, which enrolled JAK‑naïve patients. In MOMENTUM, 25% of OJJAARA recipients achieved a ≥50% reduction in...

There Are Two GLP-1 Side Effects Your Doctor Doesn’t Know About, and They Can Affect Your Workouts
A new *Nature Health* study used AI to scan 400,000 Reddit posts, finding that roughly 70,000 users were taking GLP‑1 drugs such as Ozempic, Wegovy, or Zepbound. While nausea and fatigue remain the most common side effects, about 4% of...

Independent Doctors Applaud President Trump and the White House for Surgeon General Nominee Dr. Nicole Saphier
President Trump nominated radiologist and bestselling author Dr. Nicole Saphier as U.S. Surgeon General, prompting applause from the Independent Medical Alliance (IMA), the nation’s largest coalition of independent physicians. The IMA praised Saphier’s patient‑first advocacy and her willingness to challenge...

Seven Things Every Medical Device Manufacturer Must Know Before Integrating AI
Artificial intelligence has moved from experimental to production‑ready in healthcare, enabling devices like predictive glucose monitors and AI‑driven oncology tools. For medical‑device makers, integrating AI is now a business imperative but also a complex regulatory undertaking. The article outlines seven...

Justices Poised to Protect Generics Manufacturers From Liability for Decisions of Pharmacists About Prescribing Their Products
The Supreme Court heard arguments in Hikma Pharmaceuticals USA v. Amarin Pharma, where Amarin seeks to hold generic maker Hikma liable for pharmacists dispensing its generic product in ways that infringe Amarin’s Vascepa patents. The Court appeared skeptical that Hikma’s...

Interim PFS Review Confirms Study Continuation
$SMMT ivo HARMONi-3 update on squamous subgroup interim PFS analysis: "At this early interim PFS analysis reviewed exclusively by the Independent Data Monitoring Committee (iDMC), the iDMC recommended that the study continue as planned."