Today's Healthcare Pulse

FDA greenlights durvalumab combo for high‑risk bladder cancer
The FDA approved durvalumab (Imfinzi) combined with Bacillus Calmette‑Guerin for BCG‑naïve, high‑risk non‑muscle invasive bladder cancer. The POTOMAC trial enrolled 1,018 patients and showed a 32% reduction in disease recurrence risk (hazard ratio 0.68, p=0.015). Durvalumab is given at 1,500 mg IV every four weeks for up to 13 cycles.
Also developing:
By the numbers: Apogee Therapeutics raises $1.3B royalty financing

Who Is Nicole Saphier?
President Trump withdrew Casey Means and, within minutes, nominated Dr. Nicole B. Saphier, a breast‑imaging oncologist at Memorial Sloan Kettering, as his third Surgeon General pick in fifteen months. Saphier is the author of the 2020 bestseller *Make America Healthy Again*, which frames chronic disease as a trillion‑dollar crisis driven by lifestyle and fee‑for‑service incentives. Her public record shows support for COVID‑19 vaccines in high‑risk groups but criticism of universal pediatric boosters, and she regularly appears on Fox News. The nomination repositions the Surgeon General’s office toward prevention‑focused messaging while sidestepping the vaccine‑policy showdown that stalled Means.

MedCity FemFwd: Inside Partum Health’s Partnership with UChicago Medicine
Partum Health has partnered with UChicago Medicine in Hyde Park to provide 24/7, Medicaid‑reimbursed doula support for labor, delivery, and postpartum care. The program offers in‑person doulas without referrals, aiming to improve birth outcomes and patient satisfaction for a largely Medicaid‑eligible...

People in Low-Income Areas Are Less Likely to Get Cancer Screenings
A new study of 1,300 federally qualified health centers (FQHCs) serving 29.8 million patients reveals stark gaps in cancer screening rates across socioeconomic lines. Residents of the most resource‑poor neighborhoods receive colorectal screening about 15 percentage points less, breast screening 11 points less,...

Cybersecurity Tactics for Medical IoT Devices
The Internet of Medical Things (IoMT) is projected to surge from $60 billion in 2024 to $814 billion by 2032, driven by real‑time patient monitoring and cost‑saving benefits. However, 2024 saw over 14,000 IoMT IPs exposed, with 36% stemming from unsecured medical...

The Prostate Cancer Recovery Few Men Are Warned About
Physiatrist Dr. Francisco M. Torres, a prostate‑cancer survivor, recounts his unexpected post‑operative challenges after robot‑assisted radical prostatectomy. His experience revealed a systemic failure: patients receive scant guidance on pre‑habilitation and pelvic‑floor rehabilitation, despite strong evidence that these interventions improve continence...

Sorting Through Medicare Myths in Federal Retirement Decisions
Federal retirees face a maze of Medicare myths that can affect their health‑care costs. Adding Medicare Part B costs a base $202.90 per month, but many FEHB plans waive deductibles, copays and coinsurance when Medicare is primary, potentially offsetting the premium....

Asembia ASX26: The Strain on the Development of Infrastructure for Sustainable C&G Therapy
Cell and gene therapies promise transformative treatments but remain hampered by steep costs across research, manufacturing, and delivery. At the Asembia ASX26 Summit, Aradigm Health CEO Will Shrank highlighted the fragmented U.S. payment landscape as a core barrier to sustainable...

FDA's Oncology Advisors Vote Against 'New Paradigm' In AstraZeneca Trial
The FDA’s oncology advisory committee voted 6‑3 that AstraZeneca’s late‑stage trial of camizestrant, an oral selective estrogen‑receptor degrader (SERD), did not demonstrate a clinically meaningful benefit. AstraZeneca had positioned camizestrant as a potential first‑in‑class oral therapy for hormone‑receptor‑positive metastatic breast...
Siemens Healthineers to Receive $60M in Federal Funding for Key Cancer Therapy
Siemens Healthineers will receive up to $60 million from ARPA‑H over five years, complemented by a $23 million cost‑share, totaling about $83 million. The funding targets development of photon flash therapy, an experimental radiation technique that delivers doses 100 times faster than conventional methods...
Task Switching Raises Risk in Transplant Surgeries, Study Finds
A Virginia Tech analysis of more than 300,000 transplant operations shows that surgeons who switch organ types between consecutive procedures raise one‑year patient mortality by 14.8%. The risk spikes when the switch occurs on the same day, lifting mortality from 4.5%...

Higher Tubular Phosphate Levels Linked to Faster Five-Year Kidney Decline
Researchers at the University of Tsukuba analyzed 308 Japanese participants and found that higher estimated proximal tubular fluid phosphate (ePTFp) levels are linked to a faster decline in kidney function over five years. The ePTFp metric, derived from routine serum...

Scaling the Agentforce Life Sciences Ecosystem to Drive the Future of Pharma and MedTech
Salesforce announced a major expansion of its Agentforce Life Sciences ecosystem, forging deeper collaborations with global systems integrators, content partners, and independent software vendors to accelerate adoption of the Agentforce 360 platform. Leading SIs such as Accenture, Deloitte Digital, Cognizant...

States Modernize PA Practice Laws to Expand Healthcare Workforces
A wave of state legislation is expanding physician assistant (PA) autonomy as part of the federal Rural Health Transformation Program (RHTP). Seven states, including Iowa and Kentucky, have removed mandatory supervisory agreements, allowing PAs to practice independently and, in Kentucky,...

From Toddlers to Teens: The Hidden Complexities of Bringing Pediatric Wearables to Market
Pediatric wearables face unique challenges beyond miniaturization, requiring designs that adapt to rapidly changing physiological, cognitive, and behavioral traits from infancy to adolescence. Companies must address divergent safety, usability, data reliability, and algorithm performance needs across developmental stages, while managing...

Heart Rhythm 2026: Electrophysiologist Mina Chung Discusses CPR, PFA and Much More
At Heart Rhythm 2026, HRS President Mina Chung outlined a multi‑pronged agenda that includes a new task force to expand CPR and AED training in high schools, leveraging digital‑health apps for continuous skill retention. The society also launched a real‑world...

Trump Withdraws Wellness Influencer and MAHA Activist Casey Means as Surgeon General Nominee
President Donald Trump withdrew wellness influencer Casey Means from the surgeon‑general nomination and named radiologist and Fox News contributor Nicole Saphier as his third pick. Means faced Senate opposition over her anti‑vaccine, abortion‑pill, and alternative‑medicine positions, while Saphier brings clinical...

Healthcare Affordability Part 4: How Annuitants Can Use Expected Healthcare Costs to Help Choose the Right Health Plan
Retirees who receive federal annuities must navigate Medicare Parts A and B while deciding between traditional FEHB coverage, Medicare Advantage (MA) offered through FEHB carriers, or commercial MA plans that require suspending FEHB. Part B carries a $202.90 monthly premium, with...

After Reductions, VA Chief Says Facilities Can 'Hire Where They Need and What They Need'
Veterans Affairs Secretary Doug Collins told the Senate Appropriations Committee that no VA facility faces hiring constraints, despite the department’s recent staffing caps and a 30,000‑person workforce reduction. He emphasized that baseline FTE limits are administrative and will not block...

Ride in Support of People Living with Diabetes at the 2026 Tour De Cure: Pacific Northwest
On May 2, 2026 the American Diabetes Association will host the Tour de Cure Pacific Northwest in Bothell, Washington. The one‑day cycling fundraiser expects 168 participants to ride from 7 a.m. to 2 p.m., raising money for diabetes research, advocacy and local programming....

Genmab A/S (NASDAQ:GMAB) Upgraded at Truist Financial
Truist Financial upgraded Danish biotech Genmab A/S (NASDAQ:GMAB) to a strong‑buy, joining recent upgrades from Goldman Sachs and BNP Paribas. The stock currently trades around $26.11, well below its 52‑week high of $35.43, with a market cap of $16.78 billion and a...
Doctors Agree: There’s a Lot to Love About Cardiology’s Future
A Medscape survey of nearly 6,000 U.S. physicians shows 59% of cardiologists are optimistic about the short‑term outlook and 48% about the long‑term, placing cardiology in the top ten specialties for confidence. Only 10% express short‑term pessimism and 24% long‑term...
Vaccine Hesitancy in an Era of Misinformation
The CDC revised its autism‑vaccine statement in November 2025, moving from a definitive “no link” stance to an uncertainty‑based message. A Science‑journal survey of 2,900 U.S. adults found that exposure to the new wording raised perceived vaccine risks and lowered...
Communication From the CDC Fuels Skepticism About Vaccines and Science, Research Suggests
The CDC altered its website language to suggest that a link between vaccines and autism cannot be ruled out, diverging from the long‑standing consensus that no causal relationship exists. An international team led by the University of Vienna surveyed 2,989...

Caregivers of Ovarian Cancer Patients Face High Stress and Major Gaps in Support
A scoping review by the University of Toronto, published in PLOS One, examined 32 studies over 25 years and found that caregivers of ovarian cancer patients endure high anxiety, depression, grief, and burnout. The analysis highlighted practical barriers such as financial...

Omada Health Expands Nationwide via OptumRx Weight Engage
A milestone for @OmadaHealth: we're now available through @OptumRx's Weight Engage — and with it, employers can purchase Omada through all 3 of the nation's largest PBMs, reaching the majority of commercially insured lives in the U.S. Expanding reach has been...
BIO Panel at HERS Explores Women’s Health Biotech Ecosystem
The Biotechnology Innovation Organization (BIO) convened a panel at the inaugural Health Executive and Research Summit in San Diego to spotlight the women’s health biotech ecosystem. Speakers noted that only six percent of private healthcare investment goes to women’s health,...

Novartis Finalizes US Expansion with Seventh Facility to Strengthen End-to-End Drug Manufacturing
Novartas has completed its US expansion plan by adding a seventh facility in Morrisville, North Carolina, dedicated to API production for oral solid‑dose and RNA‑based therapies. The site is part of a $23 billion multi‑year investment to build an end‑to‑end domestic...

Eli Lilly Shares Surge on a Beat-and-Raise, Proving the Stock's Swoon Was a Mistake
Eli Lilly reported first‑quarter 2026 revenue of $19.8 billion, up 56% year‑over‑year and well above the $17.6 billion consensus, while adjusted earnings per share jumped to $8.55, more than double the prior year and beating the $6.66 estimate. The strong top‑ and bottom‑line...

Redo Mitral Valve Replacement Surgery Outdoes ViV Over the Long Term
A retrospective analysis of 229 patients treated for failed bioprosthetic mitral valves found that 30‑day mortality and readmission rates were similar for repeat surgical mitral valve replacement (SMVR) and transcatheter valve‑in‑valve (ViV) procedures. However, at five years SMVR patients experienced...
Rare Myocarditis After mRNA Vaccination: Mitochondrial Stress Identified as a Key Factor
Researchers at the University of Tsukuba identified mitochondrial stress as a key driver of the rare myocarditis cases observed after COVID‑19 mRNA vaccination. By analyzing heart biopsies and a mouse model with subclinical mitochondrial impairment, they showed that vaccine lipid...

At UToledo Health, Ambient AI Decreases Open Charts, Improves Documentation
UToledo Health piloted Nabla’s ambient AI documentation tool with 40 providers across multiple specialties. Over an eight‑week period the system captured real‑time clinical conversations, generating structured notes that were integrated into Epic, resulting in more than 3,000 documented encounters. The...

Cigna Sez Sayonara: Another Major Carrier Bailing on ACA Exchanges Next Year
Cigna announced it will exit the individual health‑insurance market under the Affordable Care Act in 2027, leaving roughly 369,000 members in 11 states to find new coverage. The move follows a 17% year‑over‑year drop in Cigna’s ACA enrollment, even as...
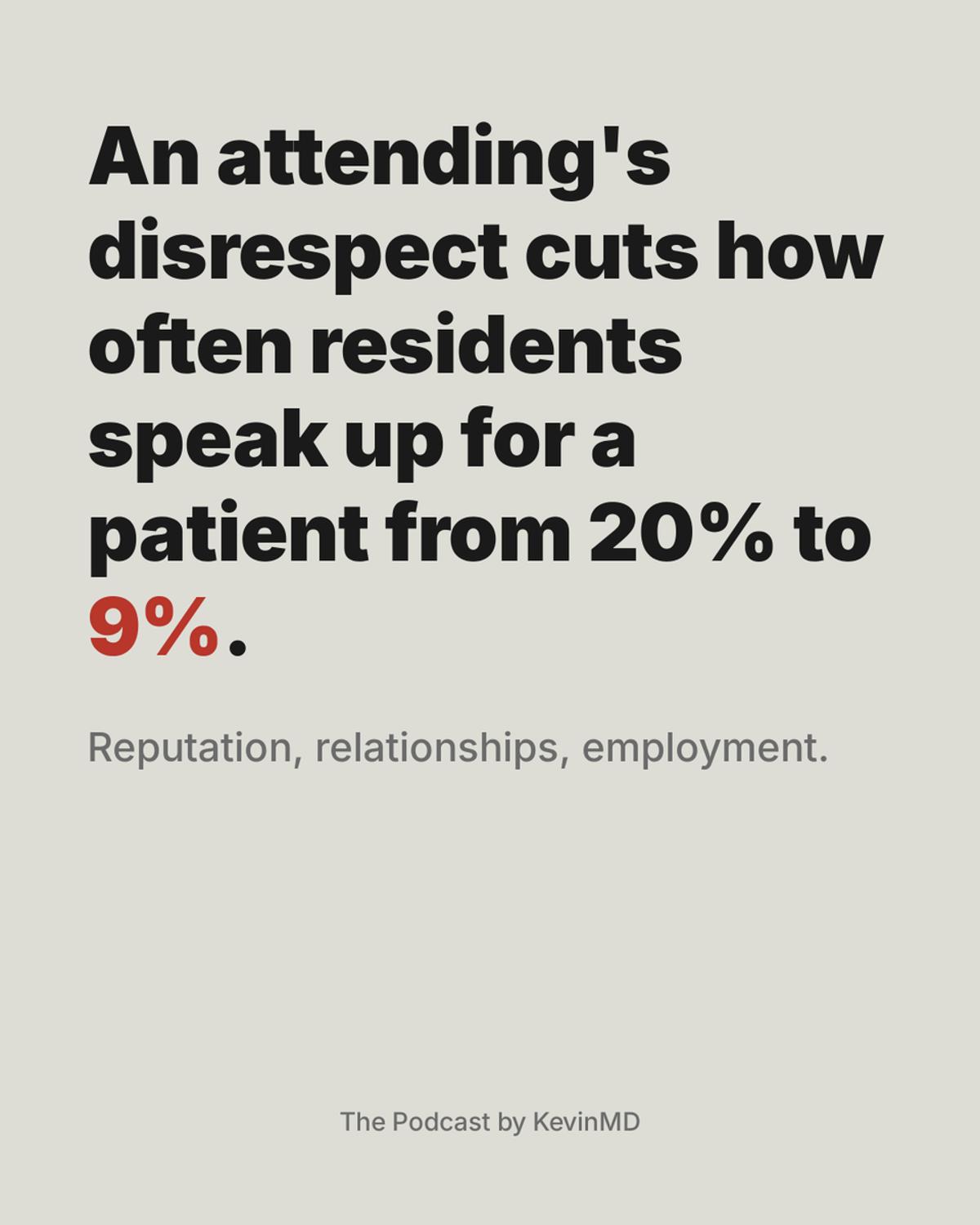
Disrespectful Attendings Silence Residents, Undermining Patient Safety
Only 9% of residents will challenge an attending about to harm a patient, if that attending has been disrespectful to them before. When the attending is respectful and patient harm is the only variable, around 20% of residents will speak...
Diabetes Flips Immune Cells From Repair to Inflammation in Peripheral Artery Disease, Study Finds
A new study published in Science Translational Medicine shows that type 2 diabetes reprograms TREM2‑positive macrophages from a tissue‑repair mode to a pro‑inflammatory state, worsening peripheral artery disease (PAD). Researchers used single‑cell RNA sequencing and spatial transcriptomics on human arteries and...
Avalyn Pharma Takes a Breath to Raise $300M in IPO Cash for Lung Drug Trials
Avalyn Pharma completed a $300 million IPO, pricing 16.6 million shares at $18 each and listing on Nasdaq under the ticker AVLN. The Boston‑based biotech will use the proceeds to advance inhalable versions of pirfenidone (AP01) and nintedanib (AP02), plus a combination...

Driving Value-Based Practice Transformation Through Care Management
Community oncology practices are turning to care‑management models such as Principal Care Management, Transitional Care Management, and Principal Illness Navigation to deliver patient‑centric, value‑based care without assuming full financial risk. Real‑world examples—from cCARE in Fresno addressing food insecurity to Rocky...

Pharma Power Shifts: Controlling Diseases, Not Just Assets
Most pharma pipeline analysis is spreadsheet theater. I used AI to look at the 2020 → 2026 shift. The real question is not: Who has the most assets? It is: Who controls the disease category? GLP-1s are a metabolic land grab. Oncology is becoming a platform stack. Trials...

All Eyes on Fauci as Top Aide Indicted Over Scheme to Hide COVID Emails
Former NIAID senior adviser Dr. David Morens was indicted on federal charges for conspiring to hide COVID-19-related communications by using private Gmail accounts and deleting records. Prosecutors allege he created a backchannel to Dr. Anthony Fauci to evade FOIA requests...

Pharma Pulse: The Ember Cube 2 and a $1 Trillion Prescription Spending Projection for 2026
Ember LifeSciences unveiled the Ember Cube 2, a modular cold‑chain solution with real‑time monitoring, GPS tracking and cloud visibility, earning a Red Dot “Best of the Best” award. At the Asembia AXS26 summit, policy experts warned that new pricing legislation is compressing post‑launch...
Your Oral Microbiome Could Affect Your Weight, Liver and Diabetes Risk
The study, one of the largest to date, examined the oral microbiome of thousands of participants and found distinct bacterial signatures associated with obesity, pre‑diabetes and non‑alcoholic fatty liver disease. Researchers identified specific taxa whose abundance correlated with insulin resistance...

This Gas Provides Rapid Relief For Major & Treatment-Resistant Depression (M)
Recent clinical investigations show that inhaled nitrous oxide can alleviate symptoms of major depressive disorder, including cases resistant to standard therapies, within hours of administration. Across multiple small‑scale trials, roughly 40% of participants achieved remission after a single session, with...

Physician Career Choices Come Down to Risk Tolerance
Physician career decisions often hinge on risk tolerance, with most doctors opting for stable, modest pay raises (Career A) rather than high‑potential, uncertain roles (Career B). Dr. Stanley Liu argues that a robust financial plan—tax optimization, reserve funds, debt strategies, and contract...
Surgeon Wears Apple Vision Pro to Fix Cataract in Medical First
In October 2025, Dr. Eric Rosenberg performed the world’s first cataract surgery using Apple’s Vision Pro mixed‑reality headset paired with a custom ScopeXR app. The system streams 3D microscope feeds to the headset, allowing the surgeon to see a stereoscopic...

FDA Expedites Review of New HIV Treatment Combo
The FDA has granted priority review to Gilead Sciences’ new single‑tablet HIV regimen that combines bictegravir and lenacapavir. The application is backed by phase 3 ARTISTRY‑1 and ARTISTRY‑2 trials, which showed sustained viral suppression in adults already virologically controlled. If approved,...
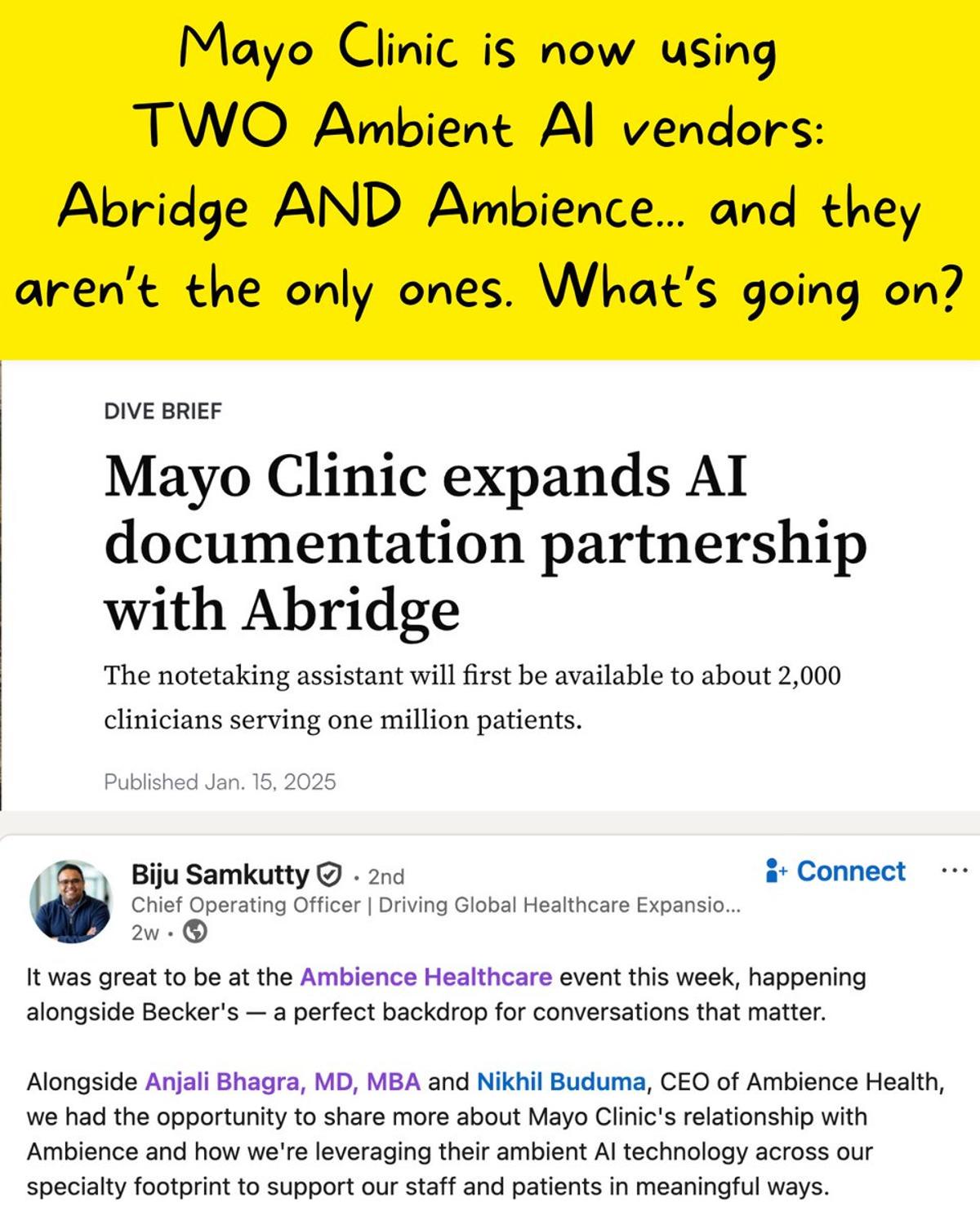
Health Systems Add Multiple AI Vendors for Specialty Precision
Mayo Clinic partnered with Abridge initially - and now is adding Ambience for specialty care. That’s MORE point solutions - not less. What’s going on here? Recently I spoke to a CMIO whose health system is also planning to add a...
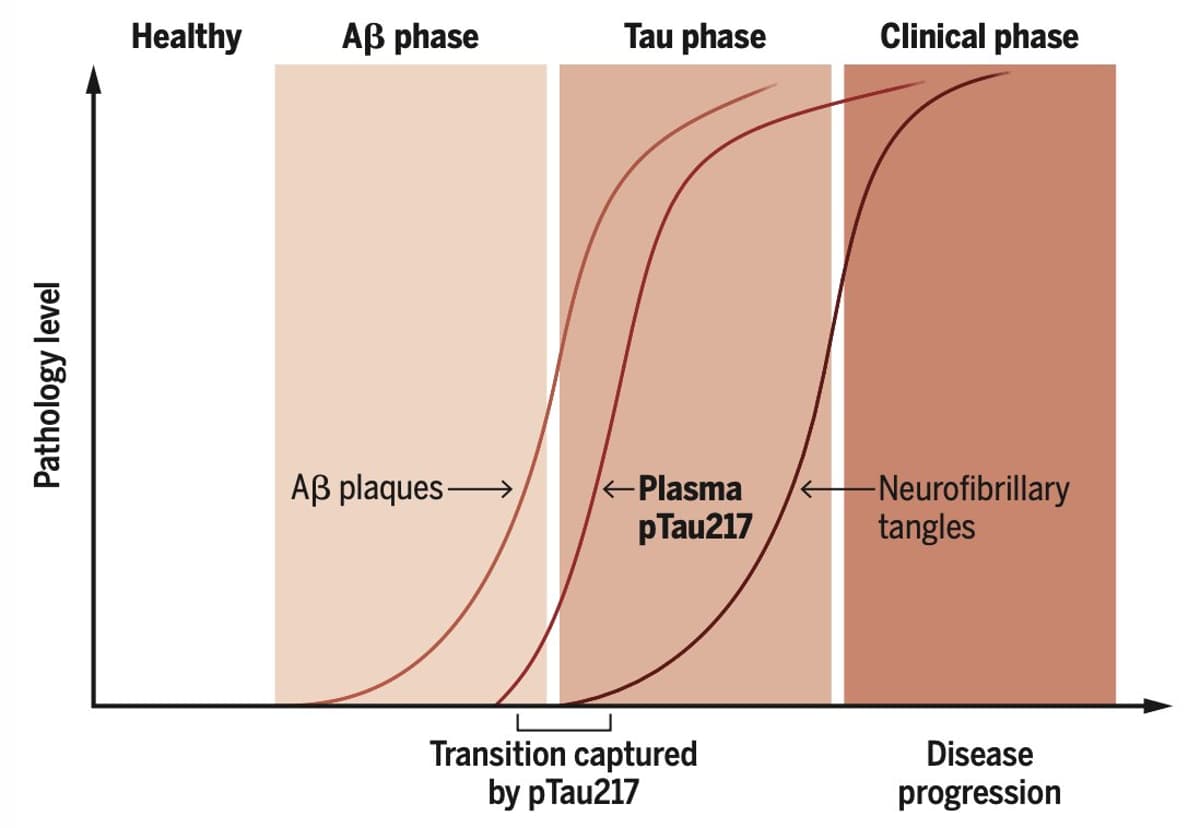
P‑tau217 Blood Test Predicts Alzheimer’s Risk Early
Good summary of p-tau217, the breakthrough blood test to predict risk of Alzheimer's in people well before onset of symptoms @ScienceMagazine https://t.co/qVJtYR4bnz https://t.co/OhjLExTroG

FDA Moves to Speed Review Support for Psychedelic-Related Treatments
The FDA announced a new fast‑track mechanism for psychedelic‑based therapies, issuing Commissioner’s National Priority Vouchers to three programs targeting depression and PTSD with psilocybin or methylone. It also granted an IND for DemeRx NB to study noribogaine hydrochloride as a...

Outsourced Teams Can Seamlessly Blend Into Health Systems
Can outsourced teams feel like part of your health system? Gene Scheurer of Optimum Healthcare IT says yes—with secure, branded environments that mirror your organization. Full interview from ViVE 2026 🔗 https://t.co/cPkAwasnxg @OptimumHIT #ViVE2026 #HITSM https://t.co/1cw3rf1g6W
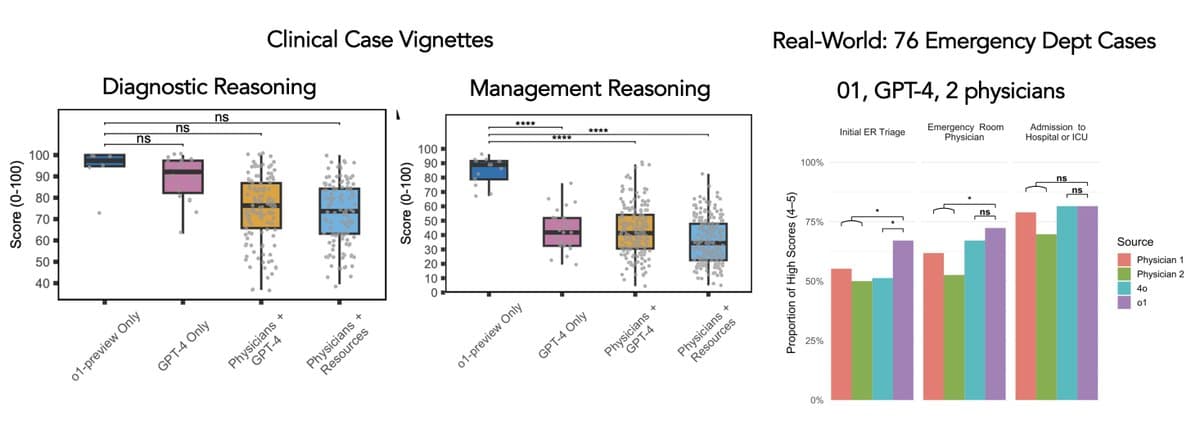
O‑1 Model Beats GPT‑4 and Doctors in Triage
New @ScienceMagazine The o-1 reasoning model (text only, from @OpenAI, releasedSept 2024) exceeded performance cf GPT-4 and physicians for clinical vignette management reasoning and in a real-world emergency department assessment for initial triage @AdamRodmanMD @PeterBrodeurMD @arjunmanrai @jonc101x https://t.co/tPZqZAE8cd
Trump Replaces Surgeon General Pick with Radiologist Nicole Saphier
Trump drops Casey Means as surgeon general nominee, opts for radiologist Nicole Saphier https://t.co/mvCUOymapm via @statnews