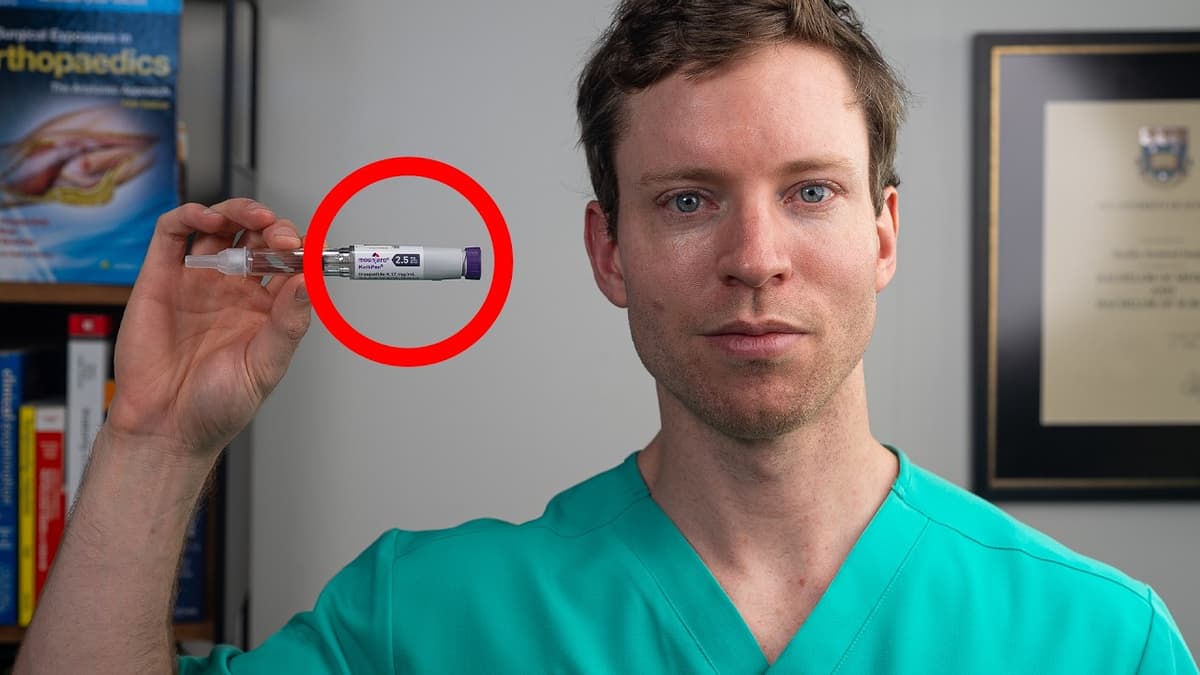
The Low-Dose Peptide I Take
The video follows a 34‑year‑old general practitioner who injects 1.25 mg of tirzepatide weekly despite lacking diabetes or obesity, using the drug to tap into emerging evidence that GLP‑1 and GIP agonists confer health benefits beyond weight loss. Recent large‑scale trials—SELECT, FLOW, and STEP‑9—show semaglutide cuts major cardiovascular events by 20 % in non‑diabetic patients, reduces kidney‑failure composites by 24 % independent of weight loss, and alleviates osteoarthritis pain. Parallel mouse studies demonstrated cartilage preservation that could not be explained by weight reduction alone. In the SELECT trial 17,000 overweight adults saw cardiovascular risk fall even when weight loss was minimal. The FLOW trial reported similar kidney benefits. A Shenzhen team gave semaglutide to mice with osteoarthritis and matched‑weight‑loss controls; only the drug‑treated group retained cartilage. A pilot MRI study in 20 humans showed a 17 % increase in knee cartilage thickness after 24 weeks of tirzepatide plus hyaluronic acid. These findings suggest GLP‑1/GIP agonists may act on receptors in heart, kidney and cartilage cells, offering therapeutic avenues for lean, non‑diabetic individuals. While the physician’s self‑experiment highlights potential personal benefits—reduced cravings, lower blood pressure, and minimal side effects—it also underscores the need for controlled trials before broader off‑label adoption.

Shining a Light on Bladder Cancer Detection
MIT researchers have engineered a urinary catheter coated with carbon‑nanotube nanosensors that light up when they encounter bladder‑cancer‑specific protein biomarkers. The device scans the bladder with a laser‑based system, producing a fluorescent “chemical image” that pinpoints where malignant molecules are...

Keeping Cancer Locked Up
The video spotlights Wakako, a post‑baccalaureate fellow at the National Institutes of Health, who is investigating the earliest mechanical steps of cancer metastasis. She explains that most cancer fatalities stem from tumor cells breaking away from the primary site, forming...

Neuralink's DJ Seo: Inside the Race to Connect Brains and AI
The video showcases Neuralink co‑founder DJ Seo discussing the company’s flagship brain‑computer interface (BCI) products—Telepathy, which lets locked‑in ALS patients control a computer with thought, and the upcoming BlindSight system that could restore vision by stimulating the visual cortex with...
Better to Freeze the Body or Just the Brain? #podcast #startup #cryopreservation
The podcast explores whether cryopreserving just the brain, rather than the entire body, is a more rational approach for future mind‑upload or revival technologies. Host and guests discuss the core premise that personal identity, consciousness, and personality are rooted entirely...

The Startup Freezing Humans For Tomorrow - Interview with Dr Emil Kendziorra, CEO of Tomorrow.bio
The EU Startups podcast featured an interview with Dr. Emil Kendziora, CEO of Tomorrow.bio, a European startup that provides cryopreservation services for both humans and pets. The conversation explored the company’s mission, funding, and the broader context of longevity research. Kendziora,...

EMV Capital Advances XF-73 Toward Late-Stage Trials for Surgical Infection Prevention
EMV Capital announced that its AMR Bio subsidiary is moving XF‑73, a novel anti‑infective, into the final FDA‑design phase of a Phase 3 trial aimed at preventing surgical site infections. XF‑73 has already cleared a successful Phase 2b study, showing near‑100% efficacy in...

Migraine Relief: How Brain Imaging Could Lead to Better Treatment | 90 Seconds W/ Lisa Kim
The video highlights emerging research that combines functional brain imaging, blood and spinal‑fluid biomarkers, and AI‑driven data analysis to redefine how migraines are diagnosed and treated. Migraine affects roughly one in four adults and imposes over $20 billion in U.S. productivity...

Does Ozempic Raise Testosterone?
The video examines whether Ozempic and other GLP‑1 receptor agonists raise testosterone in men, focusing on weight‑loss‑driven hormonal changes. Data show a 10% body‑weight reduction lifts testosterone by roughly 84 ng/dL, while bariatric surgery‑induced 20‑30% loss can add about 250 ng/dL. GLP‑1 drugs...

Is There an Effective Vaccine for the Ebola Outbreak? | Asked & Answered
An Ebola outbreak in the eastern Democratic Republic of Congo, identified as the Bundibugyo strain, has sparked urgent questions about vaccine availability. Health officials confirm no approved vaccine exists that reliably protects against this specific strain, and the existing Zaire‑targeted...

Could a Pill Prevent the World’s Deadliest Cancer?
The video explores a emerging strategy to prevent lung cancer by targeting inflammation rather than solely focusing on genetic mutations. Researchers at Mount Sinai, led by Dr. Miriam Merad and Dr. Tom Marron, argue that up to 80% of lung...
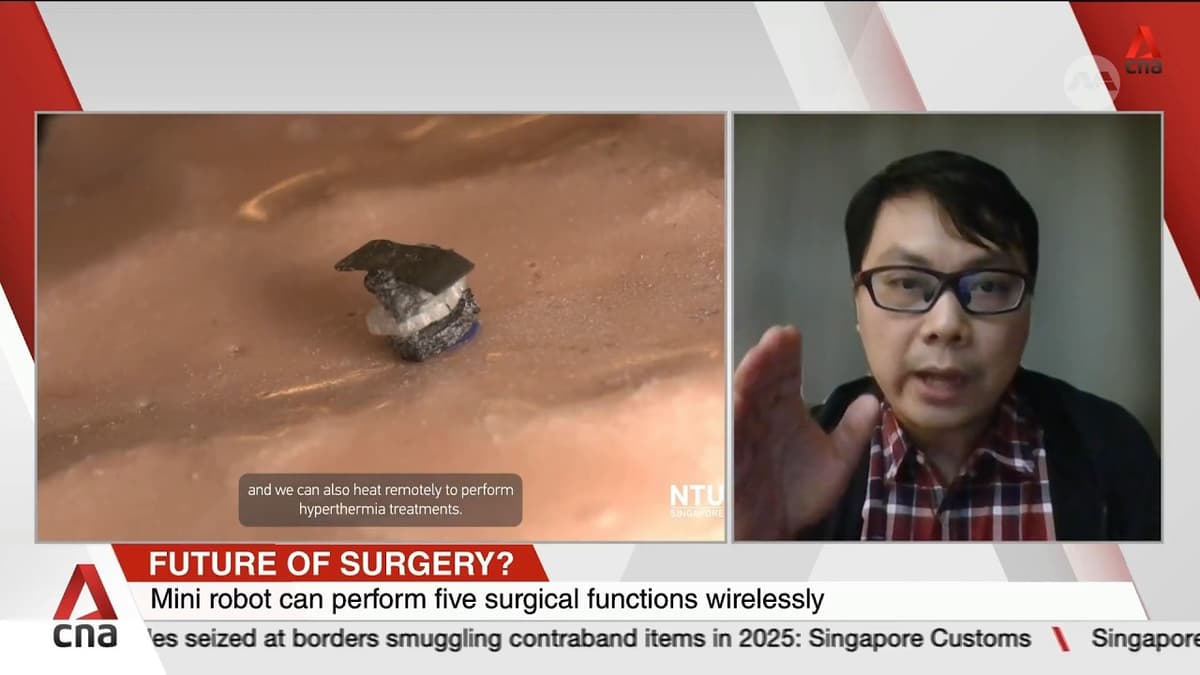
NTU Scientists Develop Seed-Sized Surgical Robot
Researchers at Nanyang Technological University in Singapore have unveiled a seed‑sized robot that can be injected into the body and perform a suite of surgical tasks. The device is steered by external magnetic fields that can be modulated to move, cut...

HERology | GLP-1 Hormones and Weight Loss
The HERology podcast from Mount Sinai explores the surge of GLP‑1 hormone agonists, focusing on their role in weight management and broader health implications for midlife women. Host Dr. Joanne Stone and co‑hosts Dr. Anu Lala and Dr. Anna Barbieri...

Rare Disease Drug Commercialization With Zevra Therapeutics' Neil McFarlane
In a Business of Biotech interview, Zevra Therapeutics CEO Neil McFarlane outlined the company’s recent rebrand from KemPharm and its sharpened focus on developing and commercializing rare‑disease therapeutics. He traced his unconventional path—from Army Reserve medic to transplant nurse practitioner—to...
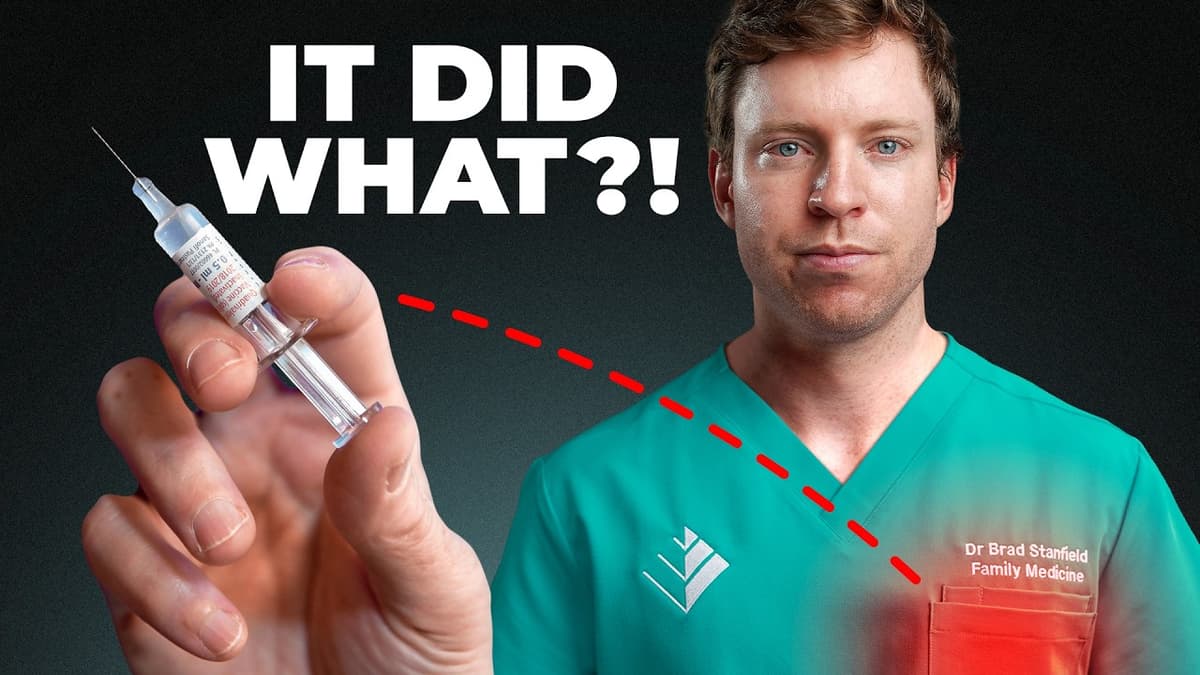
This Vaccine Is Quietly Doing Something to Your Heart
Recent research highlights that the shingles vaccine Shingrix, already approved for preventing herpes zoster, also appears to confer significant heart health benefits. Large‑scale meta‑analyses and real‑world studies report up to a 30% reduction in stroke and a comparable drop in...

Accelerating Genome Analysis @ RECOMB-ARCH 2026
At RECOMB-ARCH 2026, the speaker traced two decades of work at the intersection of computer architecture and bioinformatics, highlighting community growth, education efforts, and the role of hardware-aware system design in genomics. He focused narrowly on accelerating genome sequence analysis—particularly...
Could Telomeres Reverse Aging?
The video explores how telomeres—protective caps on chromosome ends—govern cellular longevity and, by extension, organismal aging. It highlights the biological limit on cell division imposed by telomere shortening and introduces Telomere Pharmaceuticals’ experimental drug designed to lengthen telomeres directly. Key points...

New S$1.4b Biomedical Sciences Hub to House A*STAR, R&D Firms
Singapore announced a S$1.4 billion biomedical sciences hub that will house the nation’s public research agency A*STAR alongside leading multinational biotech firms. The development is part of a broader S$37 billion commitment to research, innovation and enterprise over the next five years,...
Thirty Years of Free Therapy: Sanofi's Rare Humanitarian Program - Life Sciences Today Podcast Ep 62
The Life Sciences Today podcast highlighted Sanofi’s Rare Humanitarian Program, now in its 35th year, which provides free access to its orphan‑drug portfolio for patients in countries without commercial availability. Since its 1991 launch, the initiative has served more than 4,000...

GLP‑1 Gene Therapy: A One‑Time Fix for Obesity & Diabetes? | Longevity News Roundup — Week 21, 2026
The longevity roundup highlighted several breakthrough therapies, most notably Fractyl Health securing European approval for RJVA-001, a GLP‑1 gene therapy that programs pancreatic beta cells to secrete GLP‑1 after meals, potentially eliminating the need for chronic injections in obesity and...
![[Audio Descriptions] Faculty In Focus: Jeff Nivala](/cdn-cgi/image/width=1200,quality=75,format=auto,fit=cover/https://i.ytimg.com/vi/663CzfAzKZc/maxresdefault.jpg)
[Audio Descriptions] Faculty In Focus: Jeff Nivala
Jeff Nivala, an assistant professor at the Paul G. Allen School of Computer Science & Engineering, leads the Molecular Information Systems Lab developing technologies to read individual, full-length protein molecules end-to-end. His interdisciplinary team—combining molecular biology, biochemistry, microbiology, machine learning,...

Magic Lifescience at NIH POCTRN | Shark Tank Pitch for Point-of-Care Diagnostics
Magic Lifescience, a Stanford spin‑out, delivered a Shark‑Tank‑style pitch at the NIH POCTRN showcase, positioning its platform as a next‑generation point‑of‑care (POC) diagnostic solution. The founders, CSO Elaine Ng and CFO Tianhao, explained how their original cancer‑focused research was re‑engineered...
Gut Health Is Becoming Programmable
The video outlines an emerging field where the gut microbiome can be programmed, moving beyond generic probiotic advice to precision interventions. Researchers now identify optimal microbial compositions for individuals and deliver them via personalized transplants, currently in early human trials. Companies...
The Next Era of Healthcare Is Personal
The Mackenzie podcast episode spotlights Madna’s vision of a personal, information‑driven healthcare era. CEO Stefan Benell explains how the company’s mRNA platform treats DNA as an information molecule, allowing rapid design of vaccines, cancer immunotherapies, and treatments for rare genetic...

Strategy& Insider Podcast - Episode 46 with Lara Gervaise and Edoardo Guidice
The Strategy& Insider podcast featured Lara Gervaise and Edoardo Guidice, co‑founders of Vuosis AI, a Swiss EPFL spin‑off that uses voice analysis to flag early signs of fatal diseases, burnout and cognitive decline. Vuosis AI’s platform extracts hundreds of acoustic features—tone,...

Grey Matters Launches US Brain PET Clinics for Alzheimer’s Diagnostics
Grey Matters Health announced the opening of its first U.S. brain PET imaging clinics, branded NovaScan, to provide amyloid plaque detection for Alzheimer’s diagnosis. The company highlighted a letter of intent with Catalyst MedTech for at least 200 scans at...

The Lizard that Helped Create Ozempic #science #ozempic #podcast
Researchers tracing how certain reptiles survive long fasting periods discovered hormones in the Gila monster that inspired GLP-1–based drugs such as Ozempic and Mounjaro. The conversation highlights how basic biological research on these lizards led to first-in-class therapies for weight...

Where Organ on Chips Will Add the Most Value in 3–5 Years
The discussion centers on how contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) can accelerate the adoption of organ‑on‑chip technologies over the next three to five years. Participants stress that scaling commercial systems and creating a unified...

Audience Poll: Validation and Trust Are Biggests Barriers to Organ on Chip Adoption
An audience poll found that 46% of respondents say validation and trust are the biggest barriers to broader organ-on-chip adoption, with regulatory acceptance cited by 25%, cost and scalability by about 19%, and workflow integration by 9%. Panelists said the...

The Reality of Human on a Chip Systems
Researchers and teams have demonstrated multi-organ “human-on-a-chip” systems—ranging from DARPA-funded 10-organ demos to focused gut–liver and gut–liver–brain models—that reveal meaningful organ interactions and immune trafficking. These platforms have produced actionable biological insights, such as short‑chain fatty acids exacerbating inflammation in...

Should I Be Freaked Out by the Hantavirus?
The Oxford Sparks podcast tackled the recent Andes hantavirus outbreak aboard a cruise ship, featuring senior researcher Daniel Wright from Oxford’s vaccine group. Wright explained that hantaviruses are a family of rodent‑borne pathogens, with the Andes strain capable of occasional...

The Vitals | Pioneering Ketamine Treatment for Depression
The Vitals episode brings together Mount Sinai psychiatrists to discuss ketamine’s emergence as a fast‑acting antidepressant and its expanding role in treating depression and PTSD. Dr. Dennis Charney recounts the mid‑1990s Yale experiments that showed a single sub‑anesthetic ketamine infusion lifted...

Using AI to Outsmart Drug-Resistant Bacteria
The video addresses the escalating crisis of antimicrobial resistance, describing it as a silent global pandemic that forces the medical community to rethink how antibiotics are discovered and deployed. Traditional drug development struggles to keep pace as bacteria rapidly evolve,...

Envisioning Our Future for Children: "Al and Microphysiological Systems Transforming Biomedicine"
Professor Thomas Hartung opened the session by framing a historic transition: after 35 years of advocating alternatives, AI and microphysiological systems are now poised to supplant traditional animal testing in biomedical research. He highlighted how organ‑on‑a‑chip technologies and advanced computational...
The AI Super Scientist
The video introduces Alex Zhavoronkov, founder and CEO of In Silico Medicine, showcasing how the company leverages artificial‑intelligence‑driven drug discovery to tackle complex diseases. Using AI, the firm mined massive biomedical datasets to map idiopathic pulmonary fibrosis (IPF), identified key pathogenic...

Science Can't Wait: A Discovery Series | Part 3 | Featuring Cancer Researcher Daniel Hollern
The Science Can’t Wait webinar’s third installment spotlighted Salk Institute researcher Daniel Hollern’s work on leveraging the immune system—specifically B cells—to combat breast cancer. The session framed the effort as part of a broader interdisciplinary push, where basic questions...
One Pill Changed Everything
A clinical-trial medication from Telomere Pharmaceuticals given as a once-daily pill reportedly produced rapid, dramatic improvements in two rescue dogs: Zeus, a 12-year-old German Shepherd with terminal cancer, regained energy and appetite days after starting treatment, and Benson, a severely...
What if Aging Organs Could Actually Repair Themselves?
The video explores emerging regenerative therapies that pair stem‑cell delivery with epigenetic reprogramming and retinoic‑acid signaling to repair age‑related organ damage rather than replace organs outright. Researchers argue that activating retinoic‑acid pathways in kidney tissue, together with guided stem‑cell progenitors, could...
Regenerative Healing Gene: Rewriting Aging Rules! #shorts
The video highlights a breakthrough study that identified a single gene, ALDH1A2, capable of restoring scar‑free, complete tissue regeneration in mammals. By comparing regenerating and non‑regenerating rodents, researchers pinpointed ALDH1A2 as the most dramatically up‑regulated gene after injury, linking its...

Age-Related Macular Degeneration in 90 Seconds
The video explains age‑related macular degeneration (AMD), the leading cause of permanent vision loss among adults in the United States and many other nations. AMD develops when lipid‑rich drusen accumulate beneath the retina and the retinal pigment epithelium (RPE), impairing...

Preventing Dementia with Shingles Vaccination? | MGR | 22 April 2026
The video outlines a series of natural‑experiment studies suggesting that the live‑attenuated shingles vaccine may significantly lower dementia incidence. By exploiting birth‑date eligibility cut‑offs used in the UK, Australia, Canada and New Zealand, researchers compare cohorts that differ only in...

The Future of Fungi
The Stanford Engineering podcast explores the emerging frontier of fungi, hosted by Russ Altman and featuring bioengineer‑chef Vayu Hill‑Maini. Hill‑Maini argues that mushrooms and molds are poised to become a cornerstone of future food, pharmaceuticals, and novel materials, leveraging their...
This Compound Reverses Immune Aging — Here's Why Scientists Are Stunned
The video focuses on urolithin A, a gut‑derived metabolite of ellagitannins found in pomegranate juice, that activates mitophagy – the selective removal of defective mitochondria – and appears to rejuvenate multiple physiological systems. Researchers highlighted how this compound boosts immune health...
Building Companies at the Edge of Science and Market - Life Sciences Today Podcast Episode 61
The Life Sciences Today podcast features Jennifer Ernst, a rare hybrid who has moved from high‑tech device work at Xerox PARC to bio‑electronic medicine. Her career is defined by matching breakthrough science with clear market opportunities, from printed‑electronics roll‑to‑roll manufacturing...
TheraCryf CEO on Why Latest Toxicology Results Represent a Major Milestone
The CEO of Theracryf announced that the company has reached a pivotal pre‑clinical milestone, completing a high‑dose toxicology study in rodents and preparing a parallel study in mini‑pigs. This marks the final set of safety assessments required before filing an...

Jumping Genes: How Mobile DNA Is Reshaping Pathogens and Therapies | MGR | 29 April 2026
The talk centered on mobile genetic elements—commonly called jumping genes—and their role in reshaping bacterial pathogens within the human gut, especially in hematopoietic cell‑transplant patients. By sequencing stool and blood isolates, the speaker showed that roughly 40% of bloodstream infections...

Abortion Access In The High Court, Again | Katie Keith
The podcast focuses on a fresh legal battle over the FDA’s 2023 decision to allow mifepristone – the drug used for medication abortions – to be prescribed via telehealth and dispensed at pharmacies. Louisiana’s attorney general, joined by a private...

Sean Spencer, MD, PhD, Fellow ’20, Postdoc ’22 | Harnessing Gut Microbes to Heal Patients
Dr. Sean Spencer, a Stanford gastroenterologist and physician‑scientist, presented the emerging clinical frontier of gut‑microbe therapeutics. He outlined how advances in sequencing, culturing and sampling are moving the microbiome from a research curiosity to a practical diagnostic and therapeutic tool. Three...

Jean Tang, MD ’99, PhD ’03, Resident ’07 | Personalized Gene Therapy to Treat Rare Disease
Dr. Jean Tang, a Stanford dermatologist, detailed her two‑decade journey developing a personalized gene‑therapy for epidermolysis bullosa (EB), a rare disorder affecting one in 100,000 where patients lack functional collagen VII. Using a retroviral vector to deliver the 9 kb...

Faculty In Focus: Jeff Nivala
Assistant Professor Jeff Nivala of the Paul G. Allen School outlines a new technology that reads individual protein molecules end‑to‑end, preserving their native structure. The Molecular Information Systems Lab sits at the crossroads of computer science and biotechnology, aiming to...