
AbbVie Tops Q1 Estimates, Raises Outlook and Discontinues Cancer Candidate
AbbVie reported $15 billion in first‑quarter revenue, a 12.4% year‑over‑year increase, driven by strong immunology and a 26% surge in neuroscience sales. Immunology products Skyrizi and Rinvoq generated $7.29 billion, while the migraine drug Qulipta boosted neuroscience revenue to $2.87 billion. The company lifted its 2026 adjusted EPS outlook to $14.08‑$14.28 and announced a $650 million upfront deal for a PD‑1/VEGF bispecific, plus a potential $1.45 billion KRAS inhibitor acquisition. It also discontinued the BTK degrader ABBV‑101 after Phase 1 testing.
Genome Editing Can Be Risky. Meet the Epigenome Editors
Scientists are turning to epigenome editors—tools that rewrite gene activity without altering the DNA sequence—to sidestep the safety concerns of traditional genome editing. By coupling dead Cas9 (dCas9) with epigenetic modifiers, researchers can turn genes on or off with high...

FDA to Pilot Real-Time Clinical Drug Trials Through Cloud and AI
The FDA announced a pilot that will stream clinical‑trial data to the agency in real time using cloud platforms and artificial‑intelligence analytics. Commissioner Marty Makary said the effort could shave up to 40% off the time between Phase 1 trials and...

Supply Chain Digital Twins: An Evolution, Not a Breakthrough
Researchers at NIST and EMD Millipore argue that digital twins can model the intricate biopharmaceutical supply chain, from demand shocks to distribution bottlenecks. By creating in‑silico replicas of cells, raw materials, and logistics flows, twins could identify alternative distribution centers and...

Milk Exosomes Transform Therapeutic Bioprocessing
Milk-derived extracellular vesicles, known as milk exosomes, are emerging as a biocompatible platform for therapeutic delivery. Researchers have loaded the JAK inhibitor tofacitinib into exosomes (mEXOs@TOF) for ulcerative colitis, achieving high drug‑loading efficiency, stability and strong anti‑inflammatory effects without toxicity....

Hopes Raised for More Sustainable Oligonucleotide Manufacturing
QurAlis CTO Hagen Cramer says enzymatic synthesis could make large‑scale oligonucleotide production far more sustainable than the solvent‑intensive solid‑phase method. While solid‑phase synthesis remains fast and automated, it generates high process mass intensity due to extensive solvent washes. Enzymatic, aqueous‑based...

Astellas’s R&D Head Talks Strategy Ahead of Looming Patent Cliff
Astellas Pharma faces a looming patent cliff as its blockbuster prostate‑cancer drug Xtandi, which generated about $4.6 billion in the 2025 fiscal year, loses U.S. patent protection next year. To offset the expected revenue hit, chief R&D officer Tadaaki Taniguchi is accelerating...
Henlius and Organon Gain EC Approval for Pertuzumab Biosimilar
Henlius and Organon have received the first European marketing authorization for a pertuzumab biosimilar, Poherdy, targeting HER2‑positive breast cancer. The EC approval mirrors the product’s earlier U.S. clearance and covers all indications of Roche’s reference drug, Perjeta, including metastatic, neoadjuvant...

Pfizer Earns Positive Phase 3 in Multiple Myeloma; ICON Overstated Revenue
Pfizer announced that its antibody‑drug conjugate Elrexfio achieved a statistically significant and clinically meaningful improvement in progression‑free survival for patients with double‑class exposed relapsed or refractory multiple myeloma. The Phase 3 trial met its primary endpoint, positioning the drug as a...

After Apellis Deal, Biogen Looks to Fill Early-Stage Pipeline
Biogen announced a $5.6 billion acquisition of Apellis Pharmaceuticals, adding a complement‑inhibition platform to its portfolio. While the deal closes this month, the company emphasized that its growth engine will now hinge on early‑stage development and research rather than late‑stage launches....

Brenntag Pharma Unveiled BYPHAR, the New Regulated Biopharma Manufacturing Brand
Brenntag Pharma introduced BYPHAR, a new regulated biopharma manufacturing brand that consolidates high‑quality raw materials, advanced facilities, and value‑added services into a single, compliance‑ready portfolio. The brand groups materials into three tiers—Explore, Enhance, and Excel—covering non‑GMP to LBLE critical grades,...

With Growth Curve Trending up, Biogen Looks to Early-Stage Assets for ‘Next Generation’
Biogen, fresh from its $5.6 billion acquisition of Apellis, announced on its Q1 earnings call that it will pivot toward early‑stage assets to thicken a pipeline it described as “quite thin.” The shift comes as Alzheimer’s patients transition off Eli Lilly’s Kisunla,...
Circio Partners with TraffikGene Project to Advance Non-Viral circVec Delivery
Circio, an Oslo‑based circular RNA company, has partnered with the Universidad de Santiago de Compostela’s TraffikGene project to explore non‑viral delivery of its circVec circular RNA expression vectors. The collaboration merges Circio’s circVec platform with TraffikGene’s peptide amphiphile carrier system...
Biogen Ready to Catch Alzheimer’s Patients Transitioning Off Lilly’s Kisunla
Biogen is positioning its Leqembi therapy to capture Alzheimer’s patients who will finish Eli Lilly’s 18‑month Kisunla regimen and need a maintenance option. Leqembi, approved in January 2023, saw a 74% year‑over‑year sales jump to $168 million in Q1 2026, beating expectations. Biogen is...

AstraZeneca’s £300m UK Return Shows the Price Britain Paid to Win Back Pharma Money
AstraZeneca is restarting a £300 million (≈$380 million) research investment in the UK after the UK‑US pharmaceutical agreement boosted expected returns. The plan funds the completion of the Rosalind Franklin building in Cambridge and a new digital drug‑development lab, reflecting higher NHS...

Inside Servier’s New €200M Venture Fund
Servier has unveiled a new corporate venture fund, committing roughly $216 million to back early‑stage biotech focused on oncology and neurology. The fund, Servier Ventures, will target late pre‑clinical and Series A assets where it can add capital and industrial expertise, especially...

Candel Therapeutics Announces Commercial Agreement with EVERSANA to Support U.S. Launch of Prostate Cancer Therapy
Candel Therapeutics announced a commercialization agreement with EVERSANA to support the potential U.S. launch of its gene‑therapy candidate aglatimagene besadenovec (CAN‑2409) for intermediate‑ to high‑risk localized prostate cancer. EVERSANA will deliver a full suite of services, including data analytics, medical...

Chiesi Signs $1.9B Deal to Acquire KalVista and Its Approved Drug
Italian pharmaceutical group Chiesi announced a $1.9 billion cash acquisition of U.S. biotech KalVista Pharmaceuticals, paying $27 per share. KalVista brings an FDA‑approved therapy for a rare disease into Chiesi’s portfolio, bolstering its specialty drug offerings. The deal is part of...

Chiesi Expands Rare Disease Portfolio, Acquires KalVista for $1.9 Billion
Chiesi Group announced a $1.9 billion cash acquisition of KalVista Pharmaceuticals, its largest deal to date. The purchase adds EKTERLY (sebetralstat), the first oral, on‑demand therapy for hereditary angioedema (HAE), to Chiesi’s rare‑disease pipeline. EKTERLY is already approved in major markets...

STAT+: Pharmalittle: We’re Reading About the FDA Speeding up Trials, a Supreme Court Hearing on ‘Skinny Labels,’ and More
The FDA announced a pilot program that will review real‑time data from AstraZeneca and Amgen cancer trials using a platform built by Paradigm Health, aiming to speed regulatory feedback. The agency also opened a public comment period on a separate...

Pfizer Settles with Generic Drugmakers to Protect Blockbuster Drug Until 2031
Pfizer reached settlements with three generic manufacturers—Dexcel Pharma, Hikma Pharmaceuticals and Cipla—to postpone the launch of off‑patent copies of its heart‑disease drug Vyndamax until mid‑2031. The agreement stabilizes U.S. sales, which hit $3.8 billion in 2025, and averts the steep revenue...

When ADCs Meet Targeted Protein Degraders: The Emerging Field of Degrader-Antibody Conjugates
The biotech sector is exploring degrader‑antibody conjugates (DACs), a hybrid that merges antibody‑drug conjugate targeting with catalytic protein‑degradation payloads. C4 Therapeutics has expanded its partnership with Roche to co‑develop two undisclosed oncology DAC programs, while Orum Therapeutics secured $100 million to...

Should Pharma Be Swinging Bigger with AI? One Top Researcher Wants to Try
Kyunghyun Cho, a leading AI researcher known for co‑authoring the attention mechanism, used his platform at the International Conference on Learning Representations to argue that pharmaceutical companies should dramatically increase AI investment. He outlined a plan for a $50 million venture...

Teva to Acquire Emalex Biosciences for ~$900M
Teva Pharmaceutical Industries has signed a definitive agreement to acquire Emalex Biosciences for an estimated $900 million, comprising $700 million upfront and up to $200 million in commercial milestones. The purchase brings Emalex’s lead asset, ecopipam—a selective dopamine D1 receptor antagonist—into Teva’s neuroscience...

AstraZeneca Makes Surprise U-Turn with £300m Pharma Investment in UK
AstraZeneca announced a surprise £300 million (≈$381 million) investment in the UK, unfreezing a £200 million (≈$254 million) Cambridge expansion and allocating £100 million (≈$127 million) to a new "lab of the future" in Macclesfield. The move follows a pause last year after the company expressed...

Psychedelics Go Mainstream: Medicine, Mania Or Both?
Psychedelics are re‑emerging as a potential breakthrough in behavioral health, spurred by loosening regulations and a Trump‑issued executive order that accelerated research. Early clinical data suggest benefits for depression, PTSD and other conditions, prompting biotech stocks to rally after FDA...

Amgen, AZ Will Pilot FDA's Real-Time Clinical Trial Plan
The FDA unveiled a plan to receive clinical‑trial data in real time, aiming to accelerate drug development. AstraZeneca and Amgen have agreed to pilot the model with two studies that will stream endpoints and safety signals to regulators as they...

Revolution’s on a Pancreatic Cancer Winning Streak. What Comes Next for the Biotech?
Revolution Medicines announced that its oral RAS(ON) inhibitor daraxonrasib nearly doubled overall survival in previously treated metastatic pancreatic cancer, while its second candidate zoldonrasib showed tumor shrinkage in over half of KRAS‑driven lung cancer patients. The breakthrough data sent the...

The 20 Enterprise AI Drug Discovery & Life Sciences CEOs You Need to Know in 2026
The AI Insider has compiled a roster of the 20 most influential CEOs steering enterprise AI drug discovery and life‑sciences firms in 2026. The list showcases companies that are compressing decade‑long development cycles into months through generative protein design, AI‑optimized...

Drug Trials Snapshot: DAXXIFY
Revance Therapeutics’ DAXXIFY, a daxibotulinumtoxin A formulation, received FDA approval on September 7 2022 for temporary reduction of moderate to severe glabellar lines. Approval was based on two randomized, double‑blind, placebo‑controlled trials (GL‑1 and GL‑2) that enrolled 609 adults across the United States...

HUTCHMED Reports NMPA’s NDA Acceptance Under Priority Review for Sovleplenib to Treat wAIHA
HUTCHMED’s oral JAK2 inhibitor sovleplenib has received NDA acceptance and priority review from China’s National Medical Products Administration for warm‑antibody autoimmune hemolytic anemia (wAIHA). The submission is backed by Phase II/III data showing a 43.8% overall response rate versus 0%...

Drug Trials Snapshots: VOQUEZNA TRIPLE PAK, VOQUEZNA DUAL PAK
Phathom Pharmaceuticals’ VOQUEZNA TRIPLE PAK (vonoprazan, amoxicillin, clarithromycin) and VOQUEZNA DUAL PAK (vonoprazan, amoxicillin) received FDA approval for adult Helicobacter pylori infection based on a 992‑patient trial across six countries. In the modified intent‑to‑treat population, the triple regimen eradicated the...

GSK CEO Says Team Reorg Could Be in the Cards Pending Upcoming Phase 3 Readouts
GlaxoSmithKline (GSK) is banking on a slate of late‑stage Phase 3 trials slated for 2024, spanning oncology drugs and next‑generation vaccines. CEO Luke Miels warned on the first‑quarter earnings call that a series of missed readouts could trigger a reshuffle of...

Drug Trials Snapshot: OPDUALAG
OPDUALAG, a fixed‑dose combo of nivolumab and relatlimab, received FDA approval in March 2022 for adults and adolescents with unresectable or metastatic melanoma. The pivotal trial enrolled 714 patients and showed a median progression‑free survival of 10.1 months versus 4.6...

Drug Trials Snapshots: VONJO
VONJO (pacritinib) received FDA accelerated approval on Feb. 28, 2022 for adult myelofibrosis patients with platelet counts ≤50,000/µL. The decision rests on a single trial that enrolled 63 low‑platelet patients, of whom 31 received VONJO and 32 received best available therapy. In...

Drug Trials Snapshot: PYRUKYND
AGIOS Pharmaceuticals’ mitapivat, marketed as PYRUKYND, received FDA approval in February 2022 for treating hemolytic anemia caused by pyruvate kinase deficiency. In pivotal trials, 40% of non‑transfused adults achieved a ≥1.5 g/dL hemoglobin increase versus none on placebo, while 33% of...

Drug Trials Snapshots: ENJAYMO
ENJAYMO (sutimlimab‑jome) received FDA approval in February 2022 as the first therapy to reduce red‑blood‑cell transfusions in adults with cold agglutinin disease (CAD). Approval was based on a single open‑label, single‑arm trial (CARDINAL) that enrolled 24 patients across eight countries. After...

Drug Trials Snapshots: VABYSMO
VABYSMO (faricimab‑svoa), approved in January 2022, was evaluated in four pivotal trials involving 2,591 patients with neovascular age‑related macular degeneration (nAMD) and diabetic macular edema (DME). The drug was administered monthly for four doses before transitioning to a personalized schedule based...

Drug Trials Snapshot: CIBINQO
Pfizer's oral JAK inhibitor CIBINQO (abrocitinib) received FDA approval in January 2022 for adults with refractory moderate‑to‑severe atopic dermatitis. The approval rests on three phase‑3 trials involving 1,615 patients across 18 countries, which demonstrated robust skin‑clearance outcomes at week 12, especially with...

Drug Trials Snapshot: IMAAVY
IMAAVY (nipocalimab‑aahu) received FDA approval on April 29, 2025 for generalized myasthenia gravis in patients 12 years and older with AChR or MuSK antibodies. In a pivotal 24‑week Phase III trial of 196 adults, the drug achieved a statistically significant 1.5‑point improvement...

MPM Has Collected Three China Drugs for Its ‘Best of Both Worlds’ Strategy
MPM BioImpact, a biotech investment firm, has finalized the acquisition of three China‑origin drug candidates as part of its "best of both worlds" strategy. The deals were brokered through its portfolio company K2 Therapeutics, which now controls three distinct programs...

The Strategic Investments Expanding CDMO Capabilities for HPAPIs and ADCs
Contract development and manufacturing organisations (CDMOs) are accelerating investments to meet soaring demand for highly potent active pharmaceutical ingredients (HPAPIs) and antibody‑drug conjugates (ADCs). The focus is on backward integration, high‑containment infrastructure, and advanced processing such as chromatography and lyophilisation...

Leipzig’s Primogene Raises €4.1 Million to Scale Enzymatic Biomanufacturing of Complex Bioactive Molecules
Leipzig‑based biotech Primogene announced a €4.1 million (≈$4.5 million) seed round led by High‑Tech Gründerfonds to expand its enzymatic biomanufacturing platform. The funding will be used to scale production, grow the team, and broaden its IP portfolio and strategic partnerships. Primogene’s enzyme‑driven...

Arcera and Fosun Sign MoU for Neuroscience Innovation
Arcera Life Sciences and Fosun Pharma have signed a memorandum of understanding to create a long‑term strategic partnership focused on licensing, technology sharing, and neuroscience innovation. The deal taps Fosun’s research and manufacturing capabilities and Arcera’s access to international markets,...

How a Supreme Court Fight over Fish Oil Could Raise Your Prescription Drug Costs
U.S. Supreme Court is hearing Hikma Pharmaceuticals’ challenge to Amarin’s patent enforcement over Vascepa, a fish‑oil drug. The dispute centers on “skinny labeling,” which lets generics launch for unpatented uses while brand patents remain. A decision favoring Amarin could tighten...

Massive Bio Partners with OpenAI to Broaden Clinical Trial Access
Massive Bio has teamed up with OpenAI under the Impact Hours programme to automate clinical‑trial eligibility screening. The AI engine translates complex sponsor criteria into machine‑readable parameters, enabling real‑time, automated patient pre‑screening for oncology and haematology studies. The partnership includes...

Rocket to Sell PRV for $180m to Advance Gene Therapy Pipeline
Rocket Pharmaceuticals has agreed to sell its rare‑pediatric disease priority review voucher for $180 million after the FDA granted accelerated approval for its Kresladi gene therapy. The voucher, issued for addressing a rare paediatric condition, can be used to speed up...

CAR-T Cell Therapies Going in Vivo
Ex‑vivo CAR‑T therapies have saved tens of thousands of patients, but the industry is now pivoting to in‑vivo approaches that can be administered off‑the‑shelf. Over the past year, major pharma players have poured billions into in‑vivo CAR‑T platforms, highlighted by...
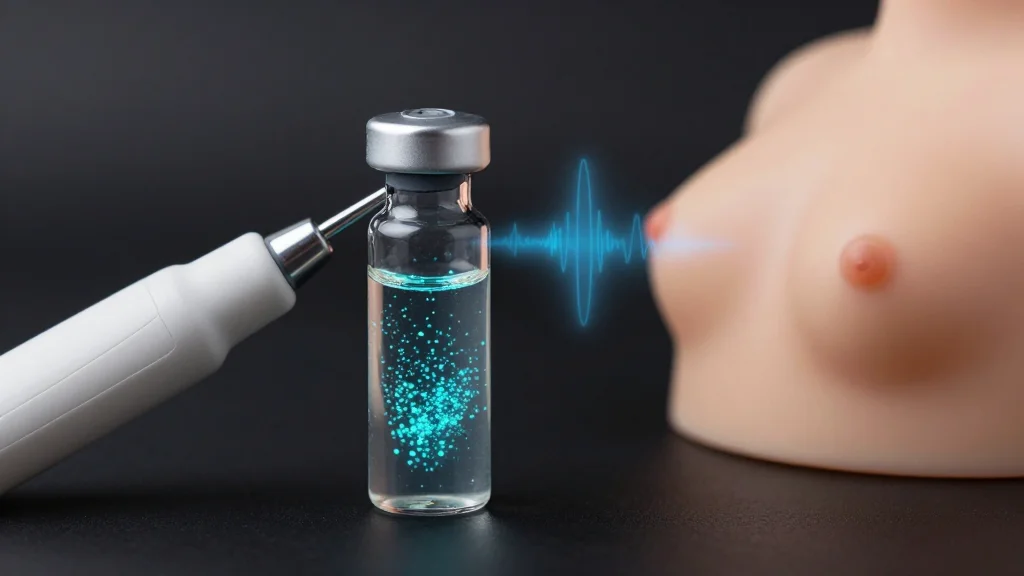
Tumor/Lymph Node Dual‐Targeting Ultrasonic Nanoconverter Orchestrates Spatiotemporal ROS Regulation for Dual‐Zone Programmed Sono‐STING Immunotherapy
Researchers have engineered a dual‑targeting ultrasonic nanoconverter (OPD@PSF) that co‑delivers the sonosensitizer protoporphyrin IX and the STING agonist Vadimezan to breast tumors and their draining lymph nodes. High‑power ultrasound at the tumor site generates abundant reactive oxygen species, inducing immunogenic...

DOJ Reclassifies Medical Marijuana to Schedule III: Key Implications for Florida and National Cannabis Operators
The U.S. Department of Justice has reclassified state‑licensed medical marijuana from Schedule I to Schedule III, the most significant federal cannabis reform in five decades. The change applies only to FDA‑approved cannabis products and state‑run medical programs, leaving adult‑use and illicit markets...