
How Saudi Arabia Is Developing as a Middle East Hub for Clinical Trials
Saudi Arabia is positioning itself as the Middle East’s primary hub for clinical trials, backed by robust state investment and the Vision 2030 Health Sector Transformation Programme. Healthcare spending is forecast to reach $61.05 million in 2026, while pharmaceutical sales are expected to climb to $17.23 billion, reflecting a 6.07% growth rate. The launch of the SEHA Virtual Hospital and expanding private‑public partnerships are enhancing patient access, data capture, and site readiness. Logistics specialist Oximio has opened the kingdom’s first GDP‑compliant facility, addressing supply‑chain and temperature‑control challenges for multicentre studies.
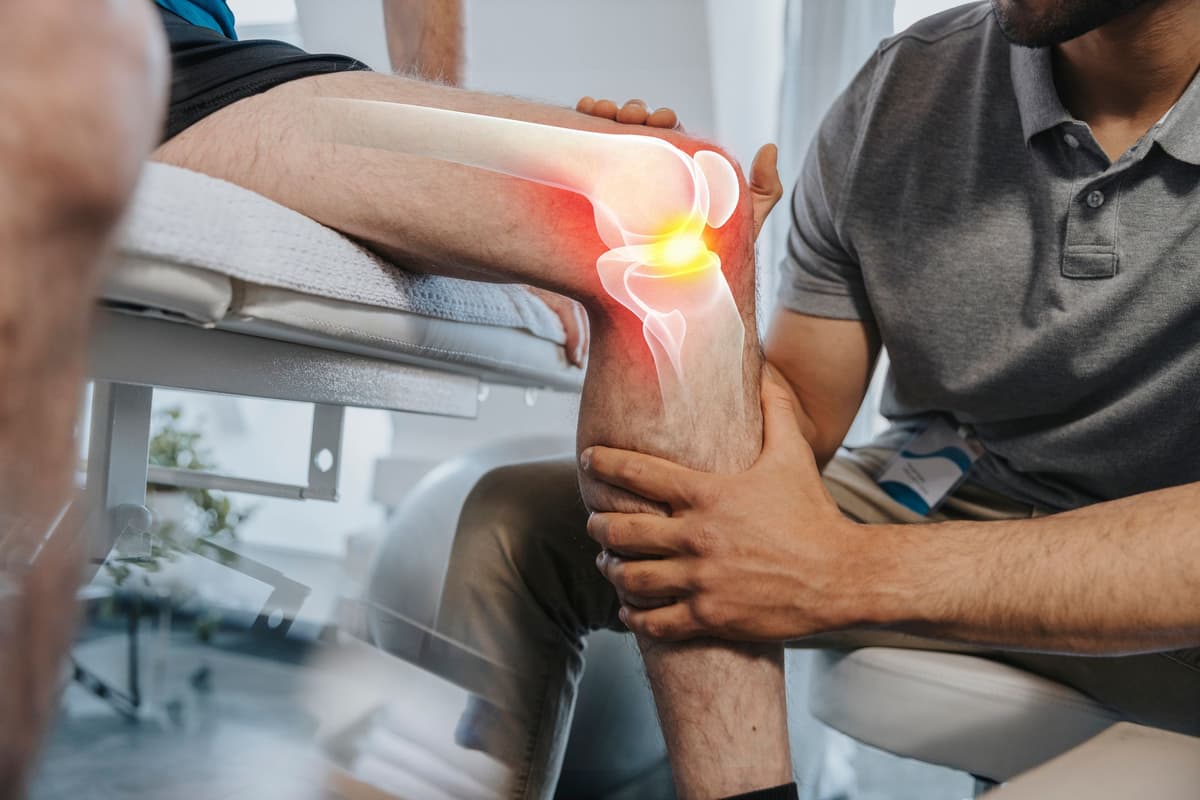
This Treatment Could Reverse Osteoarthritis Joint Damage With a Single Injection
Researchers at the University of Colorado Boulder have secured a $33.5 million ARPA‑H grant to develop a regenerative osteoarthritis therapy that could reverse joint damage with a single injection. The approach uses a controlled‑release particle system to deliver an approved drug...

BMS Brings the Buzz Around Late-Stage Readouts Amid a So-So Q1
Bristol Myers Squibb’s Q1 earnings showed modest 1% revenue growth to $11.5 bn, beating forecasts, while its growth portfolio surged 9% to $6.2 bn. The company highlighted late‑stage readouts for milvexian, a next‑generation anticoagulant, and Cobenfy, a schizophrenia therapy, as potential blockbuster...

Solvent‐Free Preparation of Pharmaceutically Active Diindolylmethane Derivatives by Stable Triple‐Interpenetrated Heterometallic‐Organic Frameworks With Sc‐Pd/Pt Synergistic Sites
Researchers reported a one‑pot synthesis of triple‑interpenetrated heterometallic‑organic frameworks (HMOFs) containing Sc‑Pd or Sc‑Pt synergistic sites. The resulting ScPd‑INA, ScPt‑INA and ScPt‑MCA catalysts achieved up to 99% conversion in solvent‑free Friedel‑Crafts alkylation of diindolylmethane (DIM) precursors. Single‑crystal X‑ray diffraction confirmed...
STAT+: Her Daughter Mila Got a Bespoke Medicine. Now She’s Starting a New Biotech to Make More
Julia Vitarello, whose eight‑year‑old daughter Mila was treated with a tailor‑made gene therapy, announced she is launching a new biotech to scale individualized medicines. Her previous company, EveryONE Medicines, folded after FDA guidance on custom therapies proved insufficient for investors....
State Tele-Buprenorphine Prescribing Policies by Medical Professional Type
A RAND‑led legal mapping of all 50 states and D.C. reveals stark differences in Medicaid policies governing fully virtual buprenorphine prescribing for opioid use disorder. While 32 states permit physicians, nurse practitioners and physician assistants to prescribe via telemedicine, 12...

What Happens When AI Transforms a Specialized Field Overnight?
In 2020 DeepMind released AlphaFold2, an AI that predicts protein structures with laboratory‑grade accuracy, quickly generating models for over 200 million proteins—a 1,500‑fold jump from prior data. The breakthrough earned its creators a 2024 Nobel Prize in Chemistry and sparked a...

LEO Pharma to Acquire Replay for Rare Skin Disease Gene Therapy Platform
LEO Pharma has signed a definitive agreement to acquire gene‑therapy specialist Replay for an upfront cash payment of $50 million, with additional milestone fees and tiered single‑digit royalties. Replay brings a herpes simplex virus (HSV) platform that delivers large therapeutic genes...

Scientists Build Drug-Carrying DNA Robots to Target Diseases
Scientists have engineered microscopic DNA robots that can carry therapeutic payloads and seek out viruses, acting as nano‑surgeons within the bloodstream. By applying origami‑inspired rigid joints and flexible components, the robots achieve nanometer‑scale precision. Movement is programmed through DNA strand...
Beacon Biosignals Is Mapping the Brain During Sleep
Beacon Biosignals has launched an FDA‑cleared, lightweight EEG headband that records clinical‑grade brain activity while users sleep at home. The device’s machine‑learning platform extracts detailed sleep‑stage metrics and subtle architecture changes, supporting more than 40 global clinical trials for conditions...

Explosion in UK Demand for GLP-1s Risks NHS Capacity Crisis
The Food Foundation’s January survey estimates 8.25 million people in the UK have used or are considering GLP‑1 weight‑loss drugs, representing about 7% of the population. NHS targets to treat 220,000 patients over three years from June 2025 and 3.4 million over 12...
![[Comment] GLP-1 Therapies: An Emerging Approach for Alcohol Reduction?](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/f628ae5758874c46e9810dd10e2c007b.webp)
[Comment] GLP-1 Therapies: An Emerging Approach for Alcohol Reduction?
Alcohol use disorder (AUD) remains one of the world’s most prevalent yet undertreated conditions, with fewer than 2 % of affected Americans receiving an FDA‑approved medication. Recent randomized trials of once‑weekly GLP‑1 receptor agonists, especially semaglutide, have demonstrated statistically significant reductions...

Gentler Treatment Improves Survival in Children with Relapsed Leukemia
A UK‑wide trial (UKALL Rel2020) tested a gentler regimen for children and young adults with relapsed acute lymphoblastic leukemia, using reduced‑intensity chemotherapy followed by the targeted immunotherapy blinatumomab. The study enrolled 188 patients across 25 centres and achieved a 92%...

FDA, up Close and Personal: A Banker’s Recalibration
Former FDA senior advisor Peter Reikes argues that the agency’s perceived slowness stems from its core principles of independence, caution, and process discipline rather than dysfunction. He notes that despite recent leadership turnover and political pressure, a deep bench of...

The CRISPR Medicine That Cured a Child: How Yuri Milner’s Breakthrough Prize Brought Gene Therapy to Hollywood
At the 2026 Breakthrough Prize ceremony, Baby KJ was celebrated after receiving Casgevy, the first FDA‑approved CRISPR gene‑editing therapy for sickle cell disease and beta‑thalassemia. The $3 million prize honored researchers Stuart Orkin, Swee Lay Thein and others whose work on the BCL11A...

GLP-1s Reduce Heavy Drinking Days in Patients With Obesity, Alcohol Use Disorder
A randomized, double‑blind trial in Copenhagen found that once‑weekly semaglutide 2.4 mg significantly reduced heavy‑drinking days in patients with alcohol use disorder and obesity. Over 26 weeks, the semaglutide group saw a 41.1‑percentage‑point drop in heavy‑drinking days versus 26.4 points for...
Single-Vesicle Profiling Could Push Liquid Biopsies Toward Everyday Clinical Use
Researchers from Incheon National University and the University of Pennsylvania reviewed cutting‑edge single‑extracellular vesicle (EV) profiling technologies that isolate and analyze vesicles one at a time. The review, published in TrAC Trends in Analytical Chemistry, highlights substrate‑based, droplet‑based and solution‑based...

The BioPharm Brief: Metabolic Phase III Progress, HER2 Oncology Momentum, and US Manufacturing Expansion Drive Industry Scale
Zealand Pharma and Roche are moving petrelintide, an amylin analog, into Phase 3 trials for chronic weight management, with enrollment slated for the second half of 2026 after earlier studies showed double‑digit weight loss and tolerability comparable to placebo. The FDA...

HYTN Successfully Exports GMP-Manufactured Vape Cartridges
HYTN Innovations announced the export of 1,000 one‑gram live‑resin vape cartridges manufactured under Good Manufacturing Practices to Australia’s regulated medical cannabis market. The shipment represents the company’s first full commercial batch of finished, non‑sterile cannabis‑oil products, following a Health Canada‑approved...

Drug Trials Snapshots: BIMZELX
UCB’s BIMZELX, a dual IL‑17A/F monoclonal antibody, received FDA approval on Oct 17 2023 for adults with moderate‑to‑severe plaque psoriasis. The approval was based on two phase‑3 placebo‑controlled trials that enrolled 839 patients across 13 countries, with a demographic profile of 72%...

Drug Trials Snapshots: AGAMREE
Santhera Pharmaceuticals received FDA approval on October 26, 2023 for AGAMREE (vamorolone), a corticosteroid designed to treat Duchenne muscular dystrophy (DMD) in patients aged two years and older. The approval rests on a multinational, double‑blind, 24‑week trial of 121 male patients that...

Drug Trials Snapshot: TALVEY
TALVEY (talquetamab‑tgvs), a bispecific antibody from Janssen Biotech, received FDA approval on August 10 2023 for adult patients with relapsed or refractory multiple myeloma who have exhausted at least four prior therapies. The pivotal trial enrolled 339 participants across 47 sites, with...

Drug Trials Snapshots: OJJAARA
OJJAARA (momelotinib) received FDA approval on September 15 2023 for adults with myelofibrosis‑related anemia. The approval rests on two pivotal trials—MOMENTUM, which enrolled JAK‑inhibitor‑experienced patients, and SIMPLIFY‑1, which enrolled JAK‑naïve patients. In MOMENTUM, 25% of OJJAARA recipients achieved a ≥50% reduction in...

There Are Two GLP-1 Side Effects Your Doctor Doesn’t Know About, and They Can Affect Your Workouts
A new *Nature Health* study used AI to scan 400,000 Reddit posts, finding that roughly 70,000 users were taking GLP‑1 drugs such as Ozempic, Wegovy, or Zepbound. While nausea and fatigue remain the most common side effects, about 4% of...

RNA-Built Droplets Create Customizable Organelles Inside Living Cells
UCLA researchers have engineered programmable artificial organelles by assembling RNA nanostars into droplet‑like condensates inside living cells. The RNA sequences encode assembly instructions, allowing precise control over condensate size, composition, and subcellular location. Published in Nature Nanotechnology, the study demonstrates...

Drug Trials Snapshots: ELREXFIO
Pfizer’s ELREXFIO (elranatamab‑bcmm) received accelerated FDA approval on Aug 14 2023 for relapsed/refractory multiple myeloma after at least four prior therapies. The approval rests on a single‑arm trial of 187 patients, with 97 efficacy‑eligible participants showing a 57.7% objective response rate, including...

Active-Duty US Soldiers to Receive MDMA Therapy for PTSD Next Year
The Department of Defense has approved two MDMA‑assisted therapy trials for active‑duty service members, allocating $4.9 million to each of Walter Reed and an Emory‑UT Health partnership. A total of 186 soldiers with PTSD will receive up to three MDMA doses...

Drug Trials Snapshots: VEOPOZ
VEOPOZ (pozelimab‑bbfg) received FDA approval on August 18, 2023 to treat CD55‑deficient protein‑losing enteropathy (CHAPLE disease) in patients of all ages. The decision was based on a single‑arm trial of ten patients enrolled in Turkey, Thailand and the United States, with a...

Axsome Gets FDA Approval for First Non-Antipsychotic Drug to Treat Agitation Associated with Alzheimer’s Dementia
The FDA has expanded the indication for Axsome Therapeutics’ Auvelity, approving it to treat agitation associated with Alzheimer’s disease dementia. This marks the first non‑antipsychotic medication cleared for this distressing symptom. Auvelity, originally approved in 2022 for major depressive disorder,...
Axsome Hits Record High On Alzheimer's Agitation Approval
Axsome Therapeutics saw its stock surge to a record $207.75 after the FDA approved Auvelity for treating agitation in Alzheimer’s disease patients. The drug, already cleared for major depressive disorder, now gains a second indication, expanding its market reach. This...

FDA Proposal Would Exclude Bulk Compounding of Weight-Loss Drugs Semaglutide, Tirzepatide, and Liraglutide
The FDA announced a proposal to remove semaglutide, tirzepatide and liraglutide from the 503B bulk drug list, concluding there is no clinical need for outsourcing facilities to compound these GLP‑1 weight‑loss agents from bulk substances. Because FDA‑approved versions are already...

Drug Trials Snapshots: EXXUA
Fabre‑Kramer Pharmaceuticals received FDA approval for EXXUA (gepirone) on September 22, 2023 as a once‑daily oral antidepressant for adult major depressive disorder. The approval was based on two placebo‑controlled, flexible‑dose Phase III trials that enrolled 456 patients, with 442 forming the...

The Future of Biomanufacturing: Key Highlights From INTERPHEX 2026
INTERPHEX 2026 highlighted a rapid shift toward digital integration and smarter single‑use technologies in biomanufacturing. Panels emphasized automation, continuous processing, and real‑time monitoring as ways to boost scalability while tackling data‑driven control challenges. A second discussion focused on supply‑chain resilience,...

Drug Trials Snapshots: POMBILITI
The FDA approved POMBILITI (cipaglucosidase alfa‑atga) on September 28, 2023 for adult patients with late‑onset Pompe disease (LOPD) weighing ≥ 40 kg who are not responding to existing enzyme replacement therapy. Approval is based on a randomized, double‑blind trial of 123 participants across 61 sites...

ESOC 2026: What to Expect in Maastricht
The 12th European Stroke Organisation Conference (ESOC) convenes in Maastricht from May 6‑8, drawing roughly 4,200 delegates from 100 nations. Six scientific tracks will showcase the latest clinical trial data on stroke prevention, diagnosis, and treatment, including late‑breaking sessions on pre‑hospital...

FDA's Oncology Advisors Vote Against 'New Paradigm' In AstraZeneca Trial
The FDA’s oncology advisory committee voted 6‑3 that AstraZeneca’s late‑stage trial of camizestrant, an oral selective estrogen‑receptor degrader (SERD), did not demonstrate a clinically meaningful benefit. AstraZeneca had positioned camizestrant as a potential first‑in‑class oral therapy for hormone‑receptor‑positive metastatic breast...

Novartis Finalizes US Expansion with Seventh Facility to Strengthen End-to-End Drug Manufacturing
Novartas has completed its US expansion plan by adding a seventh facility in Morrisville, North Carolina, dedicated to API production for oral solid‑dose and RNA‑based therapies. The site is part of a $23 billion multi‑year investment to build an end‑to‑end domestic...
Avalyn Pharma Takes a Breath to Raise $300M in IPO Cash for Lung Drug Trials
Avalyn Pharma completed a $300 million IPO, pricing 16.6 million shares at $18 each and listing on Nasdaq under the ticker AVLN. The Boston‑based biotech will use the proceeds to advance inhalable versions of pirfenidone (AP01) and nintedanib (AP02), plus a combination...

This Gas Provides Rapid Relief For Major & Treatment-Resistant Depression (M)
Recent clinical investigations show that inhaled nitrous oxide can alleviate symptoms of major depressive disorder, including cases resistant to standard therapies, within hours of administration. Across multiple small‑scale trials, roughly 40% of participants achieved remission after a single session, with...

FDA Expedites Review of New HIV Treatment Combo
The FDA has granted priority review to Gilead Sciences’ new single‑tablet HIV regimen that combines bictegravir and lenacapavir. The application is backed by phase 3 ARTISTRY‑1 and ARTISTRY‑2 trials, which showed sustained viral suppression in adults already virologically controlled. If approved,...

Merck Still Sees ‘Compelling’ Outlook for Terns Leukemia Drug
Merck agreed to acquire Terns Pharmaceuticals for $6.7 billion, paying $53 per share, after updated trial data showed its TERN‑701 leukemia drug achieving a major molecular response (MMR) rate north of 50% at 24 weeks. The data suggested TERN‑701 could outperform...

Other News to Note for April 30, 2026
Chinese biotech companies have reached an inflection point, gaining leverage as co‑dealmakers in cross‑border partnerships, according to multinational executives. Roche announced the discovery of new TREM2 agonist compounds aimed at modulating microglial activity for neurodegenerative diseases. Plasticity launched an advanced...

The Bangui Operation: A Story of Blood, Science and Biomedical Exploitation
In the early 1990s the Pasteur Institute in Bangui ran a covert HIV‑vaccine trial that recruited roughly 3,000 Central African soldiers, extracting over 11,000 blood samples. The research was funded by French institutions and aimed to fast‑track vaccine development at...
STAT+: Five-Way Obesity Drug Is Super Effective — in Mice
Researchers have unveiled a preclinical obesity candidate that combines five distinct mechanisms, delivering dramatic weight loss in mouse models and outperforming current GLP‑1 therapies. The multi‑modal approach targets appetite, metabolism, gut hormones, energy expenditure, and adipose tissue remodeling, achieving up...

Swiss BioTech Startup ALP Bio Raises €1.9 Million to Advance Immune Organoid and AI Platform
Swiss startup ALP Bio announced a €1.9 million (≈$2.1 million) pre‑seed round led by 42CAP to accelerate its immune organoid‑AI platform for antibody development. The platform couples human tonsil organoid readouts with generative AI to predict and reduce immunogenicity risk earlier in...

CMS Extends GENEROUS Model Deadline for Pharma and States
The Centers for Medicare & Medicaid Services has pushed back the application deadline for the GENEROUS Model, a program that lets state Medicaid programs buy drugs at prices comparable to those in other nations. Manufacturers now have until June 11 to...

Zealand Pharma and Roche Advance Petrelintide to Phase 3 for Chronic Weight Management
Zealand Pharma and Roche are moving the amylin analog petrelintide into Phase 3 trials to treat chronic overweight and obesity. The drug achieved up to 10.7% mean weight loss in the Phase 2 ZUPREME‑1 study, with tolerability comparable to placebo. A March 2025...

FDA Proposes Excluding Novo, Lilly Weight Loss Drugs From Bulk Compounding List in Win for the Companies
The FDA has proposed removing the active ingredients of Novo Nordisk’s semaglutide and liraglutide and Eli Lilly’s tirzepatide from the bulk‑compounding list used by 503B outsourcing facilities. If the rule is finalized, these high‑demand obesity and diabetes drugs could only be...

GLP-1s May Prevent Incident AF, Series of Studies Shows
Observational analyses presented at Heart Rhythm 2026 suggest GLP‑1 receptor agonists, including tirzepatide, lower the risk of incident atrial fibrillation by 33%‑47% across diverse patient groups. The benefit appears consistent in individuals with or without diabetes, obesity, and chronic kidney...

Why Protocol Complexity Keeps Increasing – and How to Fix It
Protocol complexity in late‑stage trials is spiraling, with Phase III studies now averaging about 6 million data points—a rise of roughly 11% each year. The growth is fueled by scientific advances, global regulatory demands, and the ease of capturing extra data via...