
STAT+: Pharmalittle: We’re Reading About a Supreme Court Hearing on ‘Skinny Labels,’ AstraZeneca U.K. Expansion, and More
AstraZeneca announced a $400 million investment to complete the Rosalind Franklin building in Cambridge and launch a new digital‑focused laboratory in Macclesfield, reviving UK R&D expansion after the US‑UK trade deal lifted NHS cost‑effectiveness thresholds. Meanwhile, the U.S. Supreme Court heard arguments on “skinny labeling,” a generic‑drug tactic that limits approved indications to sidestep patent infringement, featuring Amarin’s Vascepa and generic maker Hikma. The justices appeared reluctant to change the legal standard. Together, these moves signal renewed confidence in UK pharma investment and potential constraints on generic market entry in the United States.

Five Companies Pushing Biotech in Scotland
Scotland’s life‑science sector now comprises roughly 750 companies and 46,000 employees, anchored by strong university spin‑outs in Edinburgh, Glasgow and Dundee. Within this ecosystem five biotech firms are drawing attention: Cumulus Oncology’s portfolio‑driven oncology asset creation, EnteroBiotix’s microbiome‑based IBS therapy...

BeOne Receives the US FDA Priority Review to Tevimbra Combination for 1L Treatment of HER2+ GEA Cancers
The U.S. FDA granted priority review to BeOne Medicines’ Tevimbra‑Ziihera‑chemotherapy regimen for first‑line treatment of unresectable HER2‑positive gastric, gastro‑esophageal junction and esophageal adenocarcinomas. The decision follows the Phase III HERIZON‑GEA‑01 trial, which enrolled 914 patients and compared the new combos against...

Canada First in G7 to Approve Generic Ozempic
Health Canada has granted approval for the first generic version of semaglutide, the active ingredient in Ozempic, making Canada the first G7 nation to do so. The generic injection, submitted by Dr. Reddy’s Laboratories, is indicated for once‑weekly treatment of adult...

Specialty Drugs Now Consume over Half of Total Drug Spend
Specialty drugs now represent more than half of total prescription spending, despite treating a relatively small patient pool. A 2024 Pharmaceutical Strategies Group survey shows 43% of employers and health‑plan leaders cite managing specialty drug costs as their top priority,...
RAAS Inhibitors Show Benefits in Pediatric CKD Vs. CCBs
A new comparative‑effectiveness study of 2,762 children with chronic kidney disease (CKD) found that renin‑angiotensin‑aldosterone system (RAAS) inhibitors significantly outperformed calcium‑channel blockers (CCBs) in preventing progression to dialysis or transplant. Over a two‑year follow‑up, RAAS users had a 42% lower...

NDMA: A Cancer-Causing Contaminant in Meds and Meat
The FDA has identified the probable carcinogen N‑nitrosodimethylamine (NDMA) in several high‑volume drugs—including valsartan, Zantac (ranitidine) and certain metformin formulations—prompting recalls and market withdrawals. NDMA exposure limits are set at 96 nanograms per day, yet a half‑chicken breast cooked by...
Doubts Cast over 'Wild' Claim that Magnetic Control Can Turn on Genes
Researchers in South Korea announced a magnetically controlled switch that can turn on genes inside cells using an electromagnetic signal, a development touted as a potential breakthrough for non‑invasive therapies. The study appeared in a leading journal but has been...

Trump Administration Move to Reclassify Cannabis Sparks Confusion
The Trump administration issued an order that partially reschedules cannabis, moving products sold under state medical licenses and prospective FDA‑approved cannabis drugs from Schedule I to Schedule III. The change does not affect existing FDA‑approved cannabis pharmaceuticals and leaves the broader recreational...

Symptom Progression Slowed in Lewy Body Dementia with Zervimesine
Cognition Therapeutics reported that the oral, brain‑penetrant small‑molecule zervimesine slowed symptom progression in a phase 2 SHIMMER trial of 130 adults with mild‑to‑moderate dementia with Lewy bodies (DLB). Over six months, patients receiving 100 mg or 300 mg daily showed 52‑86% slower decline...

Laguna Biotherapeutics Reports the US FDA IND Clearance for LGNA-100 to Treat High-Risk Pediatric Leukemias
Laguna Biotherapeutics announced that the U.S. Food and Drug Administration has granted IND clearance for its lead candidate LGNA-100, allowing the company to begin a Phase‑I single ascending dose trial. The study will evaluate safety, tolerability and mechanism of action...

Merck Beats Quarterly Sales Expectations as Keytruda Fuels Growth
Merck reported first‑quarter sales of $8 billion, driven by a 12% increase in Keytruda revenue, surpassing analyst estimates of $7.6 billion. The company posted a loss after taking a $3.62‑per‑share charge related to its acquisition of antiviral developer Cidara Therapeutics. Shares jumped...

Huahui Health and BeOne Medicines Partner for HH160 in a ~$2.02B Deal
Huahui Health and BeOne Medicines have signed a global exclusive option, license and collaboration agreement for HH160, a novel trispecific antibody targeting oncology immunotherapy. Huahui will receive $20 million upfront, $100 million when BeOne exercises its option, and up to $1.9 billion in...

Real World Applicability of Ivermectin vs Permethrin Trial for Scabies
A recent cluster‑randomised trial found oral ivermectin more effective than 5% permethrin cream for treating classic scabies under controlled conditions. The study, however, enrolled participants from well‑resourced health centres and excluded severe dermatological cases, raising questions about its relevance to...

EU’s Joint Clinical Assessment System Still Finding Its Footing
The European Union’s Joint Clinical Assessment (JCA) system, launched in January 2025, saw its first activation with Ipsen’s Ojemda approval, initiating a 30‑day countdown for a centralized clinical report. Designed to replace duplicated health‑technology assessments across member states, the JCA currently...
ADM Research Highlights Microbial Benefits for Psychological Symptoms
A randomized, double‑blind trial examined live and heat‑inactivated probiotic strains in healthy adults with self‑reported mild anxiety. The 12‑week live blend of Bifidobacterium longum CECT 7347 and Lactobacillus rhamnosus CECT 8361 failed to meet primary anxiety endpoints, though it preserved butyrate‑producing bacteria....

Givaudan Research: Zensera Lemon Balm Supports the Mind During Stress
Givaudan’s patented Zensera lemon‑balm extract (300 mg) was tested in a double‑blind, placebo‑controlled trial with 130 healthy adults under moderate stress. The study measured mood, heart rate, blood pressure and a battery of executive‑function tasks over five hours. Participants who took...

Buried in Soil, a 100-Million-Year-Old Bacterial Toxin Could Reshape Pest Control and Antibiotic Discovery
Researchers from McMaster, Harvard, Yale and European partners have identified a new class of insect‑killing proteins, SAIPs, produced by rare Streptomyces strains. These toxins, structurally distant from diphtheria toxin, target an insect‑specific surface protein called Flower, leaving humans unharmed. The...

Precision Begins Patient Enrolment for FUNCTION-DMD Trial
Precision BioSciences has activated Arkansas Children’s Hospital as the inaugural site for its Phase I/II FUNCTION‑DMD trial, enrolling boys aged two to seven with Duchenne muscular dystrophy. The study tests PBGENE‑DMD, a first‑in‑class in‑vivo gene‑editing therapy that removes exons 45‑55 to restore...

How Epic Bio Is Leveraging CRISPR without Cutting DNA
Epic Bio, founded by Stanford professor Stanley Qi, is developing an epigenetic editing platform called GEMS that uses the smallest known Cas protein to modulate gene expression without cutting DNA. The system can be delivered in a single viral vector...

Re: Efficacy and Safety of VPM1002 and Immuvac in Preventing Tuberculosis: Phase 3 Randomised Clinical Trial (PreVenTB Trial)
The phase‑3 PreVenTB trial found that neither VPM1002 nor Immuvac reduced microbiologically confirmed tuberculosis, missing its primary efficacy endpoint. The authors of a BMJ rapid response highlight that the headline claim of 50% efficacy against extrapulmonary TB rests on only...

Henlius and Organon Receive the EC Approval for Poherdy (Biosimilar, Perjeta)
The European Commission has granted approval for Poherdy, a 420 mg/14 mL interchangeable biosimilar of pertuzumab (Perjeta), covering all approved indications. The decision rests on extensive analytical, pharmacokinetic, efficacy, safety and immunogenicity data that demonstrate equivalence to the reference product. Henlius has...

Defect-Engineered Pt/Nb2O5 Boosts Radical-Driven Benzimidazole Production and Hydrogen Evolution Efficiency
Researchers have created a defect‑engineered Pt/Nb₂O₅ catalyst with abundant oxygen vacancies and platinum nanoparticles that dramatically improves photocatalytic benzimidazole synthesis and concurrent hydrogen evolution. The system delivers 4.0 mmol g⁻¹ h⁻¹ production of 2‑methylbenzimidazole and 10.2 mmol g⁻¹ h⁻¹ hydrogen under mild light, surpassing prior benchmarks....

Follow the Money: Autoimmune, Inflammatory Disease Therapies, Antibody-Drug Conjugates, AKT1-Selective Inhibitor Programs
A wave of sizable financing rounds swept the biotech sector in late April 2026, with more than $1.3 billion pledged across 15 companies. Beeline Medicines led the pack with a $300 million Series A to advance lupus‑focused small‑molecule and protein therapeutics, while Sidewinder...
Single-Cell Sequencing Reveals Why some CAR-T Therapies Succeed While Others Fail
Researchers reviewed 44 single‑cell RNA sequencing studies covering about 500 patients to pinpoint cellular traits linked to CAR‑T therapy outcomes. The analysis identified exhaustion marker expression, low memory‑like cell fractions, and limited clonal diversity as hallmarks of relapse, while persistent,...

Tackling Drug Resistance Must Become Biotech’s Next Frontier
Drug resistance underlies roughly 90% of the 600,000 cancer deaths in the United States each year, limiting the durability of modern therapies. Kairos Pharma, founded in 2013, is focusing on the biology of resistance with its candidate ENV‑105, which aims...

How Multi-Omics Is Changing What Scientists Can See in the Human Immune System
Multi-omics technologies are reshaping human systems immunology by delivering high‑dimensional, single‑cell and spatial data that capture the full complexity of immune responses. Researchers now integrate scRNA‑seq, scATAC‑seq, CITE‑seq and spatial transcriptomics with large public atlases to identify molecular signatures predictive...

Morning Brief Podcast: Sun–Organon: The Scope, Risks, and Future of India's Biggest Pharma Deal
Sun Pharma announced an all‑cash $11.75 billion acquisition of U.S.‑based Organon, the largest overseas purchase by an Indian firm since 2007. The deal doubles Sun Pharma’s revenue to about $12.4 billion, propelling it into the top‑25 global pharmaceutical companies. It instantly gives...

New Copper Nanozyme Shows Powerful Tumor Suppression with High Precision
Researchers at the Chinese Academy of Sciences have created a coordinatively unsaturated copper single‑atom nanozyme (Cu‑N₂‑CDs) that exhibits markedly higher catalytic activity than traditional Cu‑N₄ nanozymes. The unsaturated Cu‑N₂ sites boost H₂O₂ adsorption by 3.49 times and generate hydroxyl radicals 3.62 times...

Dr. Reddy’s Announces Health Canada Approval of Generic Semaglutide Injection
Dr. Reddy’s Laboratories received a Notice of Compliance from Health Canada, becoming the first firm to secure market authorization for a generic semaglutide injection in Canada. The approval covers both 2 mg and 4 mg pen formulations, and the company is preparing...
New Genome Editing Method Could Swap Entire Genes and Correct 1000 Mutations at Once
Scientists have unveiled a new genome‑editing platform called prime assembly that can insert DNA segments up to 11,000 base pairs, enabling the replacement of entire genes rather than single‑point edits. The method uses overlapping flaps to attach donor DNA without...

Cartesian Therapeutics Inc (RNAC) Q1 2026 Earnings Call Transcript
Wave Life Sciences reported a Q1 net loss of $46.9 million, with cash reserves of $243.1 million expected to fund operations into 2027. The DMD candidate WVE‑N531 delivered a statistically significant 3.8‑second improvement in time‑to‑rise and 7.8% average dystrophin expression,...

Syndax Pharmaceuticals Inc (SNDX) Q1 2026 Earnings Call Transcript
Syndax Pharmaceuticals reported $172 million total revenue for 2025, driven by $124.8 million from its menin inhibitor RevuForge and $151.6 million from the chronic‑GVHD therapy Nictimvo. RevuForge posted 38% quarter‑over‑quarter revenue growth and reached roughly 50% penetration of the KMT2A AML population, while...

Alnylam Pharmaceuticals Inc (ALNY) Q1 2026 Earnings Call Transcript
Alnylam reported a strong Q1 2021 performance, with total product sales reaching $135.8 million, an 89% year‑over‑year increase. ONPATTRO generated $102 million in revenue, growing 13% quarter‑over‑quarter, while newly launched OXLUMO contributed $9 million and GIVLAARI $24.7 million. The company announced positive Phase 3 data...

Amgen Inc (AMGN) Q1 2026 Earnings Call Transcript
Amgen reported first‑quarter 2026 sales of $5.6 billion, a 4% year‑over‑year increase, driven by six high‑growth products that contributed 70% of total revenue. Repatha, Evenity, Test Buyer, TEPEZZA and the rare‑disease portfolio posted double‑digit growth, while legacy brands Prolia and XGEVA...
Kymera Therapeutics Inc (KYMR) Q1 2026 Earnings Call Transcript
Kymera Therapeutics reported Q1 2026 revenue of $2.8 million, entirely from its Gilead collaboration, and ended the quarter with $978.7 million in cash, extending its runway into the second half of 2028. The company completed enrollment and dosing for the Phase Ib atopic...
Angiocrine Signaling Drives Liver Fibrosis: From Mechanism to Early Clinical Translation
Researchers led by Hu et al. discovered that ROCK2 activity in liver sinusoidal endothelial cells is a pivotal driver of liver fibrosis. Single‑cell transcriptomics and knockout models revealed that ROCK2‑mediated cytoskeletal remodeling releases angiocrine factors that activate hepatic stellate cells. Early...
Long-Lived Immune Cells Show Promise Against Cancer in World-First Trial
A world‑first clinical trial tested CAR‑T therapy enriched with stem‑cell memory T cells, a long‑lived immune subset. In a small cohort of 11 patients with refractory blood cancers, five achieved complete remission and one partial remission, outperforming historical outcomes of...
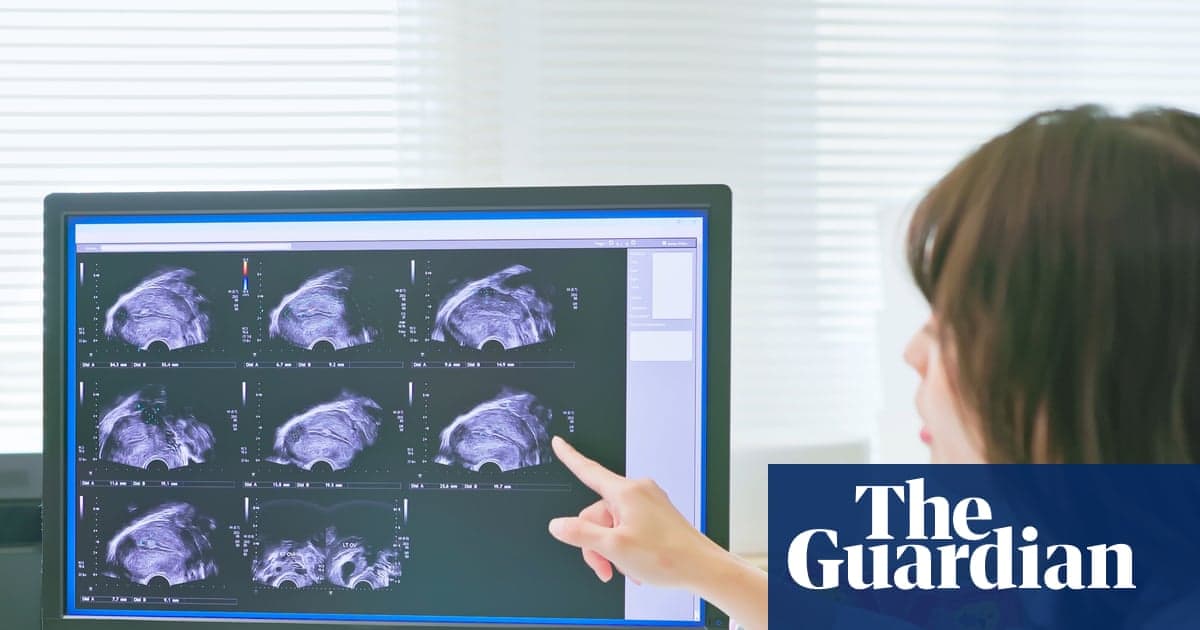
Trial of Non-Invasive Endometriosis Scan Boosts Hopes for Quicker Diagnosis
A small trial of 19 women showed that the experimental radiotracer maraciclatide can illuminate endometriotic lesions on a Spect‑CT scan, matching surgical findings in 16 cases with no false positives. Current diagnosis in the UK often requires invasive laparoscopy, leading...

Tiny Biotech’s Experience Raises Questions About FDA’s Rare Disease Policies
BioCentury’s website now publishes a detailed cookie policy that separates cookies into five categories: strictly necessary, functional, marketing, advertising, and analytics. Strictly necessary cookies are always active and essential for authentication and navigation, while functional cookies enable personalization of the...

In Chiesi’s Biggest M&A Deal yet, Pharma Buys HAE Company KalVista
Italian pharmaceutical group Chiesi announced the acquisition of U.S. biotech KalVista for approximately $1.5 billion in cash, marking its largest M&A transaction to date. KalVista, a specialist in hereditary angioedema (HAE), brings a late‑stage monoclonal‑antibody program and a pipeline of complement‑targeted...

The Trump Administration Is Shifting Federal Policy On Cannabis And Psychedelics
The Trump administration announced that marijuana will be re‑classified from Schedule I to Schedule III in states that have authorized medical use, and the Justice Department scheduled a June hearing on a nationwide re‑classification. The same administration issued executive orders to increase...
BIOTECanada Responds to Health Canada’s Gazette on Modernizing Clinical Trials Regulations
Health Canada has released a Gazette notice proposing modernized clinical‑trial regulations and draft guidance for decentralized trials. BIOTECanada welcomed the initiative but urged that the new rules align with the U.S. FDA and European EMA to avoid duplicative requirements. The...
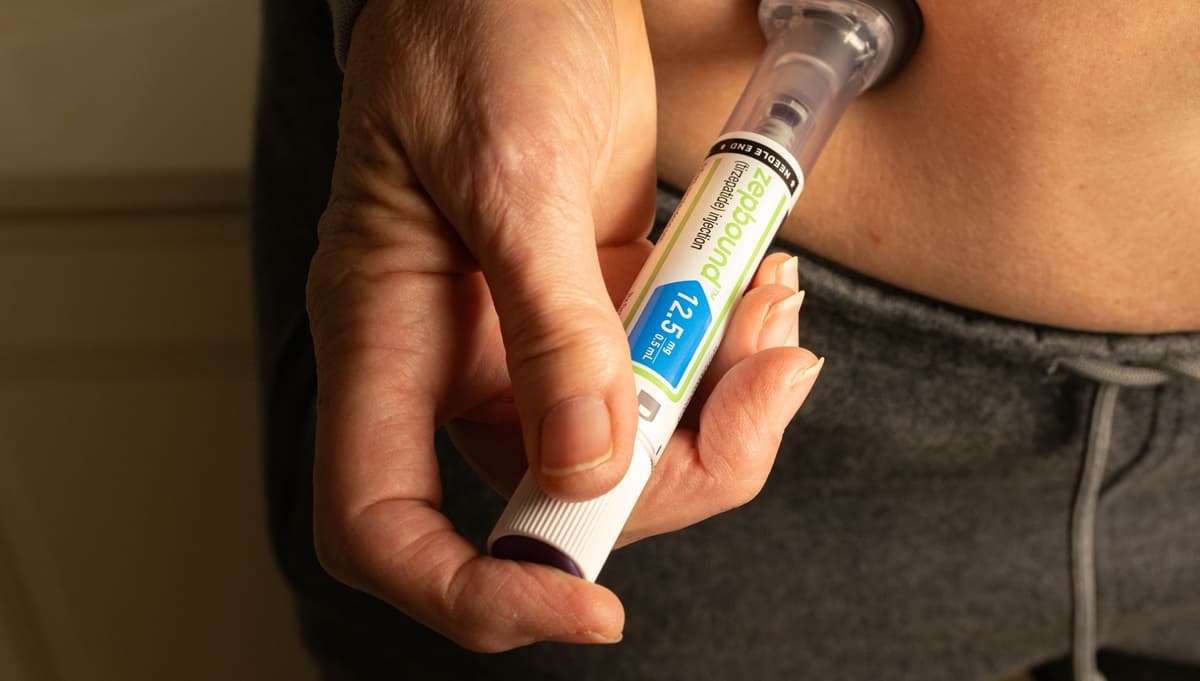
GLP-1 Drugs May Lower CV Risk in TAVI Patients With Diabetes or Obesity
A retrospective analysis of 1,708 matched TAVI patients shows that glucagon‑like peptide‑1 (GLP‑1) receptor agonists cut the relative risk of major adverse cardiovascular events (MACE) by 37% and all‑cause mortality by 39% at one year. The benefit was consistent in...

Payment Models Taking Shape for Advanced Therapies: Fran Gregory, PharmD
Advanced therapies priced up to $4 million are prompting a rethink of how they are funded. Fran Gregory of Cardinal Health says the gap between limited launch data and payer expectations can be bridged with robust real‑world evidence. The company is...

Olema Oncology Appoints Prakash Raman, Ph.D., to Board of Directors
Olema Oncology announced the appointment of Dr. Prakash Raman, a veteran biotech executive, to its Board of Directors. Raman, currently CEO of InduPro Therapeutics, brings more than two decades of experience in business development and corporate strategy, including senior roles...

Rockwell Automation and Cytiva Launch Platform to Accelerate Digital Transformation for Biopharma Companies
Rockwell Automation and Cytiva have launched Figurate, a supervisory control and data acquisition (SCADA) platform aimed at streamlining digital integration in biopharmaceutical manufacturing. The system combines Cytiva’s bioprocessing hardware with Rockwell’s FactoryTalk software, offering an open‑architecture layer that connects third‑party...

AI Drug Target Platform Pairs Prediction with Benchmarking to Improve Early Discovery
Insilico Medicine unveiled an integrated AI framework that couples its Target Identification Pro (TargetPro) predictive engine with the TargetBench 1.0 benchmarking suite to improve early‑stage drug target discovery. The system uses disease‑specific models trained on 22 omics and text scores,...

Early Data Links Wegovy to Risk of 'Eye Stroke' — Here's What to Know
Early signals from a British Journal of Ophthalmology analysis suggest Wegovy, the semaglutide‑based weight‑loss injection, may be linked to ischemic optic neuropathy (ION), a rare form of eye stroke that can cause rapid vision loss. The study examined 31,774 FDA...

AbbVie Faces Questions About Skyrizi Competition From J&J
AbbVie’s first‑quarter earnings call highlighted growing pressure on its immunology franchise as analysts probed the company’s defense against Johnson & Johnson’s upcoming IL‑23 inhibitor. Skyrizi, AbbVie’s flagship psoriasis drug, posted $1.2 billion in Q1 sales, a 12% year‑over‑year increase, but faces...