
The Company that Built TikTok’s Algorithm Is Now Designing Drugs for Diseases Pharma Called Undruggable
ByteDance’s Anew Labs showcased its first AI‑designed therapy, a small‑molecule inhibitor of IL‑17, at the American Association of Immunologists meeting in Boston. The molecule targets a protein‑protein interaction long deemed undruggable, suggesting generative AI can breach a major pharmaceutical barrier. Anew Labs also released AnewOmni, a framework trained on more than five million biomolecular complexes that claims to design functional molecules across all scales, from chemicals to nanobodies. The move places ByteDance alongside Isomorphic Labs, Anthropic and Insilico Medicine in the fast‑growing AI drug‑discovery race.
Precise Spatiotemporal Cardiac Repair and Regeneration
Researchers are advancing spatiotemporal drug delivery systems (DDS) that synchronize therapeutic release with the heart’s natural healing stages after myocardial infarction. These platforms integrate bioelectrical scaffolds, programmable degradation, and cell‑free vesicle carriers to provide phase‑specific immunomodulation, angiogenesis, and antifibrotic treatment....

FDA Approves Once-Daily Idvynso Tablet for Treating HIV
The FDA has approved Merck’s Idvynso, a once‑daily, two‑drug tablet combining doravirine and islatravir, for HIV‑1‑infected adults who are virologically suppressed. The regimen replaces existing antiretroviral therapy and is tenofovir‑free, targeting patients without prior treatment failure. Approval rests on two...
Federal Appeals Court Temporarily Halts Abortion Pill Access by Mail
A federal appeals court in the Fifth Circuit issued a temporary stay that blocks abortion providers from prescribing mifepristone via telemedicine and mailing the pills to patients. The ruling follows a lawsuit by Louisiana seeking to enforce its near‑total abortion...
Innovative Nanoparticle Technique Advances Early Detection of Pancreatic Cancer
Researchers have unveiled a novel nanoparticle‑based imaging agent that markedly improves early detection of pancreatic cancer. In pre‑clinical trials the probe identified tumors as small as 2 mm, delivering a 30% sensitivity boost over conventional CT scans. The technology earned FDA...

The Biopharm Brief: Microbiome Fast Track, Leo Pharma Gene Therapy Deal, AAV Automation Advances
The FDA has awarded Fast Track designation to a live biotherapeutic aimed at restoring gut microbiota in moderate‑to‑severe ulcerative colitis, accelerating its development path. Leo Pharma is buying Replay’s gene‑therapy program to broaden its rare‑dermatology portfolio, marking a shift toward...

Artificial Intelligence for Drug Development
The FDA’s Center for Drug Evaluation and Research (CDER) is formalizing its AI strategy through a draft 2025 guidance, an AI Council created in 2024, and extensive stakeholder engagement. CDER has reviewed more than 500 AI‑enabled drug submissions since 2016...

External Engagements with FDA for Artificial Intelligence in Drug Development
The FDA has published a comprehensive menu of early‑engagement options for sponsors leveraging artificial intelligence across the drug development lifecycle. Sponsors can request formal meetings for IND or pre‑IND programs, or use targeted programs such as C3TI for trial design,...

Regulatory and IP Considerations for Veterinary Drugs in Canada
The article outlines Canada’s regulatory framework for veterinary drugs, separating them from other animal health products and detailing the data‑protection, patent‑linkage, and supplementary‑protection regimes that apply only to veterinary drugs. It explains submission requirements, labeling rules, priority‑review criteria, and biowaiver...

Arvinas’ ‘Protac’ Breast Cancer Drug Cleared by FDA
The FDA approved Arvinas and Pfizer’s Veppanu, a PROTAC drug, as a second‑line therapy for metastatic ER‑positive, HER2‑negative breast cancer with ESR1 mutations. Veppanu is the first protein‑degrading medicine cleared in the U.S., targeting estrogen receptors for destruction. While the...

UniQure Pushes Forward As FDA Rare Disease Controversies Continue
UniQure, the gene‑therapy pioneer at the heart of FDA Commissioner Marty Makary’s rare‑disease controversy, is pressing forward with a formal request for its Huntington’s disease treatment. The company will present data at a Type B meeting with the FDA in the...
RFK Jr. Wants Stakeholders To Show Real-Time Prices Ahead Of Final Regulation
HHS Secretary Robert F. Kennedy Jr. is urging health‑care providers and health‑IT firms to deploy Real‑Time Pharmacy Benefit Technology (RTPBT) immediately, rather than waiting for formal rulemaking. RTPBT enables patients to see the exact out‑of‑pocket cost of prescription drugs at...
DNA-Containing Extracellular Vesicles Boost Antitumor Responses in Mice
Researchers at Weill Cornell Medicine discovered that extracellular vesicles released by activated T cells contain DNA that can be transferred to dendritic and tumor cells, enhancing antigen processing and presentation. In mouse models of glioblastoma, pancreatic and triple‑negative breast cancer,...

Moderna in Talks with FDA over Phase 4 Covid Vaccine Data
Moderna is actively collaborating with the U.S. Food and Drug Administration to submit Phase 4 post‑marketing data on its COVID‑19 vaccines. The company hopes the additional safety and efficacy evidence will persuade regulators to broaden the current, narrowed product labels that...

AI, Gene Therapies Drive Market Trends in Eye Care
In 2026 the ophthalmology market is being reshaped by gene‑therapy breakthroughs and the emergence of agentic AI, according to Boston Consulting Group’s Long Sha. Gene‑based treatments are moving beyond rare retinal disorders into chronic conditions such as wet age‑related macular...
Common Cholesterol Medications Do Not Alter Long-Term Dementia Risk
A massive target‑trial emulation study of more than 320,000 older adults found that statin use does not change long‑term risk of dementia. While statin users showed a 46% spike in dementia diagnoses during the first year after initiation, researchers attribute...

Ensorcell Debuts VersaWeld Platform, Wins Best New Product at INTERPHEX 2026
Ensorcell introduced the VersaWeld sterile tube welding platform at INTERPHEX 2026, marking its entry into cell and gene therapy (CGT) manufacturing equipment. The system automates thermoplastic tubing welding in closed‑system environments, delivering consistent weld integrity across multiple tube sizes and materials...

Amgen Launches Late-Stage Obesity Trial in Patients Who Switch From Rival Drugs
Amgen is initiating three Phase III trials for its long‑acting obesity injection MariTide, including a pivotal study that enrolls about 1,200 patients switching from Eli Lilly’s semaglutide or Novo Nordisk’s tirzepatide. The primary goal is a minimum 10% body‑weight loss after 68 weeks,...

F.D.A. Grants Early Access to Promising Drug for Pancreatic Cancer
On May 1, the FDA granted expanded‑access permission for daraxonrasib, an experimental oral drug from Revolution Medicines, allowing patients with previously treated metastatic pancreatic cancer to obtain the therapy outside clinical trials. The drug, taken as three pills daily, has produced...

Regulatory Actions for May 1, 2026
BioWorld released a comprehensive “Regulatory Actions for May 1, 2026” roundup, collating FDA, EMA and other global regulator updates across biopharma, medical technology, and policy domains. The collection links to data snapshots, infographics on digital analysis, and special reports covering topics such...

FDA Permits Expanded Access for Investigational Pancreatic Cancer Drug
The FDA issued a “safe to proceed” letter to Revolution Medicines, enabling an expanded access protocol for its experimental pancreatic cancer drug daraxonrasib. The request, received on April 28 and signed on April 30, targets patients with previously treated metastatic pancreatic ductal...

Acelyrin Founder Shao-Lee Lin Emerges at Cue, as It Becomes Latest to License From China
Cue Biopharma announced a strategic reboot, appointing Acelyrin founder Shao‑Lee Lin to its executive team and unveiling a new allergy drug candidate sourced from research labs in Taiwan and mainland China. The Boston‑based company, previously focused on T‑cell immunotherapies, will...

Microbiome-Based Therapy Gains FDA Fast Track in Ulcerative Colitis
Belgium‑based MRM Health announced that its investigational microbiome‑based therapy MH002 has been granted FDA fast track designation for mild‑to‑moderate ulcerative colitis. MH002 is a live biotherapeutic composed of six defined commensal bacterial strains designed to restore gut microbial balance and...
Moderna Beats Revenue Expectations with $389M, but Litigation Dogs Earnings
Moderna reported first‑quarter revenue of $389 million, roughly 50% above analysts’ expectations, while vaccine sales reached $352 million, beating consensus. A $950 million upfront settlement with Arbutus over lipid‑nanoparticle patents pushed the net loss to $1.3 billion, though it was still better than the...

Policy Watch: FDA Looks to Expand Real-Time Drug Clinical Trials
The FDA has launched an AI‑driven pilot that streams clinical‑trial data in real time for AstraZeneca and Amgen’s lymphoma and lung‑cancer studies, and is now seeking public input to broaden the approach. The Ninth Circuit Court declined to compel the...
STAT+: Axsome Wins FDA Nod for Alzheimer’s Agitation
Axsome Therapeutics announced that the U.S. Food and Drug Administration has granted a regulatory nod for its investigational therapy aimed at treating agitation in Alzheimer’s disease. The agency’s decision clears the path for accelerated clinical development, potentially moving the drug...
Peptides Are Unproven as Health Aids. FDA May Unleash Them Anyway
The FDA is poised to broaden access to injectable peptides by allowing compounding pharmacies to produce them and by considering their inclusion in oral dietary supplements. Health and Human Services Secretary Robert F. Kennedy Jr. has publicly opposed what he...

Drug Trials Snapshot: TRYNGOLZA
TRYNGOLZA (olezarsen), an APOC‑III‑directed antisense oligonucleotide, received FDA approval on December 19, 2024 for adults with familial chylomicronemia syndrome (FCS). In the pivotal Phase 3 trial (NCT04568434), 66 patients from 11 countries received monthly 80 mg injections or placebo for 53 weeks. The drug achieved...

Drug Trials Snapshot: ALYFTREK
Vertex Pharmaceuticals received FDA approval for ALYFTREK, a triple‑combination CFTR modulator, on Dec. 20, 2024. The drug targets cystic fibrosis patients aged six and older with at least one F508del or other responsive mutation. Approval was based on two 52‑week, non‑inferiority trials...
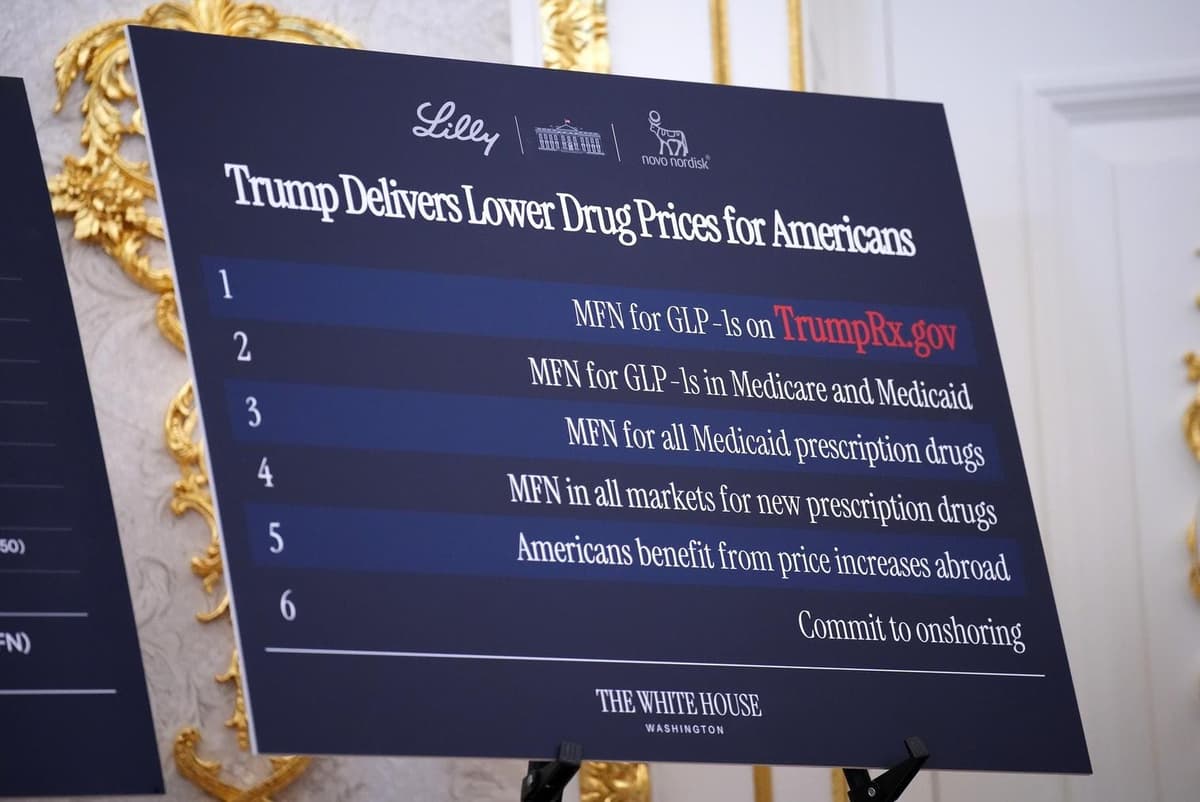
Direct-To-Consumer Drug Portals Offer Lower Prices, But Preserve Inequities
President Trump launched TrumpRx, a federal direct‑to‑consumer portal that sells dozens of prescription drugs at cash‑pay discounts. The program relies on confidential three‑year agreements with 17 manufacturers, using most‑favored‑nation pricing tied to nine wealthy nations. While the portal lowers out‑of‑pocket...

STAT+: Pharmalittle: We’re Reading About Obesity Drugs and a Compounding List, an AstraZeneca Setback, and More
The FDA proposed removing semaglutide and tirzepatide—the active ingredients in Novo Nordisk’s Wegovy/Ozempic and Eli Lilly’s Mounjaro/Zepbound—from the list of substances that compounding pharmacies may bulk‑manufacture, arguing there is no clinical need. This move effectively blocks large‑scale compounding of popular GLP‑1...

Drug Trials Snapshot: ROMVIMZA
Deciphera's ROMVIMZA (vimseltinib) received FDA approval on Feb 17 2025 for adult tenosynovial giant‑cell tumor (TGCT) when surgery is unsuitable. Approval was based on a global phase II trial of 123 patients, showing a 40% overall response rate versus 0% with placebo...

Summit Slips on Ivonescimab's Apparent Interim Miss in Sign of Investor Frustration
Summit Therapeutics’ shares plunged almost 20% after an interim analysis of its Phase 3 trial of ivonescimab showed a statistical shortfall in a key efficacy endpoint. The miss was limited to a subgroup analysis, but investors interpreted it as a signal...

Amgen Files Update to Tavneos Label as FDA Escalates Push to Withdraw
Amgen filed a supplemental application to revise the prescribing information for its rare‑disease drug Tavneos (avacopan) as the FDA intensifies its effort to withdraw the product. The label update seeks to add new safety warnings and monitoring requirements after the...

GoodRx Partners with Novo Nordisk to Offer Self-Pay Pricing for New Oral Ozempic® Pill
GoodRx has teamed with Novo Nordisk to sell the newly launched oral Ozempic® pill at transparent self‑pay rates, ranging from $149 to $299 per month depending on dosage. The tiered pricing covers the 1.5 mg, 4 mg and 9 mg strengths, letting patients...

Amgen Positions MariTide as Potential ‘Best Monthly’ Obesity Drug
Amgen is positioning its bispecific antibody‑peptide MariTide as the premier monthly or less‑frequent obesity treatment. The company launched a Phase 3 MARITIME‑Switch study to evaluate switching patients from weekly GLP‑1 injections to four‑to‑six monthly doses. Phase 2 data showed up to a...

Finland’s Algorithmiq Takes Top Spot in $50M Q4Bio Challenge with Quantum Drug Simulation Milestone
Finland’s Algorithmiq won the $2 million prize in Wellcome Leap’s $50 million Q4Bio challenge, becoming the first to demonstrate an end‑to‑end quantum‑classical workflow for drug simulation on real hardware. Using up to 100 qubits on IBM’s quantum computer, the team modeled the...

Theradaptive Receives US FDA Approval to Initiate P-III Trial of OsteoAdapt SP for Spinal Fusion Procedures
Theradaptive has secured U.S. FDA approval to launch the pivotal OASIS Phase III trial evaluating its OsteoAdapt SP device as a bone‑graft alternative in transforaminal, anterior, and lateral lumbar interbody fusion (TLIF, ALIF, LLIF) procedures. The trial follows earlier studies involving...
AstraZeneca’s Breast Cancer Drug Fails to Earn Backing of FDA Advisory Committee
The FDA’s advisory committee voted against recommending AstraZeneca’s oral SERD camizestrant for HR⁺/HER2‑ metastatic breast cancer patients with an ESR1 mutation, citing concerns over the Phase 3 SERENA‑6 trial design. The study switched patients to camizestrant at the point of mutation...

Lilly's Double-Beat Widens the GLP-1 Gap—And a New Pill Could Make It Permanent
Eli Lilly posted a strong Q1 2026, with revenue up 56% YoY to almost $20 billion, driven by $12.8 billion in GLP‑1 sales from Mounjaro and Zepbound. The FDA also approved Foundayo, the first oral GLP‑1 agonist for obesity that can be taken without...

BIO on the American Road Tours Gene Therapy Hub in Ohio
BIO President John F. Crowley toured Ohio on April 28, spotlighting the state’s emerging gene‑therapy hub. Researchers at Nationwide Children’s Hospital have delivered two of the FDA’s first eight approved gene therapies for Duchenne muscular dystrophy and spinal muscular atrophy type 1....
Corcept Ties ALS Drug to Improved 2-Year Survival as Phase 3 Start Date Nears
Corcept Therapeutics reported that its experimental ALS drug dazucorilant reduced the two‑year risk of death by roughly 87% in a Phase 2 extension study, despite missing its primary motor‑function endpoint. The survival benefit was most pronounced at the 300 mg dose, though...

MDMA-Assisted Therapy for Depression: A Promising but Early First Step
A small open‑label proof‑of‑principle study examined MDMA‑assisted therapy in 12 adults with moderate‑to‑severe major depressive disorder. Participants received two MDMA dosing sessions spaced a month apart together with nine psychotherapy sessions. At two months, 75% of participants achieved remission and...

Cue Biopharma Licenses Ascendant-221 From Ascendant Health Sciences in a ~$691.5M Deal
Cue Biopharma announced an exclusive license with Ascendant Health Sciences for Ascendant-221, an anti‑IgE monoclonal antibody aimed at allergic diseases. The agreement grants Cue worldwide rights, except Greater China, in exchange for a $15 million upfront payment and up to $676.5 million...

Approaches to Reducing Toxicity and Side Effects in Cell and Gene Therapy
Cell and gene therapies are expanding rapidly, with the market projected to exceed $9 billion in 2025 and grow over 15% annually through 2035. Safety remains a hurdle, prompting multiple strategies to curb cytokine release syndrome (CRS) and related toxicities. Companies...

FDA's ODAC Delivers One Loss, One Win for AstraZeneca
The FDA’s Oncologic Drugs Advisory Committee (ODAC) voted 6‑3 against AstraZeneca’s oral SERD camizestrant in combination with CDK‑4/6 inhibitors for first‑line HR‑positive, HER2‑negative breast cancer with ESR1 mutations, citing a lack of overall survival benefit despite a 56% progression‑free survival...
Gene Editing at Scale, Clinic Seeks Generalizable Therapies
Integrated DNA Technologies helped deliver a CRISPR therapy that rescued baby KJ Muldoon from a fatal urea‑cycle disorder, proving gene editing can correct a single disease‑causing mutation. The success highlights the field’s next hurdle: scaling personalized edits for disorders with...

Smarter AAVs Drive Gene Therapy’s Next Chapter
Gene therapy’s growth is hampered by AAV manufacturing bottlenecks, safety concerns, and high costs, prompting a wave of innovations across bioprocessing, analytics, and vector design. Companies like Thermo Fisher, PackGene, Catalent, and Asimov are deploying design‑space modeling, high‑throughput purification, and...

What’s Next in the Evolution of Standards for Biologics Development
The United States Pharmacopeia (USP) is redefining how documentary standards support biologics, moving from product‑specific monographs toward a hybrid model that blends platform‑based chapters, emerging standards, and analytical reference materials. This shift addresses the growing complexity of monoclonal antibodies, ADCs,...

The Cost of Inefficiencies in Clinical Trials
Clinical trials are hampered by fragmented technology, leading to roughly 45% higher costs compared with unified platforms. Integration fees, duplicate data entry, and managing multiple vendors add significant expense and delay timelines. Unified data‑management solutions such as CRScube claim 50‑60%...