Bio Korea 2026 Kicks Off with Spotlight on Oligonucleotides
In early May 2026, several biotech firms announced pivotal milestones. Axsome Therapeutics received FDA clearance for Auvelity (AXS‑05) to treat agitation in Alzheimer’s disease, a condition affecting up to 75% of patients. Sonire Therapeutics began its U.S. Sunrise II trial of Suizenji, a high‑intensity focused ultrasound (HIFU) platform, backed by an $18 million Series A round. Meanwhile, Summit Therapeutics moved its bispecific antibody ivonescimab into a Phase III study for non‑small cell lung cancer (NSCLC).

Second Life for Gene Therapy; Takeda Phase 2/3 Win; UK Cancer Biotech's $83M
A Cleveland‑based biotech announced a revamped gene‑therapy platform that could give a previously stalled program a second chance, while Takeda disclosed positive Phase 2/3 results for its oncology candidate. Across the Atlantic, a UK cancer‑focused biotech raised roughly $83 million to accelerate...
ADAPT OCULUS Trial Shows Promising Results in Treatment Efficacy for Ocular MG: Carolina Barnett-Tapia, MD, PhD
The ADAPT OCULUS Phase III trial evaluated efgartigimod alfa (VYVGART) in patients with ocular myasthenia gravis (oMG). In a double‑blind, placebo‑controlled arm, participants receiving the drug showed statistically significant reductions in ptosis and diplopia versus placebo. An open‑label extension confirmed continued...

Prolific Machines Sets Monoclonal Antibody (mAb) Manufacturing Record with Light-Controlled Platform
Prolific Machines, an SOSV portfolio company, announced a record 21 g/L monoclonal antibody titer in a 15‑day intensified fed‑batch CHO run using its light‑controlled optogenetic platform. The photomolecular system lets operators toggle gene expression in real time with light, offering reversible,...

FDA Flags eBay Listing over GLP-1 Claims in Rare Warning Letter
The FDA sent a rare warning letter to an eBay seller for making GLP‑1 and blood‑sugar support claims, marking only the fourth eBay‑related enforcement in recent years. Unlike typical cases, the agency did not purchase or test the product, relying...

Emcure Doubling Down on Biosimilars Pipeline: MD Satish Mehta
Emcure Pharmaceuticals is expanding its biosimilars pipeline as a wave of biologic patent expiries creates market tailwinds. The company currently sells six biosimilars through its Gennova Biopharmaceuticals subsidiary and will now seek external partners to bring additional products to India...

STAT+: Are Analysts Too Quick to Gloss over Lilly’s Liver Case?
Analysts are being criticized for downplaying a recent liver safety issue at Eli Lilly, raising concerns about market oversight. Meanwhile, biotech earnings showed mixed results, with Vertex shelving an mRNA cystic fibrosis candidate and Pfizer and Alkermes delivering near‑consensus Q1 numbers....

Viridian Reports Positive Phase III REVEAL-2 Data for Elegrobart in Chronic Thyroid Eye Disease
Viridian Therapeutics announced that its subcutaneous IGF‑1R antibody elegrobart met the primary endpoint in the phase 3 REVEAL‑2 trial for chronic thyroid eye disease (TED). Patients receiving the drug every four or eight weeks showed 50‑54% proptosis responder rates and 55‑61%...

5 Key April FDA Approvals Signal Momentum Across Rare, Chronic Diseases
April 2026 saw five FDA approvals spanning HIV, type 1 diabetes, chronic spontaneous urticaria, genetic hearing loss, and systemic lupus erythematosus. Merck’s Idvysno introduced the first tenofovir‑free, non‑INSTI two‑drug HIV regimen, while Sanofi’s teplizumab received clearance for children as young as...

Key Biosimilars Events of April 2026
In April 2026 the biosimilar landscape saw a wave of regulatory approvals and strategic deals. The European Commission cleared Poherdy (pertuzumab) and Tuyory (tocilizumab), while Health Canada and China’s NMPA approved multiple denosumab biosimilars and granted IND clearances for Henlius’...

Foundayo’s Liver Failure Blip Weighs Down Lilly Shares but Analysts Unconcerned
A single hepatic failure case linked to Eli Lilly’s new weight‑loss pill Foundayo appeared in the FDA’s adverse‑event database, prompting a brief sell‑off that pushed the stock down about 3% before rebounding to close up 0.48%. Lilly quickly investigated and...

CareFusion 213, LLC - 722729 - 04/30/2026
The FDA issued Warning Letter 320‑26‑72 to CareFusion 213, LLC, a BD subsidiary, citing extensive CGMP violations at its El Paso sterile drug facility. Inspectors documented over 2,500 customer complaints involving foreign particles, missing components, and compromised seals, and found the company’s investigations,...

Active Cosmetics Manufacturing Inc. - 722408 - 04/22/2026
The FDA issued a warning letter to Active Cosmetics Manufacturing Inc. after an October 2025 inspection uncovered multiple Current Good Manufacturing Practice (CGMP) violations. The firm failed to conduct thorough investigations of out‑of‑specification microbial results, used an inadequately validated rapid...

Intelligent Remedies, Inc. - 681941 - 01/23/2026
The FDA issued a warning letter (CMS #681941) to Intelligent Remedies, Inc., alleging that ten of its products—including Pryenda, Athrombosyn, and VIRAsol—are marketed with therapeutic claims that classify them as new drugs. The agency says the claims violate the Federal Food,...

Respilon Production S.R.O. - 719705 - 04/20/2026
The U.S. Food and Drug Administration issued Warning Letter 320‑26‑69 to Respilon Production S.R.O., a Czech over‑the‑counter drug manufacturer, citing multiple Current Good Manufacturing Practice (CGMP) violations. The FDA found failures in identity testing of components, lack of stability studies,...

Ray’s Vitamins - 726694 - 04/24/2026
The FDA issued a warning letter to Ray’s Vitamins after discovering that its product “Yeicob Ácido Hialurónico” contains undeclared diclofenac and dexamethasone, classifying it as an unapproved new drug and a misbranded drug. The agency cited violations of sections 301(d), 505(a) and...

STAT+: Pharmalittle: We’re Reading About RFK Jr. Targeting Antidepressants, J&J Pushing an IBD Drug, and More
U.S. Health and Human Services Secretary Robert F. Kennedy Jr. announced a new HHS initiative aimed at curbing the widespread prescription of selective serotonin reuptake inhibitors (SSRIs), which currently treat roughly 16.7% of American adults. The plan introduces clinician training,...
Phase 3 FUZION Data Show Guselkumab Benefit in Perianal Fistulizing Crohn Disease
Late‑breaking Phase 3 FUZION data presented at DDW 2026 show that guselkumab significantly improves combined fistula remission in adults with perianal fistulizing Crohn disease. At 24 weeks, remission rates were 28.3% with 100 mg every eight weeks and 27.0% with 200 mg every four weeks,...
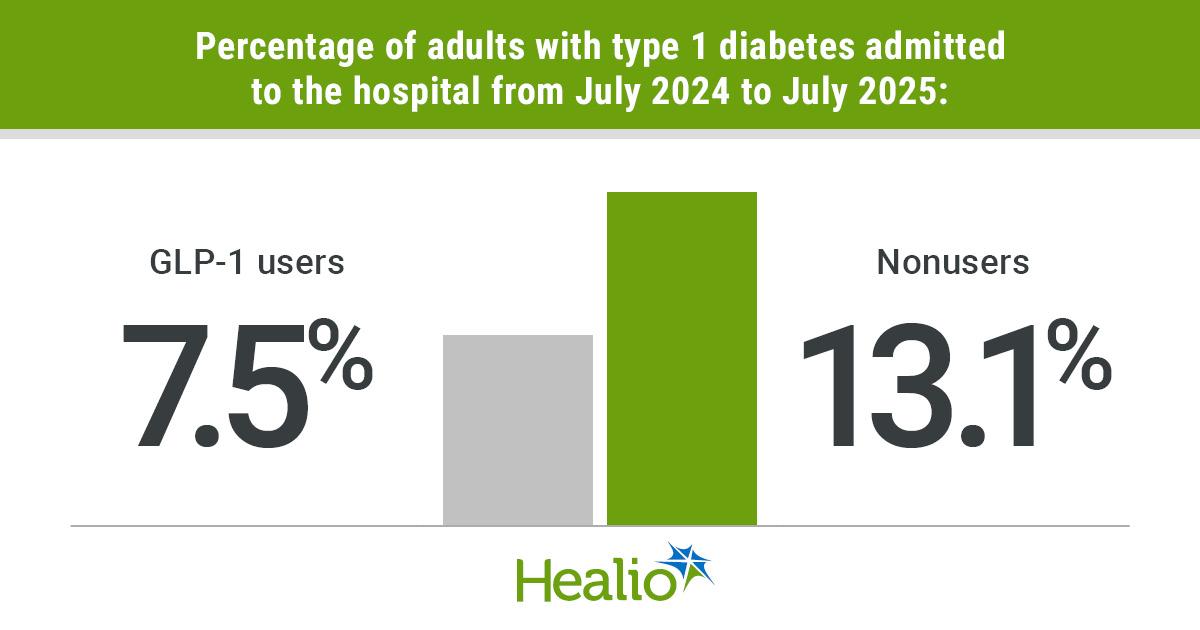
GLP-1s May Not Raise DKA, Pancreatitis Risk in Type 1 Diabetes
A single‑center study of 7,377 adults with type 1 diabetes found that none of the 255 patients using GLP‑1 receptor agonists were hospitalized for diabetic ketoacidosis (DKA) or pancreatitis over a one‑year period. Overall hospital admission rates were significantly lower for...

Disc Medicine Reports First Quarter 2026 Financial Results and Provides Business Update
Disc Medicine reported Q1 2026 results, highlighting the completion of enrollment in its Phase 3 APOLLO trial of bitopertin for erythropoietic protoporphyria, with topline data expected in Q4 2026. The company also announced that Phase 2 data for DISC‑0974 in myelofibrosis‑related anemia will be...

Do We Really Need Smarter AI to Cure Cancer?
The debate over AI’s role in curing cancer intensifies as Emilia Javorsky argues that over‑investment in AGI/ASI distracts from practical AI tools and data infrastructure needed for personalized oncology. She stresses cancer’s heterogeneity, urging focus on early detection, trial acceleration,...
Minerva Neurosciences Provides First Quarter 2026 Financial Results and Business Updates
Minerva Neurosciences announced the start of its global confirmatory Phase 3 trial of roluperidone for negative symptoms of schizophrenia, enrolling about 380 patients across 40 sites, with the first patient screened in March 2026. The company reported a GAAP net loss...

STAT+: Cytokinetics Drug Myqorzo Meets Twin Efficacy Goals in Study of Genetic Heart Disease
Cytokinetics announced that its Phase 3 ACACIA trial met both primary efficacy endpoints for Myqorzo in patients with non‑obstructive hypertrophic cardiomyopathy, showing significant symptom relief and improved cardiovascular fitness. Myqorzo is already approved for the obstructive form of HCM, and this...
Slow Alzheimer’s Diagnoses ‘Mean UK Patients Missing Out on Experimental Treatments’
Alzheimer's Research UK warns that delayed or imprecise diagnoses are keeping UK patients out of a surge of experimental drug trials. While global trials hit a record 192 this year, fewer than 1,000 UK participants are enrolled in phase‑3 studies....
U.S. Courts Are Once Again Litigating Abortion Pill’s Distribution By Mail
U.S. courts are once again debating the mail distribution of the abortion pill mifepristone. The Fifth Circuit Court of Appeals ruled that the drug can only be dispensed in person at clinics, overturning the FDA’s 2021 rule that allowed mail...

Lannett Company and Sunshine Lake Pharma Report US FDA Approval of Langlara (Biosimilar, Lantus)
The U.S. Food and Drug Administration has granted interchangeable biosimilar status to Langlara, a copy of Sanofi’s Lantus insulin glargine. Langlara is approved for adults and children with type 1 diabetes and for adults with type 2 diabetes, based on data showing...

Mirum Pharmaceuticals Reports P-IIb (VISTAS) Trial Data on Volixibat in Primary Sclerosing Cholangitis (PSC) Patients with Cholestatic Pruritus
Mirum Pharmaceuticals announced that its Phase IIb VISTAS trial met the primary endpoint, showing volixibat (20 mg BID) reduced cholestatic pruritus in primary sclerosing cholangitis (PSC) patients by 2.72 points on the Adult ItchRO scale versus a -1.08 point change with...

STAT+: Johnson & Johnson Advances IBD Therapy, Despite Trial Miss
Johnson & Johnson's combination therapy JNJ-4804, pairing Tremfya and Simponi, failed to meet the primary clinical remission endpoint in two Phase 2b trials for ulcerative colitis and Crohn's disease, though it outperformed each drug alone. The DUET study results were...
Sertraline Manufacturer Recalls Antidepressant Batch After UK Packaging Mix-Up
Amarox, a subsidiary of India’s Hetero Group, is recalling a batch of 100 mg sertraline tablets after a packaging error placed citalopram strips in the same cartons. The UK Medicines and Healthcare products Regulatory Agency (MHRA) issued the recall following a...

Comparing Antibiotic Outcomes in Preterm Infants
A recent multicenter cohort study examined how different antibiotic regimens affect outcomes in preterm infants born before 32 weeks. Researchers compared broad‑spectrum empiric therapy with a targeted, shorter‑duration approach, tracking mortality, necrotizing enterocolitis (NEC), late‑onset sepsis, and antimicrobial resistance. The...
Disruptions Threaten Progress in Inclusive Clinical Trials and Health Equity
JMIR Publications highlighted recent disruptions to inclusive clinical trials after a 2025 White House executive order targeting DEI programs. The order led to the termination and delayed funding of NIH‑funded studies, such as Dr. Mohottige’s kidney disease trial, affecting 383...

TGA Updates Australian Manufacturing Licences with New Approvals and Regulatory Actions
The Therapeutic Goods Administration (TGA) released its latest licensing decisions, granting 14 new manufacturing licences for therapeutic goods while suspending two and revoking eight existing licences. The approvals cover a diverse set of entities, including pharmaceutical firms, biotech innovators, logistics...

Advances in Neonatal Cell Therapies: 2025 Update
The 2025 Neonatal Cell Therapies Symposium underscored the rapid maturation of manufacturing for cord blood, placental and amniotic‑derived cellular products, now operating under GMP certification. Robust quality‑management systems integrate donor eligibility, informed consent and continuous environmental monitoring to guarantee batch...
Special Packaging Enables Effective Mitochondrial Delivery
Researchers have engineered "mito‑capsules" by wrapping donor mitochondria in erythrocyte‑derived plasma membranes, a technique that markedly improves delivery and integration into recipient cells. In vitro, the capsules restored bioenergetic function in mitochondrial disease models, while in vivo studies demonstrated functional...
Cytokinetics Inc (CYTK) Q1 2026 Earnings Call Transcript
Cytokinetics reported a strong commercial debut for MYCorzo, generating $4.8 million in product revenue within the first nine weeks and securing an $11.9 million Bayer milestone payment. The drug saw rapid adoption, with over 425 prescribers and 70% of dispensed...

Grail Inc (GRAL) Q1 2026 Earnings Call Transcript
GRAIL reported Q1 2026 results showing revenue of $36.2 million, a 26% year‑over‑year increase, driven by 45,000 Gallery tests sold—a 39% volume rise. The company completed a $325 million private placement and secured a pending $110 million equity investment from Samsung to accelerate...

Ocugen Inc (OCGN) Q1 2026 Earnings Call Transcript
Ocugen reported major clinical progress across its gene‑therapy pipeline, completing enrollment for the OCU400 Phase 3 Limelight trial and delivering strong 12‑month efficacy data for OCU410 in geographic atrophy and OCU410ST in Stargardt disease. The company announced a rolling Biologics License...
Cumberland Pharmaceuticals Inc (CPIX) Q1 2026 Earnings Call Transcript
Cumberland Pharmaceuticals reported an 18% revenue increase to $44.5 million for 2025, driven by strong sales of Vibativ, Sancuso and the newly launched Talicia. Adjusted earnings turned positive at $1.7 million and cash flow from operations rose to $4.9 million, narrowing the annual...

Alkermes Plc (ALKS) Q1 2026 Earnings Call Transcript
Alkermes reported $1.5 billion in 2025 revenue, driven by a 9% rise in its proprietary product portfolio, and closed the $775 million cash Avadel acquisition in February 2026, adding the LUMRIZE sleep‑medicine platform. The company forecast 2026 total revenue of $1.73‑$1.84 billion with...

Exelixis Inc (EXEL) Q1 2026 Earnings Call Transcript
Exelixis reported first‑quarter 2026 revenue of $611 million, driven by an $555 million cabozantinib franchise that grew 8% year‑over‑year and lifted global sales to $764 million, a 12.5% increase. The company posted record new patient starts for CABOMETYX, expanding its renal cell carcinoma...

Supernus Pharmaceuticals Inc (SUPN) Q1 2026 Earnings Call Transcript
Supernus Pharmaceuticals announced FDA approval of Qelbree, a non‑controlled ADHD medication for children aged 6‑17, with a U.S. launch planned for the second quarter of 2021. The company reported first‑quarter revenue of $130.9 million, a 38% year‑over‑year increase, driven by growth...

Neurocrine Biosciences Inc (NBIX) Q1 2026 Earnings Call Transcript
Neurocrine Biosciences reported Q1 2026 results with total product sales exceeding $2.8 billion, a 22% year‑over‑year increase driven by strong performance of INGREZZA and the first‑year launch of CRENESSITY. INGREZZA generated over $2.5 billion, up 9%, while CRENESSITY posted more than $300 million, covering...

Vertex Drops mRNA Cystic Fibrosis Program over 'Tolerability' Issues
Vertex Pharmaceuticals announced it is halting development of its mRNA‑based cystic fibrosis (CF) therapy after encountering tolerability and delivery challenges. The decision follows similar setbacks at other biotech firms pursuing mRNA treatments for CF. Vertex will refocus resources on its...

DOJ Moves Certain Marijuana Products to Schedule III, Sets June Rescheduling Hearing
The U.S. Department of Justice issued an order on April 22, 2026 moving FDA‑approved and state‑licensed medical marijuana products into Schedule III of the Controlled Substances Act. A separate order sets a June 29, 2026 hearing to consider broader rescheduling of marijuana from Schedule I to...

FDA Expectations Create Potential Friction in New Form 483 Response Guidance
The FDA has issued a draft guidance outlining heightened expectations for Form 483 response submissions, emphasizing greater detail and faster turnaround. Cooley’s life‑sciences regulatory chair, Sonia Nath, warned that these demands could generate friction between regulators and manufacturers. She urged...

Protein Biomarkers in Practice: Strategies to Reduce Drug Development Risk
Protein biomarkers are emerging as pivotal tools for reducing risk across the drug development lifecycle. Advances in high‑throughput proteomic platforms now allow real‑time functional insights, enabling stronger target validation, patient segmentation, and measurable efficacy signals. An eBook from GEN compiles...

Axsome Therapeutics, Inc. (AXSM) Q1 2026 Earnings Call Transcript
Axsome Therapeutics held its first‑quarter 2026 earnings conference call on May 4, 2026. CEO Herriot Tabuteau, CFO Nick Pizzie and CCO Ari Maizel presented the company’s financial results, commercial strategy and pipeline milestones. The discussion featured forward‑looking statements about upcoming Phase 3 data and reaffirmed...

Pharma Exports Surpass $31 Billion in FY26 Despite Global Headwinds
India’s pharmaceutical exports exceeded $31 billion in FY 26, marking a robust overall performance despite a sharp 23% drop in March shipments. The decline was driven by a 10% fall in U.S. exports and an 11.5% dip to China, both grappling with...

AAPS NBC 2026 To Highlight Predictive Tools in Drug Discovery with Opening Plenary
The AAPS National Biotechnology Conference 2026 will open with a plenary by Johns Hopkins professor Thomas Hartung, focusing on artificial intelligence and new‑approach methods (NAMs) that enhance predictive toxicology and human‑relevant models. Hartung will detail how AI‑driven in‑vitro systems, organoids...

Novo Nordisk’s Wegovy Head Start on Pills Forces Investors to Rethink Eli Lilly's GLP-1 Dominance
Novo Nordisk’s oral Wegovy pill, launched in January at $149 a month, quickly doubled patient intake for telehealth provider LifeMD and attracted tens of thousands of new GLP‑1 users. The strong start has forced investors to reassess Eli Lilly’s position after...