
PYC Therapeutics Advances PKD Program with Phase 1b Multiple Ascending Dose Study Initiation
PYC Therapeutics has dosed the first patient in a Phase 1b multiple‑ascending‑dose (MAD) study of its PKD candidate PYC‑003, targeting safety, tolerability and early efficacy signals such as urinary PC1 protein, total kidney volume and eGFR. Results from the earlier Phase 1a single‑ascending‑dose (SAD) cohort are expected in the second half of calendar year 2026, with MAD readouts slated for 2027. The company also highlighted progress in its ophthalmology portfolio, where RNA‑peptide therapies VP‑001 and PYC‑001 showed safety and emerging efficacy at the ARVO 2026 conference. A recent $47 million capital raise underpins these clinical advances and positions PYC for a potential registrational Phase 2/3 trial.
The Peptide Problem: Hype Is Outrunning the Evidence
Health Canada has warned Canadians against buying and injecting unauthorized peptide products such as BPC‑157, CJC‑1295, ipamorelin, TB‑500 and retatrutide, and has already seized several shipments. Influencers on Instagram and TikTok are promoting these compounds for anti‑aging, weight loss and...

Akebia Therapeutics Inc (AKBA) Q1 2026 Earnings Call Transcript
Akebia Therapeutics reported 2025 total revenue of $236.2 million, a 47% increase year‑over‑year, driven by the commercial launch of its oral HIF‑PH inhibitor Vafseo and higher sales of the phosphate binder Auryxia. Vafseo now reaches 290,000 dialysis patients across 24 organizations,...

Monte Rosa Therapeutics Inc (GLUE) Q1 2026 Earnings Call Transcript
Monte Rosa Therapeutics reported Q1 2026 worldwide revenue of $141.2 million, a 14.3% year‑over‑year increase, driven primarily by strong U.S. sales of its AtriClip and Cryosphere product lines. Adjusted EBITDA rose to $17 million, nearly doubling the prior‑year quarter, while gross margin...
Ironwood Pharmaceuticals Inc (IRWD) Q1 2026 Earnings Call Transcript
Ironwood Pharmaceuticals reported 2025 LINZESS U.S. net sales of $865 million, matching guidance despite a 6% price erosion tied to Medicare Part D changes. Prescription demand grew 11% for the second year, maintaining a 45% market share in the IBS‑C and chronic...

There Is No Vaccine for Deadly Hantavirus: What that Means for Future Outbreaks
An outbreak of Andes hantavirus on the cruise ship MV Hondius left three confirmed cases and three deaths, highlighting the deadly potential of the rodent‑borne virus. The World Health Organization confirmed the strain has no approved treatments or vaccines. Virologist...

Corvus Pharmaceuticals Inc (CRVS) Q1 2026 Earnings Call Transcript
Corvus Pharmaceuticals reported Q1 2026 results highlighting accelerated R&D spending and a strengthened balance sheet after a $200 million public offering, giving the company cash runway into the second quarter of 2028. Clinical data from the Phase I/Ib atopic dermatitis trial...

INmune Bio Inc (INMB) Q1 2026 Earnings Call Transcript
INmune Bio reported Q1 2026 results showing a cash balance of $21.4 million, sufficient to sustain operations through Q1 2027, and a narrowed net loss of $5.4 million. The company highlighted significant progress on its CORDStrom platform, including approval of the U.K. Pediatric Investigation...

Zymeworks Inc (ZYME) Q1 2026 Earnings Call Transcript
Zymeworks reported a PDUFA deadline of August 25, 2026 for its zanidatumab HER2‑positive gastroesophageal adenocarcinoma (GEA) indication, with a supplemental BLA filed in China. The company stands to receive up to $265 million in milestone payments contingent on U.S. and Chinese...
Absci Corp (ABSI) Q1 2026 Earnings Call Transcript
Absci Corp (ABSI) reported Q1 2026 revenue of $200 K while R&D expenses rose to $19.3 M as the company advances its lead prolactin‑receptor antibody ABS‑201. The Phase 1/2a androgenic alopecia trial is on track, with safety data due next month and interim...

Harmony Biosciences Holdings Inc (HRMY) Q1 2026 Earnings Call Transcript
Harmony Biosciences reported record Q1 product revenue of $243.8 million, a 21% increase year‑over‑year, driven by sustained demand for its Wakix sleep‑wake therapy. The company added roughly 400 patients per quarter, reaching about 8,500 on Wakix, and reaffirmed 2026 revenue guidance...

Lexicon Pharmaceuticals Inc (LXRX) Q1 2026 Earnings Call Transcript
Lexicon Pharmaceuticals reported Q1 2026 revenue of $5.5 million, largely from a $4.3 million Novo Nordisk licensing deal and INPEFA sales. The company cut its quarterly net loss to $15.5 million and reduced annual operating expenses by $129.5 million, boosting cash to $125 million after...

Crinetics Pharmaceuticals Inc (CRNX) Q1 2026 Earnings Call Transcript
Crinetics reported $10.3 million net product revenue from Palsonify in Q1 2026, driven by 232 new patient enrollments and a prescriber base that grew to 263 unique physicians. Treatment‑naive patients rose to 15 % of enrollments, indicating expanding first‑line use. Reimbursement coverage...

Arrowhead Pharmaceuticals Inc (ARWR) Q2 2026 Earnings Call Transcript
Arrowhead Pharmaceuticals reported accelerating uptake of its newly launched triglyceride‑lowering therapy REDEMPLO, surpassing 400 prescriptions with a 40% surge in the last month. The company announced a price reduction to $45,000 per patient per year and secured payer policies that...

Scholar Rock Holding Corp (SRRK) Q1 2026 Earnings Call Transcript
Scholar Rock Holding Corp reported $368 million in cash and secured a $550 million debt facility, providing ample liquidity for 2026 initiatives. The FDA completed a reinspection of the Catalent Indiana site with no further requests, clearing the path for an imminent...

Caris Life Sciences Inc (CAI) Q1 2026 Earnings Call Transcript
Caris Life Sciences reported Q1 2026 revenue of $216 million, up 79% year‑over‑year, propelled by an 85% surge in molecular profiling revenue. Gross margin improved to 65% on a GAAP basis, reflecting a 61% rise in clinical average selling price and...
SIGA Technologies Inc (SIGA) Q1 2026 Earnings Call Transcript
SIGA Technologies reported Q1 2026 revenue of roughly $6 million, a pretax loss of $5 million and a cash balance of $146 million with no debt. The company announced a $13 million oral TPOXX order for an Asia‑Pacific customer in Q2 and sealed an...

Insmed Inc (INSM) Q1 2026 Earnings Call Transcript
Insmed reported that its newly launched Brinsupri generated $144.6 million in its first full quarter and now projects at least $1 billion in U.S. revenue for 2026. The company highlighted strong early adoption, with over 11,500 new patients in 2025, 90%+ payer...

Science Spotlight: New Prime Editing Tools for Large DNA Insertions
BioCentury’s website employs a tiered cookie framework that classifies cookies into strictly necessary, functional, marketing, advertising, and analytics groups. Strictly necessary cookies power core services such as authentication, registration, and user‑preference management, while functional cookies enhance site personalization. Marketing and...

An Essential Framework to Navigating Technical Challenges in Contemporary OSD Development
Oral solid dosage (OSD) development faces mounting technical hurdles as biopharma pipelines become richer in poorly soluble compounds, with 70‑90% of new small‑molecule candidates classified as BCS Class II or IV. Early‑risk identification—through comprehensive physicochemical profiling, excipient compatibility testing, and Quality...

The BioPharm Brief: Precision, Immunotherapy, Expansion
Madrigal Pharmaceuticals is licensing Arrowhead’s RNA‑interference candidate ARO‑PNPLA3 to broaden its precision‑medicine pipeline for metabolic dysfunction‑associated steatohepatitis (MASH). LTZ Therapeutics secured $38 million to push forward myeloid‑engager immunotherapies that activate innate immune cells against cancer. Amgen and Eli Lilly announced major U.S....

Dems Probe Withholding Of Vaccine Safety Studies At CDC, FDA
Democrats on the House Energy & Commerce Committee are pressing HHS Secretary Robert F. Kennedy Jr. for an explanation after a CDC study that found COVID‑19 vaccines cut hospitalizations during the latest respiratory virus season was not released. The committee...

MFN Drug Pricing Proposal Raises Questions Around Access, Innovation, and Commercial Coverage
The White House’s most‑favored‑nation (MFN) drug pricing framework projects roughly $600 billion in savings over the next decade, tying U.S. prices to the second‑lowest net price among eight high‑income reference countries. The plan distinguishes prospective launches, which could slash net prices...

Self-Organizing ‘Pencil Beam’ Laser Could Help Scientists Design Brain-Targeted Therapies
MIT researchers discovered that under high power, chaotic laser light in a multimode fiber can spontaneously self‑organize into a tightly focused “pencil beam.” Using this beam, they captured three‑dimensional images of a human blood‑brain barrier model 25 times faster than...

DNA-Guided CRISPR Flips Gene Editing Script, Opening a New Path for Precise Diagnosis and Antivirals
Researchers at Hong Kong University of Science and Technology have created the first DNA‑guided CRISPR‑Cas12a system that can programmatically target and cleave RNA. The new platform, called SLEUTH, combines the DNA‑guided enzyme with isothermal amplification to achieve attomolar‑level detection of...

What’s New in Clinical Trial Innovation
The FDA’s Center for Clinical Trial Innovation (C3TI) publishes a newsletter that highlights new developments, opportunities, and initiatives in clinical‑trial innovation. Readers can subscribe via a public.govdelivery.com form to receive updates directly in their inbox. The site also provides free...
The Human Genome Encodes for a New Category of Molecule
Scientists have identified a previously unrecognized class of molecules encoded within the human genome, arising from short open reading frames once deemed non‑coding. These micro‑proteins, often called micropeptides, exhibit distinct biochemical activity and appear to regulate cellular pathways. Early experimental...
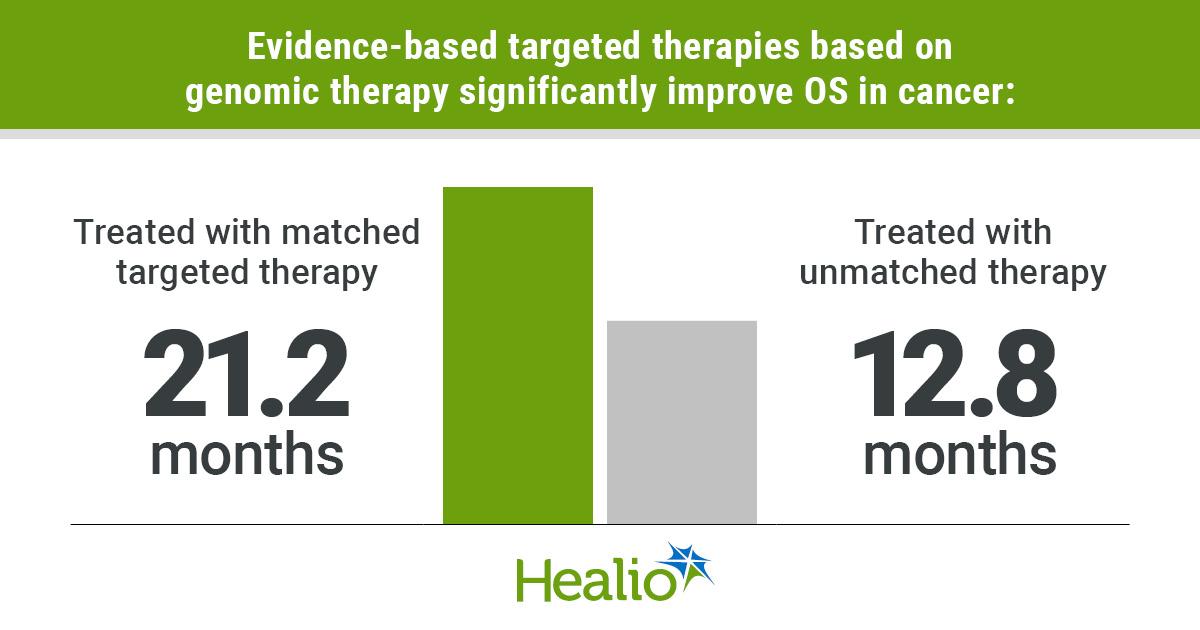
Treating Cancer Based on Mutation Alone Does Not Improve Survival
A large Australian study of 3,383 advanced‑cancer patients found that targeted therapies approved for a specific mutation within the same tumor type improved overall survival by roughly 40%, while using a drug solely because of a shared mutation across different...

OECD Blog Item Explores Why Biotech Start-Ups Lag in Europe
The OECD’s May 6 blog highlights that Europe continues to fall behind the United States and Asia in biotech start‑up formation, patenting, and venture‑capital funding. It points to the European Commission’s December 2025 Biotech Act, which introduces regulatory sandboxes, pre‑submission consultations, a...

Bayer Sees Opportunity With $300M Acquisition of Eye Drug Biotech
Bayer announced a $300 million upfront acquisition of Perfuse Therapeutics, a clinical‑stage biotech developing PER‑001, an endothelin‑receptor blocker delivered via a six‑month intravitreal implant. Phase 2 trials showed significant vision improvement in glaucoma and diabetic retinopathy, positioning the drug as a potential...

HOPE Supports Tailored Approach to BP After Stroke Thrombectomy
The HOPE trial, presented at the European Stroke Organisation Conference 2026, tested a reperfusion‑guided blood‑pressure strategy after endovascular thrombectomy for acute ischemic stroke. Patients whose post‑procedure mTICI score was 2b were targeted to a systolic BP of 140‑160 mm Hg, while those...
Brolucizumab Superior in Preserving Visual Acuity in Proliferative Diabetic Retinopathy
A phase‑3 CONDOR trial of 689 adults with proliferative diabetic retinopathy (PDR) showed that intravitreal brolucizumab outperformed panretinal laser photocoagulation (PRP) in preserving visual acuity over 54 weeks. The brolucizumab arm gained a mean BCVA change of +0.2 letters versus...

Clinical Trials Day
Clinical Trials Day on May 20 commemorates James Lind’s 1747 scurvy experiment, widely regarded as the first randomized clinical trial. The observance highlights the enduring role of trials in generating the safety and efficacy data that underpin FDA drug approvals. FDA’s...

BsUFA IV: Fiscal Years 2028-2032
The FDA announced the kickoff of the fourth Biosimilar User Fee Act (BsUFA IV) reauthorization, covering fiscal years 2028‑2032. A public meeting was held on December 3, 2025 to launch the process, following the 2022 law that reauthorized BsUFA II. BsUFA III expires in September 2027,...

In a First, Scientists Are Rewinding Human Cells Back to a ‘Youthful’ State. Is This the Dawn of Immortality?
Scientists are advancing partial cellular reprogramming to reverse age‑related decline while preserving cell identity. YouthBio Therapeutics is preparing a first‑in‑human trial of its brain‑targeted YB002 program for Alzheimer’s after receiving FDA feedback. Parallel efforts such as Life Biosciences’ ER‑100 aim...

FDA Vaccine Studies Censored by Trump Admin After Finding Benefits of Shots
The Food and Drug Administration, under the Department of Health and Human Services, blocked the publication of two internal studies that demonstrated the safety and efficacy of COVID‑19 vaccines, and prevented two Shingrix (shingles vaccine) abstracts from being presented at...

Tofersen, a New Treatment for A.L.S., Reverses Symptoms for Some
Tofersen, the first FDA‑approved therapy targeting the SOD1 genetic form of ALS, is showing functional gains in a subset of patients. In a case highlighted by the New York Times, 58‑year‑old Amanda Sifford’s lung capacity rebounded from 48% to 86%...
LTZ Therapeutics Secures $38M to Advance Myeloid Engager Immunotherapy Pipeline
LTZ Therapeutics announced an oversubscribed $38 million financing round led by GL Ventures, bringing its total capital to roughly $130 million since 2022. The funds will propel the Universal Myeloid Cell Engager (U‑MCE) platform, supporting the Phase 1 trial of lead bispecific antibody...

Cannabis Execs Anticipate Tax Benefits From Rescheduling
The DOJ and DEA reclassified medical cannabis as a Schedule III drug on April 23, ending its Schedule I status. This change eliminates the IRS 280E restriction for medical cannabis businesses, potentially cutting tax burdens by up to half. Industry leaders Curaleaf and Green...

Heavy-Chain BsAbs More Manufacturable than Light-Chains
Bispecific antibodies (BsAbs) have grown to 19 FDA approvals and roughly 250 candidates in development, but scaling their production remains a bottleneck. A recent study by Universidad Nacional Autónoma de México researchers compared six BsAb architectures and linked design to...

Researchers’ Spinout Focuses on Simplifying Viral Vector Purification
Researchers at North Carolina State University have spun out ChromaGenix to commercialize synthetic peptide ligands for affinity purification of viral vectors used in gene therapies. The peptide ligands are cheaper, more stable and less immunogenic than traditional protein ligands, cutting...

Moss Powering the Next Drug Frontier
Eleva is commercializing a moss‑based biomanufacturing platform that can produce complex glycoproteins difficult to express in traditional CHO or yeast systems. The German firm has advanced its first candidate, a recombinant alpha‑galactosidase for Fabry disease, into clinical trials and is...
Regulators Should Rely on Peers’ GMP Audits to Cut Inspection Burden
Biopharma manufacturing sites face an average of 2.68 GMP inspections per year, each lasting up to nine days, and preparation can take six months to a year. The International Federation of Pharmaceutical Manufacturers and Associations (IFPMA) argues that regulators should...

First U.S. Patients Treated With Microrobotic Surgery For Alzheimer’s
A microrobotic surgery trial for Alzheimer’s disease began at Baptist Health in Jacksonville, treating the first patient with moderate disease. Medical Microinstruments Inc. (MMI) plans to enroll 15 participants and monitor them for a year, aiming to clear cervical lymph‑node...
Financings for May 6, 2026
UK‑based Cytospire Therapeutics announced a £61 million (≈$82.7 million) Series A round to accelerate its EGFR‑targeted T‑cell engager pipeline for solid tumours. The capital backs multiple pre‑clinical programs and reflects strong investor confidence in next‑generation immunotherapies. Bio Korea 2026 highlighted a shift in...

Other News to Note for May 6, 2026
Cytospire Therapeutics announced a £61 million (≈$82.7 million) Series A round to advance its next‑generation T‑cell engager platform targeting the epidermal growth factor receptor (EGFR) in solid tumours. The funding will accelerate pre‑clinical and early‑clinical programs aimed at delivering more selective immunotherapies. Meanwhile,...

IPLEDGE Hurdles Block some Inmates From Receiving Isotretinoin in Jails, Prisons
The FDA‑run iPLEDGE REMS program blocks many incarcerated patients from receiving isotretinoin because it requires pharmacies to register with an NCPDP number—a identifier jails and prisons do not possess. In addition, the system mandates a phone number or email for...

CoCoGraph AI Model Generates Molecules that Comply with Rules of Chemistry
Researchers at Universitat Rovira i Virgili have unveiled CoCoGraph, an AI diffusion model that generates synthetic molecules while strictly adhering to fundamental chemical rules. By progressively disordering and reconstructing real molecules, the system ensures valid bond counts and produces chemically...

A New Kind of CRISPR Could Treat Viral Infection and Cancer by Shredding Sick Cells' DNA
Researchers have engineered a novel CRISPR protein, Cas12a2, that acts as a molecular shredder, destroying DNA in cells that express a specific RNA trigger. In vitro, Cas12a2 cut the growth of KRAS‑mutant lung‑cancer cells by 50% and eliminated over 90%...
IV Ketamine Shows Rapid Benefits for Suicide Risk, Depression in Major Depressive Episodes
A new meta‑analysis of 26 randomized trials involving 1,166 patients shows that intravenous ketamine rapidly alleviates both suicidal ideation and depressive symptoms in major depressive episodes. Single infusions cut suicidal scores by an SMD of –0.69 within 24 hours and depressive...