
The VESALIUS-CV Trial
In the VESALIUS-CV trial, researchers evaluated the impact of adding the PCSK9 inhibitor evolocumab to standard lipid‑lowering therapy in patients at high cardiovascular risk who had never experienced a myocardial infarction or stroke. Over a median follow‑up of 3.2 years, evolocumab lowered LDL‑cholesterol by roughly 55% and cut the composite endpoint of cardiovascular death, non‑fatal myocardial infarction, non‑fatal stroke, or coronary revascularization by 20% compared with placebo. The absolute risk reduction was 1.2 percentage points, translating to a number needed to treat of 83 to prevent one event. Safety profiles were comparable, with no increase in adverse events reported.
Cyclin E1 and CCNE1 Shift in Ovarian Cancer Post-PARP
The British Journal of Cancer study shows high‑grade serous ovarian carcinoma tumors develop resistance to PARP inhibitors by up‑regulating Cyclin E1 and amplifying the CCNE1 gene. Researchers compared pre‑ and post‑treatment samples using FISH and IHC, finding a significant rise in...

Novel Psychedelic Compound 25C-NBF Shows Rapid Antidepressant Effects without Addictive Traits
Researchers reported that the synthetic psychedelic 25C‑NBF rapidly induces dendritic growth and reverses depressive behaviors in male rodents after a single dose. The compound binds tightly to the serotonin 5‑HT2A receptor while showing minimal activity at 5‑HT2B, reducing heart‑valve risk....

STAT+: Novo Nordisk’s Less-Bad News on Its Wegovy Pill Boosts Earnings and Share Price
Novo Nordisk reported first‑quarter results that showed a modest improvement in its outlook for the obesity drug Wegovy. The company narrowed its adjusted sales decline forecast to 4%‑12% for the year, up from a prior 5%‑13% range, and aligned its...

STAT+: Pharmalittle: We’re Reading About a PhRMA Ad Campaign Aimed at 340B, Sales of Novo’s Obesity Pill, and More
PhRMA, the leading pharmaceutical lobby, has launched a seven‑figure TV advertising campaign aimed at the 340B Drug Pricing Program, accusing hospitals of misusing the discount to benefit themselves rather than low‑income patients. Meanwhile, FDA data show the rejection rate for...

BIOTECanada Responds to CDA-AMC Guidance for Incorporating Impacts on Informal Caregivers and Productivity Outcomes in Economic Evaluations.
BIOTECanada, together with Innovative Medicines Canada, submitted a formal response to the Canadian Drug Agency’s draft guidance on incorporating informal caregiver health‑related quality of life and productivity outcomes into economic evaluations. The association recommends expanding the societal perspective to all...

Understanding Drug Recalls: What to Know and What to Do
The FDA oversees drug recalls, publishing announcements on FDA.gov and maintaining a searchable database of all recalled products. Recalls are classified into three severity levels—Class I (life‑threatening), Class II (temporary health effects), and Class III (minor labeling or packaging issues). Consumers are advised...

Top Biotech Deals in April 2026
April 2026 marked a flurry of blockbuster biotech mergers, highlighted by Sun Pharma’s $11.75 billion acquisition of Organon, giving it a foothold in women’s health across 140 countries. Eli Lilly emerged as the most active buyer, spending roughly $11.5 billion on four...

BriaCell Therapeutics Reports US FDA IND Clearance to Initiate P-I/II Trial of Bria-BRES+ in Breast Cancer
BriaCell Therapeutics announced that the U.S. Food and Drug Administration has granted IND clearance to launch a combined Phase I/II study of its Bria‑BRES+ therapy in patients with metastatic breast cancer. Preclinical results presented at AACR indicate Bria‑BRES+ stimulates both...
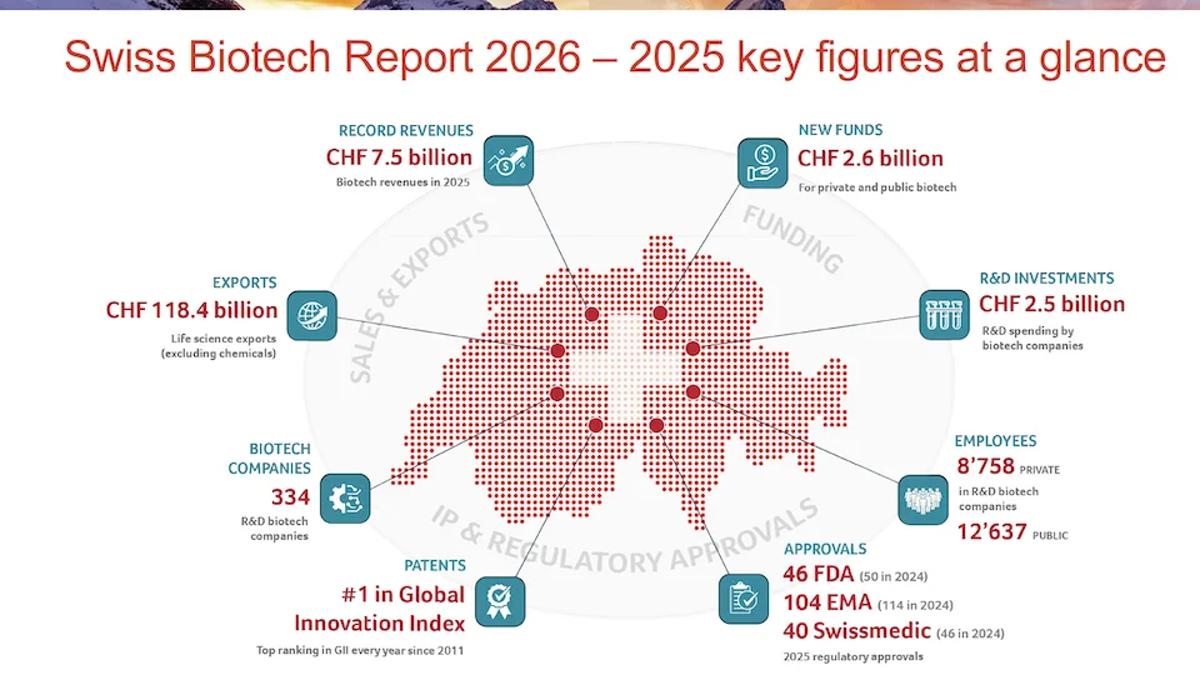
Switzerland’s Biotech Sector Defies Tough Markets with Record Revenue
Switzerland’s biotech sector posted a record CHF 7.5 billion ($8.3 billion) in revenue for 2025, up from CHF 7.2 billion the year before, driven by more market‑ready products and strong CDMO demand. Capital inflows rose 2.1% to CHF 2.6 billion ($2.9 billion), with privately financed firms leading the...

FDA Approves Selpercatinib for Medullary Thyroid Cancer with a RET Mutation
The FDA granted traditional approval on September 27, 2024, for Eli Lilly’s selpercatinib (Retevmo) to treat adult and pediatric patients two years and older with advanced or metastatic medullary thyroid cancer (MTC) harboring a RET mutation. The decision follows accelerated approvals...

SCIENTURE Announces Third Patent Grant for ARBLI™ (Losartan Potassium) Oral Suspension, Strengthening Long-Term Market Exclusivity Through 2041
Scienture Holdings announced that the U.S. Patent and Trademark Office granted a third patent (U.S. Patent No. 12,605,365) for its ARBLI™ losartan potassium oral suspension. The new patent, issued April 21, 2026, extends exclusive protection through October 7, 2041, reinforcing the company’s IP moat. ARBLI™...

Novelty Nobility Expands AGC Biologics Deal to Take Product Candidate Through GMP Manufacturing
Korea‑based Novelty Nobility has expanded its contract with CDMO AGC Biologics to move its bispecific antibody NN4101 through process development and GMP manufacturing at AGC’s Chiba, Japan facility. Cell‑line development was completed in Copenhagen and will be transferred to Chiba...
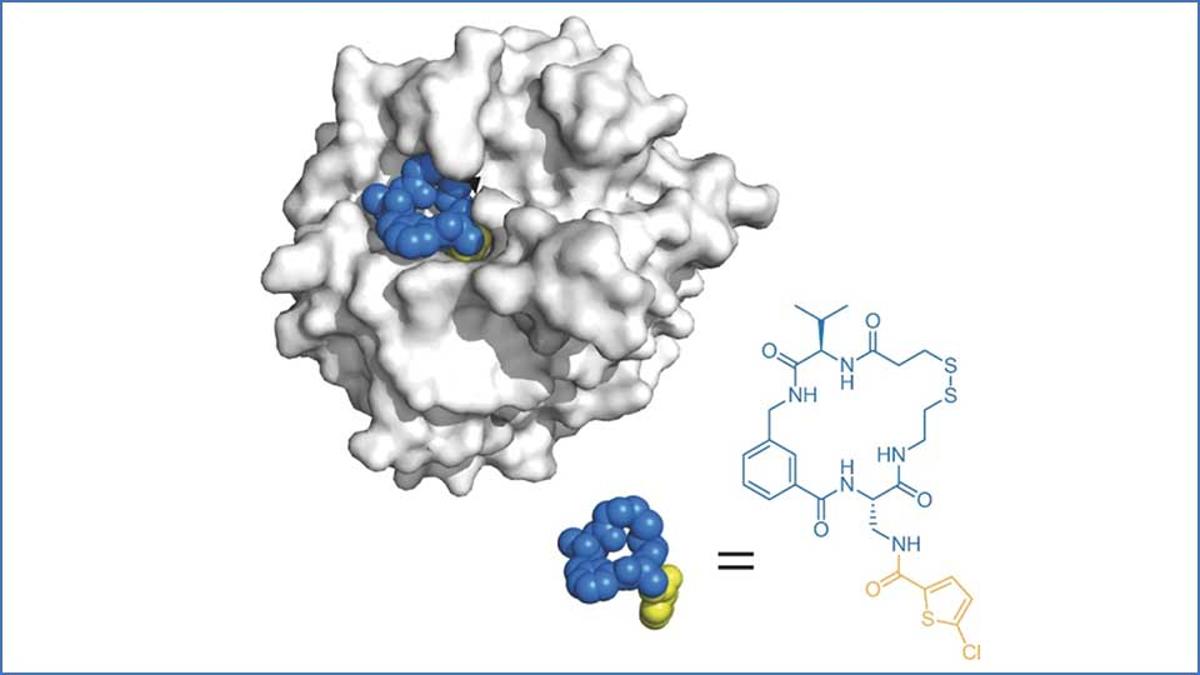
Macrocycles: Big Is the New Beautiful
Macrocycles are gaining traction as a hybrid drug modality that combines the high specificity of biologics with the oral accessibility of small molecules. Advances in synthetic chemistry, AI‑driven design, and DNA‑encoded libraries now enable rapid exploration of billions of candidates,...

Alebund Pharmaceuticals Completes Patient Enrollment in P-III (RESPOND-2) Trial of AP301 for Hyperphosphatemia
Alebund Pharmaceuticals announced that patient enrollment is complete for its global Phase III RESPOND‑2 trial of AP301, a novel therapy for hyperphosphatemia in dialysis patients. The study enrolled 282 participants—138 in the United States and 144 in China—exceeding the planned 264....

4basebio Announces Lease of an Innovation Hub and Manufacturing Facility in Cambridge, UK
4basebio PLC announced a lease for a 26,500 sq ft innovation hub and manufacturing facility in Cambridge, UK, slated to open in late summer 2026. The site will feature 7,500 sq ft of specialized laboratory space with 15 labs, advanced biosafety equipment, and a...
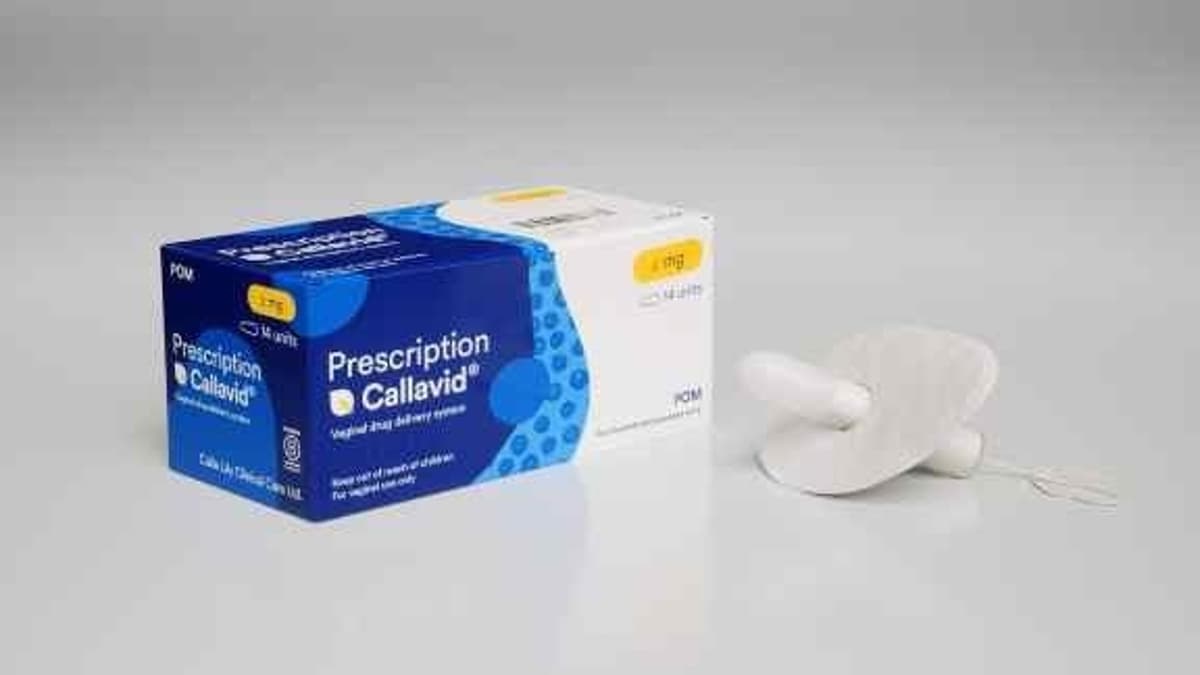
Calla Lily Clinical Care Doses First Patients in Clinical Trial for Intravaginal Drug Delivery Platform for Threatened Miscarriage
Calla Lily Clinical Care has begun dosing the first participants in the FREEDOM clinical trial, testing its 400 mg intravaginal progesterone product Callavid. The NIHR‑funded study targets women with luteal phase insufficiency, a condition linked to threatened miscarriage and infertility. Callavid’s...

CellCentric Raises $220M to Get Multiple Myeloma Pill to Market
CellCentric, a 22‑year‑old transatlantic biotech, closed a $220 million Series D round to accelerate development of its oral multiple myeloma drug. The capital will fund the completion of Phase III trials, scale manufacturing, and prepare regulatory filings. The therapy is designed for patients...
Pfizer, Lilly, More Report Q1, FDA Names Acting CBER Director and an ALS Awakening
Pfizer posted first‑quarter earnings that topped consensus estimates, yet analysts deemed the beat insufficient given higher expectations for the New York‑based giant. Eli Lilly delivered a striking 56% revenue surge, though the company noted that price reductions on several products capped its...

Bayer to Buy Perfuse Therapeutics for $300M Upfront
Bayer announced a $300 million upfront payment to acquire ophthalmology biotech Perfuse Therapeutics, securing its mid‑stage program aimed at the leading causes of blindness. The transaction represents Bayer’s first pharmaceutical merger‑and‑acquisition in several years, signaling a renewed focus on specialty eye...

Signadori Bio Raises €11M to Advance Tumour Platform
Signadori Bio announced an €11 million ($11.9 million) seed extension to fund its in‑vivo monocyte engineering platform for solid tumours. The Series A round, led by Taiho Ventures with Sofinnova and Invivo Partners, will support lead candidate nomination, platform refinement, and team expansion...

Novo Nordisk's Q1 in Four Words: Pills, Pricing, Payments and Pressure
Novo Nordisk’s first‑quarter performance centered on its obesity drug Wegovy, which now serves roughly one million patients in the United States. The company is navigating heightened pricing pressure as insurers push for larger rebates while regulators scrutinize the drug’s cost....

Sickle Cell Disease After Casgevy: Seven Companies to Watch in 2026
Casgevy’s $2.2 million, ex vivo CRISPR cure proved sickle cell disease can be edited at its genetic root, but its complex manufacturing and conditioning limit broad access. In response, a wave of innovators is targeting simpler, safer, and more scalable solutions—from Beam’s...

Cytospire Therapeutics Secures £61m to Advance Cancer Treatment
Cytospire Therapeutics, a UK biotech, closed a £61 million (≈$77 million) Series A round to accelerate its multispecific engager antibody platform. The capital will fund the development of its lead candidate, CYT X300, aimed at EGFR‑positive solid tumours such as colorectal, head‑and‑neck and non‑small‑cell...

Overcoming Material Constraints: Aseptic Filling Solutions for Rare or Expensive APIs
Aseptic fill‑finish lines can waste more than a liter of drug product per batch, translating to losses exceeding $1 million for high‑value APIs. The bulk of this loss occurs during sterile filtration and filling, where product remains trapped in vessels, tubing,...
Furin‐Mediated Intracellular Aggregation of Radioactive Molecules for Enhanced Radionuclide Imaging and Tumor Therapy
Researchers have engineered a furin‑responsive radioactive probe, RVRR‑TPE, that self‑assembles into nanoparticles inside furin‑positive cancer cells. The molecule couples a furin‑cleavable Arg‑Val‑Arg‑Arg peptide, an aggregation‑induced emission fluorophore (tetraphenylethene), and a phenol group for iodine‑125/131 labeling. In mouse models, the 125I/131I‑labeled...

STAT+: Even at a Meeting in Rome, FDA Shifts Are Top of Mind for Gene Therapy Field
At a gene‑therapy summit in Rome, Tim Hunt highlighted recent FDA approvals of rare‑disease treatments from Rocket Pharmaceuticals and Regeneron as signs of progress. He also flagged the departure of Vinay Prasad, the FDA’s top regulator for cell and gene therapies,...

Caris Launches Caris MI Clarity for AI-Powered Breast Cancer Recurrence Risk Assessment
Caris Life Sciences has introduced Caris MI Clarity, an AI‑driven prognostic test that evaluates both early (0‑5 years) and late (5‑15 years) distant recurrence risk for postmenopausal patients with HR‑positive/HER2‑negative, node‑negative early‑stage breast cancer. The assay analyzes digitized H&E pathology...
With Large DNA Fragment Assembly, Scientists Can Design Microbes that Produce Countless Complex Products
A new review in Quantitative Biology shows scientists can now reliably assemble very large DNA fragments, enabling the construction of whole metabolic pathways and even extra chromosomes inside microbes. This capability turns yeast and bacteria into efficient cell factories that...

Popular GLP-1 Drugs Significantly Reduce Major Cardiovascular Events,
A systematic review and meta‑analysis of eleven cardiovascular outcome trials involving 91,490 high‑risk patients found that GLP‑1 receptor agonists reduce major adverse cardiovascular events (MACE) by 14% compared with placebo. The therapy also lowered cardiovascular mortality by 13% and improved...

World's First Vaccine for Lyme Disease Could Be Available in 2027
Pfizer and French partner Valneva announced that their Lyme disease vaccine candidate PF‑07307405 (LB6V) achieved 73.2% efficacy in Phase III trials involving more than 9,000 participants. Although the study missed its primary statistical endpoint due to a low incidence of cases,...

EyePoint Inc (EYPT) Q1 2026 Earnings Call Transcript
EyePoint Pharmaceuticals reported a dramatic revenue drop to $600,000 for the quarter, reflecting the reversal of deferred YUTIQ license revenue, while operating expenses surged to $71 million as Phase 3 trials for its DuraVu platform intensified. The company posted a net loss...
Vanda Pharmaceuticals Inc (VNDA) Q1 2026 Earnings Call Transcript
Vanda Pharmaceuticals reported Q1 2026 net product sales of $51.7 million, a modest 3% year‑over‑year increase driven primarily by a 26% surge in Fanapt revenue and a 32% rise in prescriptions. Hetlioz sales continued to decline, falling 24% YoY amid generic...

Taysha Gene Therapies Inc (TSHA) Q1 2026 Earnings Call Transcript
Taysha Gene Therapies announced that its Rett syndrome gene therapy TSHA-102 has moved into pivotal development, securing FDA Breakthrough Therapy designation and written alignment on both the REVEAL pivotal and ASPIRE trial designs. Early Phase 1/2 data showed a 100%...

Urogen Pharma Ltd (URGN) Q1 2026 Earnings Call Transcript
UroGen Pharma reported a $15.8 million 2025 revenue for its newly launched Zasturi, with the permanent J code effective Jan. 1 2026 spurring a sharp uptick in prescriber and site adoption. The company now has 838 activated sites, over 95% payer access, and...

Sarepta Therapeutics Inc (SRPT) Q1 2026 Earnings Call Transcript
Sarepta Therapeutics reported $1.86 billion net product revenue for 2025, a 16% increase, and ended the year with $954 million in cash and investments. Management guided 2026 net product revenue to $1.2‑$1.4 billion, emphasizing a low‑end outlook while targeting operating profit and positive...

Novavax Inc (NVAX) Q1 2026 Earnings Call Transcript
Novavax reported a Q1 2019 net loss of $43.2 million, improving from the prior year, while revenue fell 59% to $4 million as the Bill & Melinda Gates Foundation grant wound down. The company highlighted clinical milestones for its RSV vaccine ResVax,...

Quanterix Corp (QTRX) Q1 2026 Earnings Call Transcript
Quanterix Corp reported fourth‑quarter revenue of $43.9 million, a 25% year‑over‑year increase and 7% sequential growth, though organic revenue fell 22% as both Simoa and Spatial platforms declined over 20%. The company highlighted a $897 CMS reimbursement rate for its Lucent...

Zevra Therapeutics Inc (ZVRA) Q1 2026 Earnings Call Transcript
Zevra Therapeutics reported Q1 2026 net revenue of $36.2 million, a 78% year‑over‑year increase, driven primarily by MyPlifer sales of $24.6 million and a $10.2 million expanded‑access program. The company completed the $50 million divestiture of its SDX portfolio, recorded a $43.3 million one‑time gain...

Mineralys Therapeutics Inc (MLYS) Q1 2026 Earnings Call Transcript
Mineralys Therapeutics reported Q1 2026 results highlighting FDA acceptance of lorundrostat’s NDA with a PDUFA target of Dec 22, 2026. The company completed five trials showing durable blood‑pressure reductions, though the EXPLORER‑OSA study missed its primary AHI endpoint, it still demonstrated a...

Cytokinetics Scores Late-Stage Win in Non-Obstructive HCM as BMS Plans Camzyos Restart
Cytokinetics announced that its experimental drug Myqorzo (aficamten) achieved both primary endpoints in the Phase III ACACIA‑HCM trial, marking the first late‑stage success for a therapy targeting non‑obstructive hypertrophic cardiomyopathy (HCM). The result propelled the company’s stock up 17%, pushing its...
Advances in Genetic Medicine Took Center Stage at INSAR
At INSAR in Prague, researchers highlighted a surge of genetic‑medicine breakthroughs aimed at autism, focusing on rare variants such as SCN2A, SHANK3 and UBE3A. Techniques ranging from CRISPR gene editing to antisense oligonucleotides and epigenome editing demonstrated tangible symptom improvements,...

OTC Monograph Drug User Fee Amendments (OMUFA): Understanding FY 2026 User Fees and Registration - 05/05/2026
The FDA’s Center for Drug Evaluation and Research hosted a May 5, 2026 webinar to explain the Over‑the‑Counter Monograph Drug User Fee Amendments (OMUFA) for fiscal year 2026. The session covered fee structures, registration requirements for OTC monograph facilities, payment timelines, and penalties...

The BioPharm Brief: Breakthrough Biologics and Long-Term Wins in IBD and Beyond
Viridian Therapeutics reported positive Phase III REVEAL‑2 data for veligrotug (VRDN‑001) in chronic thyroid eye disease, showing statistically significant reductions in proptosis and higher overall response rates. Eli Lilly presented four‑year long‑term results for mirikizumab (Omvoh) in ulcerative colitis, with a substantial...

Questions and Answers on Current Good Manufacturing Practice Regulations | Production and Process Controls
The FDA released a detailed Q&A clarifying current Good Manufacturing Practice (CGMP) expectations for pharmaceutical manufacturers. It addresses equipment labeling, warehouse sampling of containers, media‑fill contamination sources, and the number of validation batches required for new products. The guidance also...
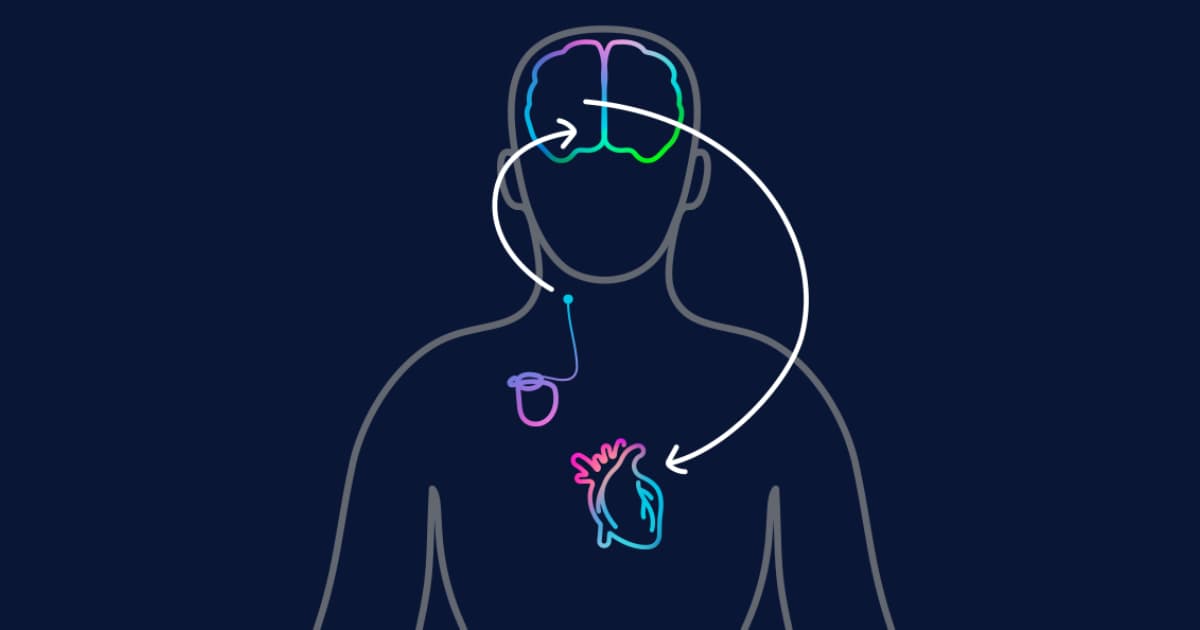
First Patient Enrolled in Massive Heart Failure Trial
CVRx has begun enrolling patients in BENEFIT‑HF, a pivotal trial of its Barostim implant for heart failure. The study plans to enroll roughly 2,500 NYHA Class II‑III patients across the United States and Germany and will run through 2032. Participants must...

F.D.A. Blocked Publication of Research Finding Covid and Shingles Vaccines Were Safe
The U.S. Food and Drug Administration blocked publication of several agency‑conducted studies that found Covid‑19 and shingles vaccines to be safe. The withdrawn research, funded with public dollars, had been slated for peer‑review journals and a drug‑safety conference. FDA officials...

White House Estimates Trump’s Big Pharma Dealmaking Will Save Americans $529 Billion over the Next 10 Years
White House economists estimate that President Trump’s “most‑favored‑nation” drug‑pricing deals could save Americans $529 billion over the next ten years, with Medicaid projected to cut $64.3 billion in costs. The analysis, prepared by the Council of Economic Advisers, assumes 17 major pharmaceutical...

Viridian Data Lift Prospects for Thyroid Eye Disease Drug
Viridian Therapeutics announced that its subcutaneous drug elegrobart met primary endpoints in a Phase 3 trial for chronic thyroid eye disease, showing 50%‑54% response rates versus 15% for placebo. The once‑monthly regimen also improved double vision in 61% of patients, while...

FDA Commissioner Defends Agency's Drug Approval Decisions After Wave of Backlash
FDA Commissioner Marty Makary faced intense media and political backlash after the agency rejected Replimune’s melanoma drug candidate. In a CNBC interview, Makary emphasized that three independent FDA panels reached the same safety conclusion and rejected accusations of bias toward...