STAT+: French Regulator Fines Novo and Lilly over Weight Loss Ad Campaigns
France’s medicines regulator ANSM imposed a $2 million fine on Novo Nordisk for misleading advertisements promoting its Wegovy and Saxenda weight‑loss drugs. Eli Lilly was also fined about $127 000 for an ad campaign that indirectly promoted prescription‑only Mounjaro. The actions reflect heightened regulator vigilance as GLP‑1 therapies dominate the obesity market. Both companies now face pressure to tighten their marketing compliance to avoid further penalties.
Cancer Patient Advocates Endorse Bill To Allow Reimbursement Of Trial Expenses
Cancer patient advocates, led by the American Cancer Society Cancer Action Network, have endorsed a new bipartisan Senate bill that would allow clinical‑trial sponsors to reimburse patients for travel and lodging costs. The legislation aims to dismantle geographic barriers that...

FDA Search for New CBER Head Focused on Small Group of Final Candidates
The U.S. Food and Drug Administration has narrowed its hunt for a new head of the Center for Biologics Evaluation and Research (CBER) to three or four finalists. CBER is the agency’s hub for overseeing vaccines, blood products, and emerging...

Celcuity Strengthens Case for ASCO-Spotlighted Breast Cancer Drug
Celcuity announced that its experimental PI3K/mTOR inhibitor gedatolisib achieved statistically significant and clinically meaningful disease‑progression delays in two‑ and three‑drug combinations for patients with PIK3CA‑mutated, hormone‑receptor‑positive, HER2‑negative breast cancer. The data will be presented at the ASCO meeting in Chicago...

Latus Bio Raises $97M to Expand Gene Therapy Pipeline
Latus Bio announced a $97 million Series A financing round led by 8VC and DCVC Bio. The funding will accelerate its gene‑therapy pipeline focused on delivering adeno‑associated virus (AAV) capsids that can reach deep brain structures at dramatically lower doses....

STAT+: Pharmalittle: We’re Reading About an Abortion Pill Controversy, Another UCB Deal, and More News
Two drugmakers, Danco Laboratories and GenBioPro, have petitioned the U.S. Supreme Court to restore mail‑order access to the abortion pill mifepristone after a lower court temporarily blocked deliveries. The appeal follows a series of lawsuits challenging the drug’s original 2000...

Choosing the Right CDMO for Long-Term Stability
Biotech firms must scrutinize contract development and manufacturing organizations (CDMOs) before committing to multi‑year projects. Dr. Patrick Meyer of Rentschler Biopharma outlines criteria such as transparent communication, accurate timelines, and a proven commercialization record. Technical expertise and a resilient supply chain...
Gene Therapy’s Evidence Problem—Lessons From Recent FDA Decisions
The FDA recently rejected REGENXBIO’s gene‑therapy candidate RGX‑121, citing an unvalidated biomarker as the primary endpoint and reliance on an external natural‑history control. The decision highlights a broader pattern of mixed regulatory outcomes for advanced therapeutics, with approvals like Sarepta’s...
Glowing Nanoparticles Exposed Hidden Cancer-Protein Behavior that Could Reshape Drug Screening
A Broad Institute team led by Sam Peng introduced upconverting nanoparticle probes that remain luminescent for minutes to hours, enabling continuous single‑molecule imaging of cancer‑related receptors in living cells. Using these probes, they captured real‑time dimerization dynamics of EGFR, HER2...

Protein Degraders Gain Speed as Arvinas Scores Landmark Approval
Arvinas received FDA approval for Veppanu, the first PROTAC therapy, marking a milestone for protein degraders. The drug showed modest benefit in a phase 3 trial, with the clearest effect in patients carrying ESR1 mutations, prompting the company to lay off...

Needle-Free Diabetes Care: 6 Devices that Painlessly Monitor Blood Sugar
Needle‑free glucose monitors are moving from research labs to commercial shelves, with six innovative devices highlighted for their non‑invasive approaches. Abbott’s FreeStyle Libre remains the market leader for interstitial sensing, while newcomers such as Occuity Indigo, D‑Pocket, Light Touch Technology,...

PCI Adds US Sterile Fill/Finish Capabilities Through $1B Global Expansion Drive
PCI Pharma Services announced a $1 billion global expansion, allocating $100 million to upgrade its San Diego campus with a high‑speed isolator line that will more than double its prefilled syringe capacity. The company also launched a GMP‑ready isolator vial and lyophilization line...

BioCryst Partners with Irish Affiliate of Neopharmed Gentili in ~$345M EU Commercialization Deal for Navenibart
BioCryst Pharmaceuticals has signed a licensing agreement with the Irish affiliate of Neopharmed Gentili, granting exclusive rights to commercialize its long‑acting plasma kallikrein inhibitor, Navenibart, across the European Union. The deal provides BioCryst with a $70 million upfront payment and potential...

Regulatory Submissions with Real-World Evidence: Successes, Challenges, and Lessons Learned - 09/23/2025
In a September 23, 2025 speech, FDA Principal Deputy Commissioner Sara Brenner outlined how real‑world evidence (RWE) has been used to support regulatory decisions and announced the agency‑wide FDA‑RWE‑ACCELERATE initiative. She highlighted recent examples from CDER, CBER and CDRH, and introduced Sentinel 3.0,...

First FDA ODAC Meeting without Pazdur Highlights Conceptual Challenges with Trial
The FDA’s Oncology Drugs Advisory Committee (ODAC) convened for the first time in nine months to review two AstraZeneca cancer therapies. The panel voted 6‑3 against approving camizestrant, an oral SERD for HER2‑negative advanced breast cancer, citing the trial’s failure...

Here's How Medication Abortion Works with Just One Drug That's Still Fully Available
A federal appeals court ordered the FDA to revert to in‑person prescribing rules for mifepristone, overturning the Biden administration’s telehealth flexibility. The decision takes effect nationwide, while the makers of mifepristone have appealed to the Supreme Court for a rapid...

Celcuity Reports the P-III (VIKTORIA-1) Trial Data on Gedatolisib Combination for HR+/HER2- PIK3CA Mutant Advanced Breast Cancer
Celcuity announced phase‑III VIKTORIA‑1 data showing that gedatolisib combined with fulvestrant, with or without palbociclib, achieved a statistically significant improvement in progression‑free survival (PFS) versus the alpelisib‑fulvestrant standard of care in HR+/HER2‑ advanced breast cancer patients harboring PIK3CA mutations. The...

Summit Shares Descend as PD-1/VEGF Asset Misses Early Survival Mark
Summit Therapeutics announced that its anti‑PD‑1/VEGF bispecific, ivonescimab, failed to meet the interim progression‑free survival threshold in the Phase 3 HARMONi‑3 trial for squamous NSCLC. The miss triggered a 26% plunge in Summit’s share price, closing at $16.12. An independent data...

Drugmaker AbbVie Chooses North Carolina for $1.4B Manufacturing Campus
AbbVie announced a $1.4 billion investment to build a 185‑acre manufacturing campus in Durham, North Carolina, targeting its immunology, neuroscience and oncology portfolios. The site will create 734 jobs over four years, incorporate AI‑driven production tools, and be operational by the...

UCB Captures Candid in $2.2B Autoimmune Deal as Pharma’s M&A Train Chugs Along
UCB announced a $2 billion acquisition of Candid Therapeutics, adding a pipeline of T‑cell engager drugs aimed at autoimmune and inflammatory diseases. The deal also includes up to $200 million in milestone payments and is expected to close by Q3 2026. Candid’s...

How a Vision-Restoring Gene Therapy Proved that We Can Treat Inherited Diseases
Luxturna, the first FDA‑approved gene‑augmenting therapy for inherited retinal disease, received the 2026 Breakthrough Prize after restoring sight to patients with Leber’s congenital amaurosis type 2. Developed by Spark Therapeutics founders Katherine High, Jean Bennett and surgeon Albert Maguire, the treatment...

OneSource Notches a Second Generic Semaglutide Approval in Canada, Through Partner
Canada approved its second generic version of Ozempic (semaglutide) through Apotex, which partnered with Orbicular Pharmaceutical Technologies and India‑based CDMO OneSource Specialty Pharma. OneSource handled end‑to‑end manufacturing, leveraging its FDA‑approved Bengaluru facility, while Orbicular led product development. The approval follows...

ARCHIMED to Acquire Esperion Therapeutics for ~$1.1B
Archimed announced a definitive agreement to acquire Esperion Therapeutics for up to $1.1 billion, taking the company private and delisting it from NASDAQ. Esperion shareholders will receive $3.16 in cash per share plus a contingent value rights (CVR) package worth roughly...

What Is Your AI Drug Repurposing Strategy Missing?
The article argues that AI‑driven drug repurposing for oncology often fails because models are fed fragmented, noisy data despite abundant datasets. It stresses that more data alone won’t improve outcomes; instead, high‑quality, curated, structured data—such as knowledge graphs linking genes,...

EU Backs Itvisma Gene Therapy for Spinal Muscular Atrophy
European regulators have issued a positive opinion on Itvisma, Novartis' onasemnogene abeparvovec gene therapy for spinal muscular atrophy (SMA). The Committee for Medicinal Products for Human Use (CHMP) recommended marketing authorisation on 23 April 2026, pending final approval by the European Commission....

Rhythm Pharmaceuticals Reports the EC Approval of Imcivree (Setmelanotide) for Acquired Hypothalamic Obesity
Rhythm Pharmaceuticals announced that the European Commission has granted marketing authorization for Imcivree (setmelanotide) to treat acquired hypothalamic obesity in patients aged four years and older. The approval is based on the Phase III TRANSCEND trial, which enrolled 120 participants...
Novo Nordisk to Launch Ozempic for Type 2 Diabetes in US
Novo Nordisk will launch an oral version of Ozempic in the United States, following FDA approval that marks the first oral GLP‑1 drug cleared for both blood‑sugar control and cardiovascular risk reduction. The tablets, available in 1.5 mg, 4 mg and 9 mg...

Arvinas and Pfizer Report the US FDA Approval of Veppanu (Vepdegestrant) for ESR1-Mutated Breast Cancer
The U.S. FDA has approved Veppanu (vepdegestrant) for adults with ESR1‑mutated ER+/HER2‑ advanced or metastatic breast cancer who have received at least one line of endocrine therapy. The approval follows the Phase III VERITAC‑2 trial, which compared Veppanu to fulvestrant...

65 % of Eligible Lung Cancer Patients Do Not Receive the Most Appropriate Targeted Therapies, Diaceutics Report Finds
Diaceutics’ 2026 Clinical Practice Gaps report shows that 65% of U.S. patients with advanced non‑small cell lung cancer still miss the most appropriate targeted therapy, a figure unchanged since 2019. While biomarker testing has improved, the biggest loss now occurs...

FDA Action Alert: Argenx, AstraZeneca/Daiichi Sankyo, Biogen/Eisai and Cingulate
May’s FDA docket is light, but the decisions on four high‑profile drugs could reshape market dynamics. Argenx is seeking to broaden Vyvgart’s label to seronegative myasthenia gravis patients, potentially adding 11,000 new users. AstraZeneca and Daiichi Sankyo aim to secure...

Queensland Funds Research Into Cell-Based Therapies for Traumatic Brain Injury
The Queensland Government has pledged A$5.5 million (about US$3.6 million) over three years to the Cure TBI initiative, a research programme focused on cell‑based therapies for traumatic brain injury. Backed by the National Injury Insurance Scheme, Queensland (NIISQ), the effort will be...
Low-Dose Drug Cuts Breast Density up to 26% with Fewer Side Effects
A Karolinska Institutet study found that low‑dose endoxifen, the active metabolite of tamoxifen, reduces mammographic breast density by up to 26%—comparable to the 18.5% reduction seen with standard 20 mg tamoxifen—while causing far fewer serious side effects. In a randomized, placebo‑controlled...

ADC Therapeutics SA (ADCT) Q1 2026 Earnings Call Transcript
ADC Therapeutics reported Q1 2026 product revenue of $15.8 million, a decline from the prior year, while GAAP net loss narrowed to $41 million ($0.30 per share). Operating expenses fell 12.1% on a non‑GAAP basis, driven by lower R&D spend. The company...

Travere Therapeutics Inc (TVTX) Q1 2026 Earnings Call Transcript
Travere Therapeutics reported record commercial performance for FILSPARI, posting $322 million in 2025 sales—a 144% year‑over‑year increase—and a Q4 record of 908 new patient start forms. The company secured a new FDA PDUFA target date of April 13, 2026 for the FSGS indication...

NHS Cancer Jab Could Save Patients Hours in Hospital
NHS England is introducing an injectable form of Keytruda, the blockbuster immunotherapy, that can be given in one to two minutes instead of the traditional hour‑long infusion. About 14,000 cancer patients in England start Keytruda each year, and most are...

Re: Limit Use of Nasal Decongestant Sprays to Five Days, UK Regulator Says
The UK Medicines and Healthcare products Regulatory Agency (MHRA) has issued new guidance limiting the use of over‑the‑counter nasal decongestant sprays containing xylometazoline or oxymetazoline to a maximum of five days. The advisory reiterates long‑standing clinical warnings that prolonged use...

Editorial. Costly Remedy
Sun Pharma announced an $11.75 billion acquisition of Organon, a U.S. pharma firm with roughly $6.2 billion in annual revenue. The deal comes as the Trump administration prepares 100% tariffs on certain patented drugs, pressuring generic exporters to secure domestic supply chains....

Liquid Biopsy Predicts Response to Breast Cancer Immunotherapy
Researchers at Vanderbilt-Ingram Cancer Center demonstrated that serial liquid biopsies analyzing peripheral blood RNA can predict response to pembrolizumab in high‑risk early‑stage HER2‑negative breast cancer. The study examined 546 blood samples from 160 patients in the I‑SPY2 trial, showing transcriptional...

The Biosimilar Market Isn’t Failing, It’s Finding Its Purpose
The biosimilar market is not collapsing; it is transitioning from a price‑driven scramble to a purpose‑focused strategy. Recent HHS data show that markets become uneconomic once five competitors vie for the same molecule, prompting a shift toward diversified product portfolios....
Blocking a Cellular Inflammation Process Could Result in Effective Therapy for Pancreatic Cancer
Scientists at The Wistar Institute and ChristianaCare identified a vulnerability in pancreatic cancer where defective mitochondria release double‑stranded RNA, triggering the TLR3/TRAF6 inflammatory pathway. The tumor cells become dependent on this inflammation for growth and survival, and blocking the pathway...

Stopping and Restarting Certain GLP-1s to Lose Weight May Make the Drug Less Effective
A preclinical study from the University of Pennsylvania found that stopping and restarting GLP‑1 weight‑loss drugs, such as semaglutide, markedly diminishes their efficacy. Overweight mice on a stop‑and‑start regimen regained weight during off periods and never recaptured their initial loss,...

This Overlooked Mineral May Play A Role In Protecting Against Alzheimer’s
Physician‑scientist David Fajgenbaum highlights emerging evidence that lithium, a long‑used mood stabilizer, may protect against Alzheimer’s disease. Human post‑mortem studies show lower lithium in the prefrontal cortex of patients with mild cognitive impairment, while mouse experiments demonstrate that dietary lithium...

The Bias in Medical Research: Africa Carries a Huge Disease Burden but Is Missing From Clinical Trials
A new analysis of 2,472 randomized controlled trials published between 2019 and 2024 reveals a stark under‑representation of Africa in top medical research. Only 3.9% of trials in the most prestigious general journals were conducted exclusively on the continent, and...

Scientists Cautiously Suggest GLP-1s Are Safe to Use Around Pregnancy
A systematic review of more than 49,000 pregnancies over two decades found that exposure to GLP‑1 receptor agonists such as Ozempic, Wegovy and Mounjaro around conception does not increase the risk of major birth defects. The meta‑analysis of ten cohort...
Development Analysis and Strategic Insights From a 505(b)(2) Reformulation Product: Enzalutamide
Astellas used the 505(b)(2) pathway to reformulate enzalutamide (XTANDI®) from a 40 mg lipid‑filled soft capsule into 40 mg and 80 mg film‑coated tablets, aiming to cut pill burden. Five biopharmaceutic studies demonstrated comparable AUC between the two dosage forms, while the tablet...

'Build AI that Can Accurately Represent the Full Complexity of Biology': Mark Zuckerberg Wants to Cure All Diseases but Needs...
Meta billionaire Mark Zuckerberg is channeling $500 million into Biohub’s Virtual Biology Initiative to amass massive cellular datasets for AI modeling. The funding splits into $100 million for global data collection and $400 million for advanced imaging and engineering tools. Partnerships with the...
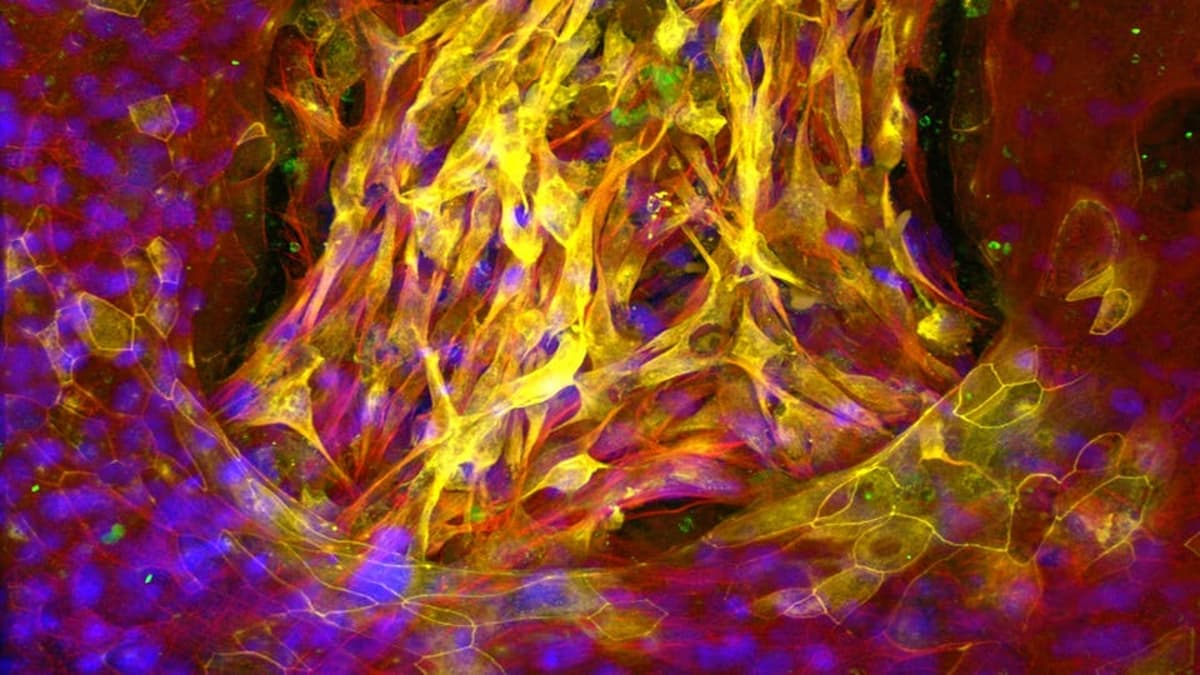
Human Organ Chip Systems Reshape Drug Development
Harvard’s Wyss Institute, led by Dr. Donald Ingber, has spent over a decade perfecting Human Organ Chip systems that mimic organ-level functions in a thumb‑drive‑sized device. Recent FDA and NIH policy shifts endorse these chips as viable alternatives to animal...

Could Ozempic Help With Alzheimer’s Disease? Scientists Are Taking a Closer Look
A new Anglia Ruskin University review of 30 preclinical studies suggests GLP‑1 drugs such as Ozempic and Wegovy can lower amyloid‑beta and tau, the hallmark proteins of Alzheimer’s disease. Twenty‑two studies reported reduced amyloid‑beta and nineteen showed decreased tau, with liraglutide...
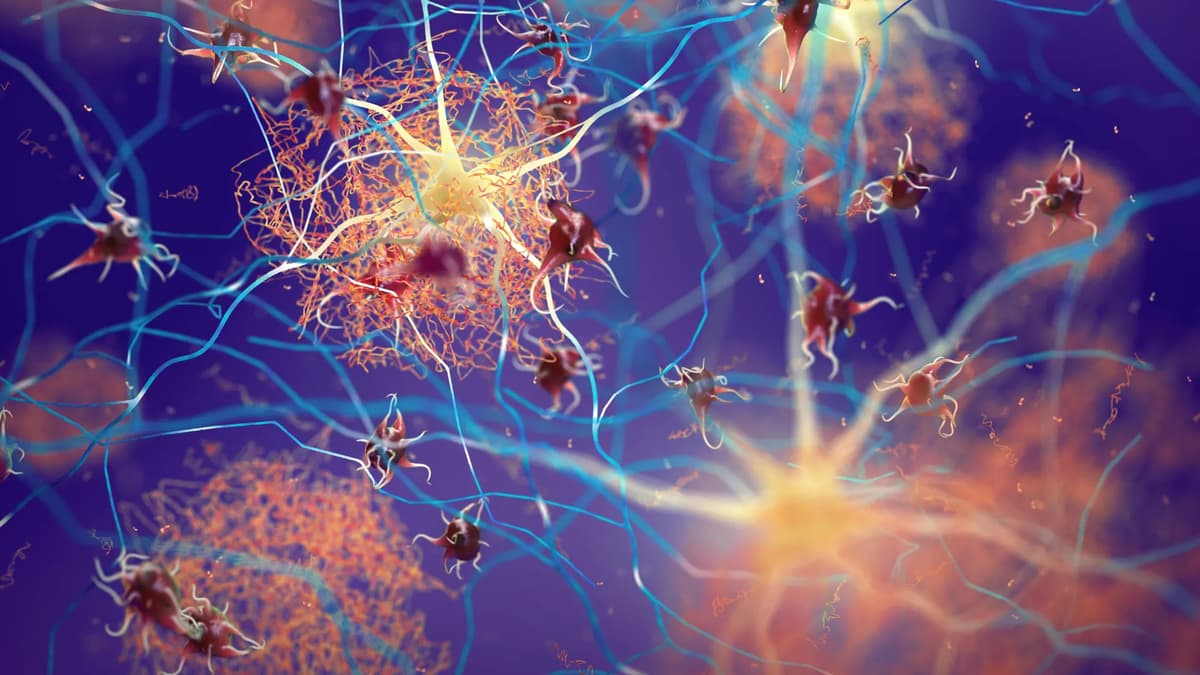
Boosting One Protein Helps the Brain Fight Alzheimer’s
Researchers at Baylor College of Medicine discovered that increasing the protein Sox9 in astrocytes enables mouse models of Alzheimer’s disease to clear existing amyloid plaques and retain memory performance. The study, published in Nature Neuroscience, showed that elevated Sox9 enhances...

Your Next Dog May Live Longer
Longevity biotech Loyal, founded by Celine Halioua, has received FDA clearance to market a daily pill that improves insulin sensitivity and could extend dogs’ lifespans. The agency deemed the drug likely effective based on a small study of about 50...