Know What's Happening in Healthcare
Know What's Happening in Healthcare

FDA approves KRESLADI, first CIRM‑backed gene‑editing therapy
The FDA cleared KRESLADI, a gene‑editing treatment from Rocket Pharmaceuticals for severe leukocyte adhesion deficiency‑I in children, eliminating the need for a bone‑marrow donor. It is the first product directly funded by California’s Institute for Regenerative Medicine to receive approval.
Also developing:
By the numbers: Syneron Bio raises $150M Series B
Kyowa Kirin announced it will cease all clinical trials of rocatinlimab, an anti‑OX40 antibody once touted as a potential eczema blockbuster, after a safety review identified emerging malignancy signals. The review, conducted with former partner Amgen, uncovered a confirmed and a suspected case of Kaposi’s sarcoma, raising concerns about the drug’s mechanism. Although the incidence is below background rates, the company concluded the risks could outweigh benefits and will halt studies after participants finish safety follow‑up. The decision follows Amgen’s earlier relinquishment of rights and mixed results for similar OX40 drugs.

Hello Heart, a digital heart‑health platform, announced a strategic collaboration with the American College of Cardiology (ACC). The ACC will convene an independent clinician workgroup to evaluate Hello Heart’s monitoring technology, coaching tools, clinician reports, and EMR integrations. Hello Heart...

Real‑world evidence from Duke’s PRECISION platform shows Pluvicto (Lu‑177 vipivotide tetraxetan) delivers a median progression‑free survival of 13.5 months in PSMA‑positive metastatic castration‑resistant prostate cancer (mCRPC) after androgen‑receptor pathway inhibitor (ARPI) therapy, matching the pivotal PSMAfore trial. Patients who received Pluvicto...

KeyCare announced a $27.4 million financing round led by HealthX Ventures, bringing its cumulative capital to over $55 million. The Epic‑integrated virtual‑care platform connects health systems with independent providers across primary, behavioral, and urgent care. Funds will be used to scale operations,...

AMITA Health St. Joseph Hospital in Elgin, Illinois tackled chronic understaffing in its emergency department by adopting General Devices' GD Solution Suite. The digital platform replaced paper EMS forms, cutting the department’s annual form‑related expenses by $8,000. By eliminating lengthy...

Small language models (SLMs) are gaining attention in the generative AI field. SLMs are trained on relatively small amounts of data and have a relatively small number of parameters. SLM can be more practical than large language models as they are...

The FDA granted priority review to Takeda’s rusfertide, a first‑in‑class hepcidin mimetic for polycythemia vera, after Phase III trials more than doubled response rates. A regulatory decision is expected in Q3 2026. The agency also expanded Novo Nordisk’s once‑weekly Sogroya to treat children...

Elsevier announced major upgrades to ClinicalKey AI, adding full‑text content from more than 130 premium journals and leading medical societies such as The Lancet, NEJM, and the American College of Cardiology. The platform now offers real‑time traceability, linking each answer...

Fresenius Kabi issued a Class I recall for its Ivenix large‑volume infusion pumps after a software flaw was discovered that misreports battery capacity and can freeze the user interface. The FDA linked the defect to two serious injuries reported in November,...

Boston Scientific’s Farapulse pulsed field ablation system received a CE‑mark label expansion in Europe, now covering persistent atrial fibrillation lasting at least seven days. The decision follows positive safety and efficacy data from the ADVANTAGE AF trial. The move mirrors...

Recent cyber incidents highlight how a single foothold can expose tens of millions of records. The Conduent Business Services breach grew to over 25 million victims, with attackers retaining access for nearly three months and exfiltrating 8.5 TB of data. Parallel attacks...

The Peterson Health Technology Institute evaluated five virtual gastrointestinal platforms, separating them into wraparound and clinician‑led models. Wraparound solutions such as Cylinder and Digbi deliver nutrition and behavioral support that improve IBS symptoms and reduce commercial spending by $1,889 per...

The FDA’s Drug Competition Action Plan (DCAP) continues to drive generic drug competition by streamlining standards for complex products, closing loopholes that allow brand‑name companies to delay approvals, and improving the overall ANDA review process. Aligned with GDUFA III, the plan...

This week on Lifers, I interviewed @othman about how he navigated @color through three massive pivots—from cancer genetics to national COVID infrastructure and now a virtual cancer clinic. Timestamps: (00:00) Preview (00:58) Intro (01:03) Lessons from Figma and betting on the tortoise over the...
Pierre Fabre seeks to revive US approval chances for spurned cell therapy https://t.co/gJeklT8asd @ByJonGardner $ATRA $QURE $RGNX
Researchers have engineered a nanocomposite incorporating silver (Ag) nanoparticles that markedly improves anticancer efficacy. The hybrid material boosts reactive oxygen species generation and enhances tumor cell uptake while sparing normal tissue. In vitro studies show synergistic activity with standard chemotherapeutics,...

Did the FDA’s individualized therapy guidance put a spike in the heart of a startup biotech? Fake news, says the HHS. Yeah, it did, says an insider. Exclusive from @RLCscienceboss and @Jared_Whitlock https://t.co/IN9o4VIp03

The FDA has approved the Teal Wand, the first at‑home cervical cancer screening device, now available nationwide for women ages 25‑65 at average risk. The wand collects a dry cervical sample at home, which is mailed to a lab for...

The Medical Technology Group’s new report reveals that 42 Integrated Care Boards (ICBs) lack basic data on gynaecology pathways, waiting times, diagnoses and outcomes. More than 750,000 women are currently waiting for hospital gynaecology services, yet most ICBs do not...
A new systematic review of LLMs in medicine @NatureMedicine @ekoermann https://t.co/npvDeqUvel “Despite thousands [4,609] of publications since late 2022, only a small fraction use real clinical data and just 19 randomized trials exist.”
New integrated safety data from 20 trials confirm that ruxolitinib 1.5% cream delivers a low incidence of serious infections, cardiovascular events, thromboembolic events, and malignancies across atopic dermatitis, vitiligo and other inflammatory skin conditions. A phase 3b trial in adults with...

A non‑randomized study of the ISHI Health AI‑enabled virtual heart‑failure platform showed significant gains in guideline‑directed medical therapy (GDMT) and improved weight stability among 747 patients across six community cardiology practices. The system collected remote biometric data, generated risk‑graded alerts,...

MedDream will demonstrate its cloud‑native, AI‑ready universal DICOM viewer, built on AWS HealthImaging, at HIMSS 2026 in Las Vegas. The solution leverages AWS’s HTJ2K transfer syntax to stream large imaging datasets quickly and securely. It supports radiology, cardiology, ophthalmology, dental...

The FDA has published a batch of newly added guidance documents, spanning drug exclusivity, post‑approval safety reporting, real‑world data studies, Bayesian trial methods, and patient‑preference research. Ten documents are highlighted, with six in draft form and four finalized, dated between...
A new Nature Metabolism study identifies extracellular matrix protein ECM1 as a pivotal early regulator of kidney fibrosis. Global ECM1 knockout mice develop spontaneous fibrosis and premature death, while AAV9‑mediated ECM1 knockdown in fibroblasts markedly lessens fibrotic burden. Mechanistically, ECM1...

Pivot Point Consulting has teamed with Microsoft’s Rural Health Resiliency Program to roll out the Dragon Copilot AI assistant to financially strained rural hospitals. Microsoft is providing a 60% discount off the standard price, while Pivot Point offers free readiness...

Cleveland Clinic CIO Sarah Hatchett will lead a HIMSS26 panel that offers a practical roadmap for building new IT infrastructure and retrofitting existing hospital spaces to accommodate emerging technologies. The session targets health‑system leaders seeking concrete steps to transition toward...

A MEDSIR‑hosted webinar titled “The Future is Collaborative: Transforming Clinical Trials” examined how collaborative‑initiated trials and investigator‑initiated trials (IITs) are reshaping oncology research. Speakers Dr. Javier Cortés and Dr. Antonio Llombart‑Cussac, both leading breast‑cancer oncologists, discussed their roles in recent...

Researchers applied Bayesian machine‑learning to uncover risk factors for hepatosplenic multimorbidity in schistosomiasis. By fusing clinical records, environmental exposure data, and host genetic markers, the model pinpointed predictors such as specific immune‑gene variants and poor sanitation. Its capacity to continuously...
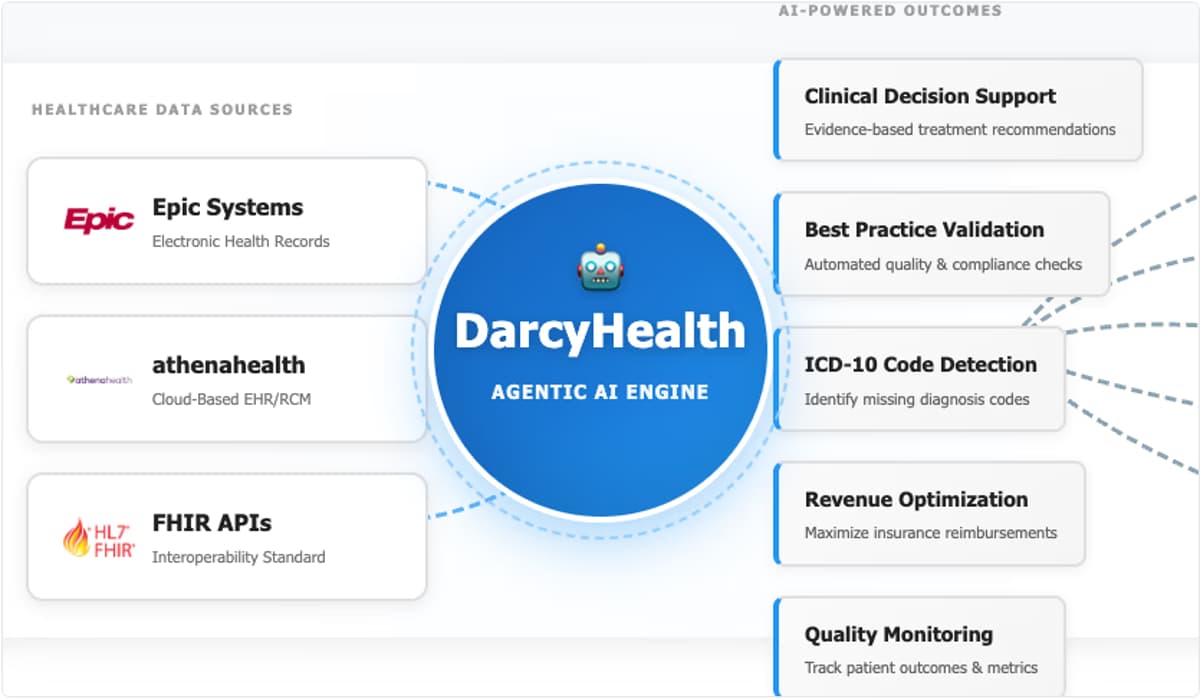
DarcyIQ, an AI‑powered revenue acceleration platform from Innovative Solutions, has launched native, HIPAA‑aligned integrations with Epic and athenahealth’s athenaOne using Model Context Protocol. The conversational layer lets clinicians and revenue teams interact with EHR data via natural language, eliminating platform...

The FDA issued a warning letter to MaxLife Technologies for false and misleading claims on its website about compounded semaglutide and tirzepatide products. The company labeled the drugs as “generic compounded medication” and implied FDA approval and that MaxLife was...

UK independent provider Ascenti Physio has launched its Ascenti Reach digital physiotherapy app in a six‑month pilot with York and Scarborough Teaching Hospitals NHS Foundation Trust. Early data from other users show wait times can be cut by up to...

Researchers at Karolinska Institutet reported that phosphorothioate antisense oligonucleotides (ASOs) directly bind key DNA‑repair enzymes, forming nuclear condensates that trigger a false DNA‑damage response. The effect was observed at concentrations commonly used in laboratory assays, though clinical doses reach lower...

Sterling Heights Fire Department launched a peak‑demand EMS unit staffed by EMTs to handle low‑acuity 911 calls, freeing paramedics for high‑severity incidents. The initiative pairs with a new five‑color dispatch system that prioritizes calls by medical urgency. Since its October...

Medicare’s 2026 Physician Fee Schedule introduces outcome‑based payments for cardiac care, rewarding providers who prevent ER visits, readmissions, or accelerate recovery. The rule also replaces the in‑person supervision requirement for the technical component of remote cardiac monitoring with virtual oversight,...

Medbridge has launched an Occupational Health Pathway within its Medbridge One Care platform, offering a hybrid (live and virtual) model to prevent musculoskeletal (MSK) injuries among industrial workers. The solution targets three high‑risk groups—production and material handlers, maintenance staff, and...

Anterior has embedded its clinician‑led AI reasoning engine into HealthEdge’s GuidingCare platform, which serves more than 70 health plans and 30 million members. The integration places AI directly inside utilization management workflows, eliminating the need for separate tools. Early production data...

An international panel of 147 oncology leaders, including Prof. John Bridgewater, issued a consensus calling for sweeping reforms in the diagnosis, treatment, and research of cholangiocarcinoma (bile duct cancer). The disease remains deadly, with three‑quarters of patients dying within a...

Postpartum depression (PPD) can surface months after birth, with research showing onset as late as 12 months postpartum. While the DSM‑5 limits PPD to the first four weeks, clinicians often treat any depression arising within the first year as postpartum‑related....

The American Medical Association’s Journal of Ethics published its final issue in December 2025, ending a 26‑year legacy of scholarly oversight. Simultaneously, the AMA launched a Center for Digital Health and Artificial Intelligence, signaling a strategic pivot toward technology. The...

Philadelphia’s biotech ecosystem, long known for CAR‑T breakthroughs, is diversifying into obesity, metabolic disease, gene editing, and solid‑tumor immunotherapies. Nine local companies illustrate this shift, from Alveus Therapeutics’ $197 million Series A obesity program to Cabaletta Bio’s autologous CD19 CAR‑T for autoimmune...
Multi‑cancer early detection (MCED) tests analyze blood, urine or saliva for tumor‑derived DNA, RNA or proteins, promising a single‑sample screen for dozens of cancers. While early‑stage detection could complement established organ‑specific programs, sensitivity varies widely, with some cancers identified in...
T-cell engagers for autoimmunity. Listen to Ken Song describe the opportunity as CEO of San Diego-based Candid Therapeutics. Sponsored by @AlphaSenseInc & Dash Bio. https://t.co/Ct2viH00c4

At ZohoDay 2026, Newcross Healthcare showcased how it built a mission‑critical care‑management application on Zoho’s governed platform, unifying CRM, finance, workforce and analytics. By leveraging Zoho Creator, the team delivered a fully integrated solution in six months, cutting licensing costs...

Cardiovascular disease remains the top global killer, accounting for 19.8 million deaths in 2022, with 85% linked to myocardial infarction or stroke. Echocardiography is essential for detecting ischemic cardiomyopathy, wall‑motion abnormalities, and left‑ventricular remodeling. Coronary artery bypass grafting (CABG) is a...

February 2026 saw a surge of biosimilar activity, with key regulatory approvals for products such as Sandoz Enzeevu (Eylea), Accord Filkri (Neupogen), and STADA Gotenfia (Simponi) across the US, Canada and the EU. Companies forged new licensing and commercialization deals that extend biosimilar...
What is the diagnostic yield of MRI in adult emergency patients with dizziness or vertigo? While MRI is efficient for detecting central causes of dizziness, its yield is low in cases of isolated dizziness without neurological signs. 👇 https://t.co/170qBbHrq7

Quell Therapeutics has launched the phase 1/2 CHILL trial of its autologous CAR‑Treg therapy QEL‑005 in rheumatoid arthritis and systemic sclerosis, after pausing the liver‑transplant program QEL‑001. The multi‑center study will enroll patients in the UK, Germany and Spain, with read‑out...

NHS England has set a firm deadline to retire the CIS1 authentication service, removing access on 28 February 2027 after reducing its SLA to silver on 1 October 2025. The move forces NHS trusts and other European hospitals to adopt the newer CIS2 platform,...

RecovryAI, emerging from stealth, received FDA Breakthrough Device Designation for its physician‑prescribed Virtual Care Assistants (VCAs), AI tools that guide patients through post‑operative recovery. The designation, reserved for devices that can substantially improve care standards, accelerates the company’s engagement with...