
Mount Sinai Introduces the Marilyn Monroe Mental Health Initiative for Arts Professionals
Mount Sinai Health System has launched the Marilyn Monroe Mental Health for the Arts Program, a $100,000 initiative aimed at the unique psychological challenges of performing artists. Housed in the Samuel J. Friedman Health Center in New York’s theater district, the program combines Mount Sinai’s clinical expertise with the Entertainment Community Fund’s social‑service network. Led by Dr. Shilpa R. Taufique and psychiatrist Dr. Nicholas Kopple‑Perry, it offers onsite consultations, emergency care, and training for social‑work staff. The effort honors Monroe’s centennial and sets a template for arts‑focused mental‑health care.

4 Types of Drugs that May Increase Your Dementia Risk
A recent analysis highlights four medication classes—anticholinergic antihistamines, antipsychotics, benzodiazepines, and proton‑pump inhibitors (PPIs)—that may elevate dementia risk, especially with chronic use. Observational studies suggest antihistamines could increase risk by roughly 50%, while antipsychotics and benzodiazepines show mixed but concerning...
Innovative Nanoparticle Technique Advances Early Detection of Pancreatic Cancer
Researchers have unveiled a novel nanoparticle‑based imaging agent that markedly improves early detection of pancreatic cancer. In pre‑clinical trials the probe identified tumors as small as 2 mm, delivering a 30% sensitivity boost over conventional CT scans. The technology earned FDA...

Ensign CEO: Nursing Home Demand Is Strong Despite Fluctuating Volume, Payer Scrutiny
Ensign Group reported record occupancy and an 18.4% year‑over‑year revenue jump to $1.33 billion in Q1, underscoring strong demand for skilled nursing despite fluctuating patient volumes and heightened payer scrutiny. The company highlighted robust growth in managed‑care and Medicare censuses, up...

Oura Unveils Menopause Insights, Birth Control Tools, US Open Partnership
Oura introduced two new hormonal health features for its Ring: a birth‑control support tool embedded in the Cycle Insights suite and a Menopause Insights module that uses a proprietary questionnaire and biometric data to assess symptoms. The company also sealed...

Confluent Medical Technologies Hires New Chief Operating Officer
Confluent Medical Technologies announced the appointment of Tom Testa as chief operating officer, effective April 29. Testa brings a track record that includes serving as CEO and CFO of Corza Medical and as VP and general manager of Nordson Medical’s...

OIG Report: 73% of Nursing Homes Face Emergency Power Failures, Many Lack Backup Systems
An Office of the Inspector General audit found that 73 percent of the United States’ more than 15,000 nursing homes operate with inadequate or unreliable emergency power. The review of 100 Medicare‑ and Medicaid‑certified facilities uncovered 119 deficiencies, including 40...
Man Dies Covered in Necrotic Lesions After Amoebas Eat Him Alive
A 78‑year‑old man developed widespread necrotic skin lesions that were ultimately traced to a disseminated infection by the free‑living amoeba Acanthamoeba. The pathogen likely entered through nasal rinsing with tap water, a route rarely linked to full‑body disease in patients...

Validated Quality Signals Will Drive Digital Health Adoption
The Digital Medicine Society’s DiMe Seal and the CARIN Code of Conduct are being adopted by CMS to highlight high‑quality digital health apps for Medicare beneficiaries. By attaching these validated quality signals to apps, CMS can surface trustworthy solutions amid...

The Biopharm Brief: Microbiome Fast Track, Leo Pharma Gene Therapy Deal, AAV Automation Advances
The FDA has awarded Fast Track designation to a live biotherapeutic aimed at restoring gut microbiota in moderate‑to‑severe ulcerative colitis, accelerating its development path. Leo Pharma is buying Replay’s gene‑therapy program to broaden its rare‑dermatology portfolio, marking a shift toward...

DiMe and CARIN Alliance Helping Apps Get Into the Medicare App Library
DiMe (Digital Medicine Society) and the CARIN Alliance are collaborating with the Centers for Medicare & Medicaid Services (CMS) to define security and trust standards for apps seeking inclusion in the Medicare app library. The partnership aims to create a...

Stroke Prevention Devices for TAVR Fail to Make an Impact
A new meta‑analysis of eight randomized trials involving more than 11,000 transcatheter aortic valve replacement (TAVR) patients found that cerebral embolic protection devices (CEPDs) did not significantly lower overall, disabling, or non‑disabling stroke rates. Roughly half of the participants received...

High Contrast, Low Risk: Black Marking for Medical Technology
Ultrashort‑pulse laser black marking has become a viable solution for UDI compliance on medical‑grade stainless steel, delivering permanent, high‑contrast codes despite polished surfaces and tiny marking areas. FOBA’s F.0100‑ir system creates nanostructured “light traps” that produce matte black marks with...

DOJ Forms Health Care Fraud Strike Force
The U.S. Department of Justice has launched the West Coast Health Care Fraud Strike Force, a multi‑district initiative that brings together the DOJ’s Health Care Fraud Section, the U.S. Attorney’s Offices in Arizona, Nevada and Northern California, the FBI, the...

Artificial Intelligence for Drug Development
The FDA’s Center for Drug Evaluation and Research (CDER) is formalizing its AI strategy through a draft 2025 guidance, an AI Council created in 2024, and extensive stakeholder engagement. CDER has reviewed more than 500 AI‑enabled drug submissions since 2016...

External Engagements with FDA for Artificial Intelligence in Drug Development
The FDA has published a comprehensive menu of early‑engagement options for sponsors leveraging artificial intelligence across the drug development lifecycle. Sponsors can request formal meetings for IND or pre‑IND programs, or use targeted programs such as C3TI for trial design,...

Regulatory and IP Considerations for Veterinary Drugs in Canada
The article outlines Canada’s regulatory framework for veterinary drugs, separating them from other animal health products and detailing the data‑protection, patent‑linkage, and supplementary‑protection regimes that apply only to veterinary drugs. It explains submission requirements, labeling rules, priority‑review criteria, and biowaiver...

Arvinas’ ‘Protac’ Breast Cancer Drug Cleared by FDA
The FDA approved Arvinas and Pfizer’s Veppanu, a PROTAC drug, as a second‑line therapy for metastatic ER‑positive, HER2‑negative breast cancer with ESR1 mutations. Veppanu is the first protein‑degrading medicine cleared in the U.S., targeting estrogen receptors for destruction. While the...

Techcyte Raises $15M to Expand AI Diagnostics Platform
Techcyte closed a $15 million funding round led by Van Tuyl Companies to scale its AI‑powered Fusion platform. The round brings strategic partners such as Zoetis and the Mayo Clinic, granting access to a 17 million‑slide dataset. Fusion integrates anatomic and clinical pathology,...

UniQure Pushes Forward As FDA Rare Disease Controversies Continue
UniQure, the gene‑therapy pioneer at the heart of FDA Commissioner Marty Makary’s rare‑disease controversy, is pressing forward with a formal request for its Huntington’s disease treatment. The company will present data at a Type B meeting with the FDA in the...
RFK Jr. Wants Stakeholders To Show Real-Time Prices Ahead Of Final Regulation
HHS Secretary Robert F. Kennedy Jr. is urging health‑care providers and health‑IT firms to deploy Real‑Time Pharmacy Benefit Technology (RTPBT) immediately, rather than waiting for formal rulemaking. RTPBT enables patients to see the exact out‑of‑pocket cost of prescription drugs at...

The World Wants to Eliminate Cervical Cancer - How Australian Scientists Led the Way
Australian scientists pioneered the Gardasil HPV vaccine, enabling the world’s first national vaccination programme in 2007 and positioning Australia to eliminate cervical cancer by 2035. The combined strategy of >80% vaccination of adolescents, HPV‑based screening every five years, and self‑sampling...

South Carolina Measles Outbreak Declared over; Utah Reaches 625 Cases
South Carolina’s measles outbreak, which infected 997 people, was declared over after a 42‑day stretch with no new cases, meeting the CDC’s containment threshold on April 26. During the crisis, statewide MMR vaccinations jumped 31.1% and Spartanburg County saw a 93.6%...
DNA-Containing Extracellular Vesicles Boost Antitumor Responses in Mice
Researchers at Weill Cornell Medicine discovered that extracellular vesicles released by activated T cells contain DNA that can be transferred to dendritic and tumor cells, enhancing antigen processing and presentation. In mouse models of glioblastoma, pancreatic and triple‑negative breast cancer,...

Rare Fatal Arrhythmias After PFA Highlighted in MAUDE Analysis
The TiFFANY analysis of FDA MAUDE reports identified fatal arrhythmic events after pulsed‑field ablation (PFA) for atrial fibrillation at roughly 3.2 per 100,000 procedures, translating to a mortality rate of 17.8 per 100,000—about twice that of thermal ablation. Overall, PFA showed a...

Advocates Sound Alarm Over Nebraska Starting Medicaid Work Reqs
Nebraska began enforcing the One Big Beautiful Bill Act’s Medicaid work requirements on May 1, earlier than many states. Advocates warn that the rollout suffers from insufficiently trained staff, an incomplete exemption list, and confusing outreach to beneficiaries. The state’s hurried...

Noninvasive Proton Beam Therapy Safe for VT Ablation: Early Series
A first‑in‑human feasibility study at Mayo Clinic showed that noninvasive proton‑beam radioablation can be delivered safely to patients with refractory ventricular tachycardia (VT). Seven high‑risk patients underwent meticulous imaging‑guided planning and received a single proton dose, with no acute cardiac...
Obamacare Enrollment Drops Sharply as Costs Rise
Enrollment in the Affordable Care Act marketplaces has plunged sharply after Congress let the enhanced subsidies lapse at the end of 2025. Industry analysts estimate a 20% drop, taking coverage from roughly 24 million to about 19 million, with some forecasts suggesting...

The Next Alzheimer’s Breakthrough Will Take More Than Just Science
Alzheimer’s research has moved from theory to treatment as anti‑amyloid antibodies like Lecanemab and Donanemab receive regulatory approval and begin reaching patients. These drugs can clear existing amyloid plaques and modestly slow cognitive decline, extending the disease trajectory from roughly...

Health Equity & Access Weekly Roundup: May 1, 2026
A House Ways and Means hearing exposed deep flaws in Medicare’s fraud‑prevention system, prompting calls for stricter provider verification and monthly claim statements. The Commonwealth Fund’s 2026 report highlighted persistent racial and ethnic health disparities, warning that Medicaid work‑requirement rollouts...
Restoring Protein Recycling Reverses T-Cell Exhaustion in Mice
Scientists at UC San Diego discovered that impaired protein recycling drives T‑cell exhaustion in mice. Restoring the activity of specific E3 ligases—NEURL3, RNF149, and WSB1—reestablished proteostasis, cleared misfolded proteins, and revived T‑cell anti‑tumor function. The findings, published in Cell, suggest...

CMS Launches Clinical Laboratory Fee Schedule Reporting Module for Data Collection
The Centers for Medicare & Medicaid Services (CMS) has launched a Clinical Laboratory Fee Schedule (CLFS) reporting module to collect private‑payor rate data. CMS provided a quick‑reference guide, an FAQ on the Protecting Access to Medicare Act (PAMA) reporting, and...

Xavier Becerra Backpedals on Single Payer as He Woos Powerful Doctors’ Lobby
Democratic gubernatorial hopeful Xavier Becerra has softened his long‑standing advocacy for a single‑payer health system after securing the endorsement of the California Medical Association, the state’s most powerful doctors’ lobby. In a private meeting, Becerra told CMA leaders he is...

Registration Opens for Virtual CMS Event on Interoperability
The Centers for Medicare & Medicaid Services (CMS) opened registration for a virtual event focused on health‑care interoperability, inviting over 1,000 hospital and health‑system leaders. The event will showcase a new CMS‑FDA collaborative pathway designed to accelerate market entry for...

A Husband and Wife Escaped From a Locked Memory-Care Unit. He Solved the Door Code Just by Listening.
In March 2020, a husband and wife living in the Elmcroft memory‑care unit in Lebanon, Tennessee, escaped by using the numeric exit code the husband had learned by listening to staff enter it on a keypad. The man, a former...
:max_bytes(150000):strip_icc():format(jpeg)/brain-shivers-as-effexor-withdrawal-symptom-1065516-final-01-32cb22b5df7e4d3b98654cba8d8a81a9.png)
What to Expect From Effexor Withdrawal
The article explains what patients can expect when stopping Effexor (venlafaxine), detailing a typical withdrawal timeline that begins within 24‑48 hours and often resolves in three weeks. It lists common physical and psychological symptoms, including nausea, dizziness, mood swings, and the...

Senators Reintroduce Bipartisan Legislation To Enhance Vetting Of Senior Care Staff
Senators reintroduced bipartisan legislation that would allow nursing homes to tap the National Practitioner Data Bank for criminal background checks on prospective caregivers. The bill also loosens current CMS rules, enabling facilities to initiate staff training immediately after a deficiency...

Sleep Health: Circadian Rhythms and Sleep Disorders 101
The column highlights the massive burden of sleep disorders in the United States, noting that 83.7 million adults (32.4% of the population) have obstructive sleep apnea (OSA) and that 30‑50% of adults suffer from insomnia, with women twice as likely as...

Moderna in Talks with FDA over Phase 4 Covid Vaccine Data
Moderna is actively collaborating with the U.S. Food and Drug Administration to submit Phase 4 post‑marketing data on its COVID‑19 vaccines. The company hopes the additional safety and efficacy evidence will persuade regulators to broaden the current, narrowed product labels that...

AI, Gene Therapies Drive Market Trends in Eye Care
In 2026 the ophthalmology market is being reshaped by gene‑therapy breakthroughs and the emergence of agentic AI, according to Boston Consulting Group’s Long Sha. Gene‑based treatments are moving beyond rare retinal disorders into chronic conditions such as wet age‑related macular...

Establishing Good Governance: Start with the Important Basics and Play the Long Game
Healthcare organizations increasingly recognize that robust board governance is essential for navigating financial strain, regulatory change, and emerging technologies like AI. The article outlines four foundational practices: establishing a Governance and Nominating committee, conducting biennial self‑evaluations, implementing proactive succession planning...
Mission, Margin and a Midterm Clock: Healthcare Signals to Watch
Healthcare leaders this week wrestled with the tension between mission and margin. Northwell Health accepted a 1.1% operating margin to fund a new behavioral‑health tower, while Epic’s founder emphasized profit as a side effect of a $6.7 billion revenue business. Tenet...

AI Is Forcing Even Insurance’s Most Cautious Players to Move Fast
AI is reshaping the insurance and healthcare sectors at a speed unprecedented for an industry built on caution. Jake Sloan, Appian’s VP of global insurance, highlighted that insurers can now move from pilot projects to full production in weeks, not...
Common Cholesterol Medications Do Not Alter Long-Term Dementia Risk
A massive target‑trial emulation study of more than 320,000 older adults found that statin use does not change long‑term risk of dementia. While statin users showed a 46% spike in dementia diagnoses during the first year after initiation, researchers attribute...
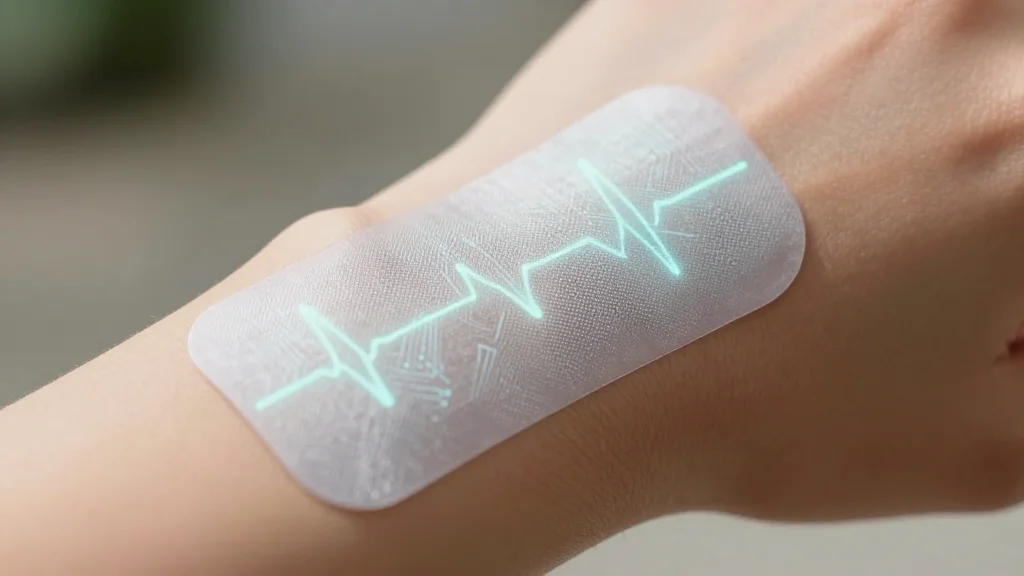
Battery-Free Skin-Conformal Wearable System Can Measure Electrocardiogram Signals
A research team led by Prof. Jerald Yoo at Seoul National University unveiled SkinECG, a skin‑conformal wearable that records electrocardiogram signals without a battery. The device uses an Orthogonal Energy Harvesting Network to wirelessly deliver power harvested from multiple on‑body...

Sequenex Announces Partnership with MedTech Innovator, Industry’s Leading Startup Accelerator
Sequenex announced a strategic partnership with MedTech Innovator, the world’s largest medtech accelerator, to support early‑stage companies developing connected medical devices. The 2026 accelerator will select 65 startups from a record 1,800 applications, and Sequenex will provide financial backing and...

Surgery Still Outperforms GLP-1 Drugs in Terms of Heart Health
A Mayo Clinic study of more than 800 patients compared metabolic and bariatric surgery (MBS) with GLP‑1 drugs such as semaglutide and tirzepatide. Surgery produced an average 28% weight loss versus 11% for medication and cut lifetime cardiovascular risk by...

Amgen Launches Late-Stage Obesity Trial in Patients Who Switch From Rival Drugs
Amgen is initiating three Phase III trials for its long‑acting obesity injection MariTide, including a pivotal study that enrolls about 1,200 patients switching from Eli Lilly’s semaglutide or Novo Nordisk’s tirzepatide. The primary goal is a minimum 10% body‑weight loss after 68 weeks,...
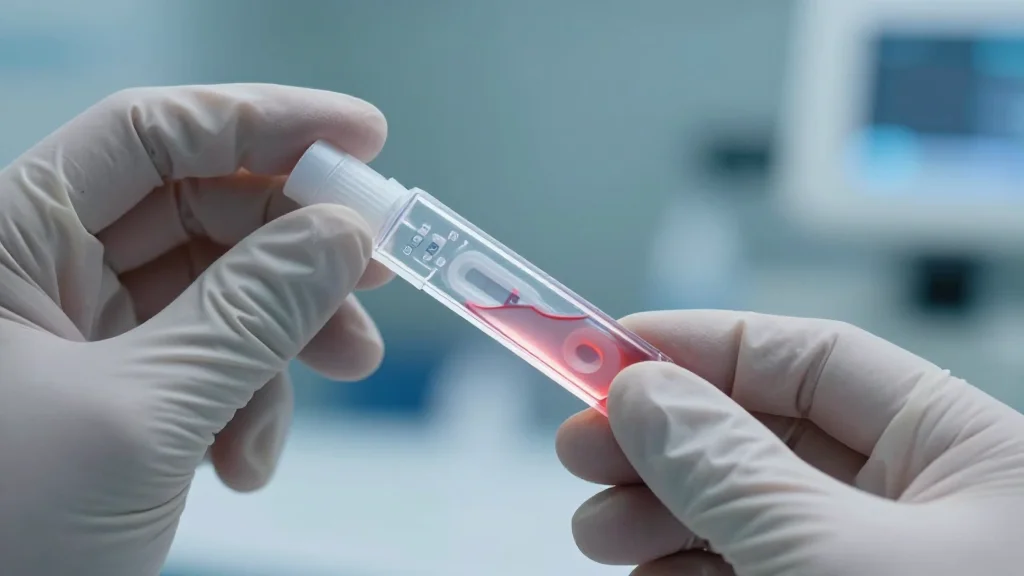
Faster and Easier Ways to Diagnose Mpox: New Approaches Improve Detection
A review in *Trends in Biotechnology* outlines new point‑of‑care (POC) diagnostic platforms for Mpox, highlighting isothermal amplification, CRISPR‑based assays, biosensors and AI‑enhanced lesion imaging. The authors argue these tools can approach PCR sensitivity while eliminating the need for complex labs....

Nemours Children’s Health Breaks Ground on Multispecialty Facility
Nemours Children’s Health broke ground on a new 34,000‑square‑foot multispecialty pediatric facility in Viera, Melbourne, Florida. The center will host roughly two dozen services, ranging from allergy and cardiology to oncology and orthopedics. Construction is set to begin this summer...