Know What's Happening in Healthcare
Know What's Happening in Healthcare

FDA approves KRESLADI, first CIRM‑backed gene‑editing therapy
The FDA cleared KRESLADI, a gene‑editing treatment from Rocket Pharmaceuticals for severe leukocyte adhesion deficiency‑I in children, eliminating the need for a bone‑marrow donor. It is the first product directly funded by California’s Institute for Regenerative Medicine to receive approval.
Also developing:
By the numbers: Syneron Bio raises $150M Series B

Valo Health is reshaping drug development by applying AI to human causal biology, aiming to raise clinical success rates from roughly 10% to 20%. The company leverages over 17 million de‑identified patient records and Mendelian randomization to pinpoint genetically validated targets. Recent collaborations include a up‑to‑$3 billion deal with Merck for Parkinson’s research, expanded work with Pfizer on autoimmune therapies, and a cardiometabolic partnership with Novo Nordisk. Valo will not commercialize drugs itself, instead licensing candidates to larger partners, and has already paused a diabetic retinopathy program after a Phase 2 failure.

A Parallels survey of 600+ global IT leaders found 94% fear vendor lock‑in, especially in health systems where platforms now underpin clinical, security and data functions. The study shows a shift from cost‑avoidance to protecting organisational freedom, prompting hybrid and...

Novo Nordisk has struck a deal with Boston biotech Vivtex worth up to $2.1 billion to develop next‑generation oral GLP‑1 obesity and diabetes pills. The partnership leverages Vivtex’s AI‑driven gastrointestinal‑on‑a‑chip platform that can boost drug absorption by orders of magnitude. Novo...

Chinese biotech firms are moving beyond licensing deals as capital markets open, highlighted by a 64% rise in the Hang Seng Biotech Index in 2025. Reforms to listing rules since 2018 have unlocked public financing, enabling a record $138 billion in...

A patient in MacroGenics' Phase 2 LINNET trial of the bispecific antibody lorigerlimab suffered grade 4 neutropenia and septic shock, leading to a fatality and prompting the FDA to place a partial clinical hold on the study. The company also reported three...

The Digital Health Rewired 2026 conference programme, featuring ten themed stages, has been released. More than 300 senior health‑IT leaders, including NHS CEOs, CIOs, CCIOs and CNIOs, will present sessions on digital transformation, cyber security, AI, and electronic patient records....

UKG Pro Workforce Management (WFM) offers a structured, data‑driven approach to healthcare scheduling, tackling the sector’s chronic staffing volatility. By enabling early offers of open shifts through advanced scheduling, organizations can pre‑empt gaps, lower overtime, and align skill sets with patient...
Researchers at Shanghai Xuhui District Stomatological Hospital and the Chinese Academy of Sciences have engineered a vibration‑activated teeth‑whitening powder (BSCT) that works with electric toothbrushes. The ceramic powder uses piezoelectric catalysis to generate reactive oxygen species for stain removal while...

O-Health, an 18‑month‑old clinical AI startup, has built a voice‑first operating system that turns doctor‑patient conversations into structured clinical intelligence in real time. The platform runs on an edge‑first, sovereign AI stack with in‑house medical ASR and small language models,...

Researchers are exploring single‑cell omics as a next‑generation tool for preventive health screening. By profiling thousands of individual cells, the technology can detect subtle genetic mutations and immune‑cell shifts years before clinical symptoms appear. Early studies have identified driver mutations...

NVIDIA’s second State of AI in Healthcare report shows AI moving from experimentation to profit across radiology, drug discovery, medical devices and digital twins. Seventy percent of respondents now actively use AI, up from 63% a year earlier, and 69%...

The Substance Abuse and Mental Health Services Administration (SAMHSA) led a massive federal response to keep behavioral health services operational during COVID‑19, awarding over $32 billion in grants from FY2020‑2023, including $8.3 billion in pandemic‑specific funding. SAMHRA provided grant extensions, telehealth prescribing...

Zifo and Maze Therapeutics have teamed up to launch an AI‑powered platform that manages, stores, and scales massive biobank datasets. The solution tackles the fragmentation of genetic, proteomic, and phenotypic data by providing a unified workflow that delivers summary statistics...

I’m proud to introduce 𝐓𝐡𝐞 𝐌𝐞𝐝𝐢𝐜𝐚𝐥 𝐅𝐮𝐭𝐮𝐫𝐢𝐬𝐭’𝐬 𝟏𝟎𝟎 𝐃𝐢𝐠𝐢𝐭𝐚𝐥 𝐇𝐞𝐚𝐥𝐭𝐡 𝐚𝐧𝐝 𝐀𝐈 𝐂𝐨𝐦𝐩𝐚𝐧𝐢𝐞𝐬 𝐨𝐟 𝟐𝟎𝟐𝟔! The digital health and AI fields are saturated with hype, and distinguishing the promising players from the noise is a challenge. The annual list is our...

Eli Lilly’s obesity drug Zepbound achieved a 25.5% weight loss in a head‑to‑head Phase 3 trial, outpacing Novo Nordisk’s CagriSema and sending Novo’s shares down 20%. The FDA launched Rare Disease Week, issuing draft guidance for a new Plausible Mechanism Pathway that...
The article argues that the brain is the rate‑limiting organ for longevity, asserting that neural network degradation drives functional decline across the body. While peripheral organ rejuvenation has advanced, preserving and repairing brain tissue remains the critical bottleneck. It proposes...

The National Safety Council introduced the Organization Safety Gap Analysis Tool, a digital assessment that translates its evidence‑based SIF Prevention Model for clinical laboratories. The ten‑to‑fifteen‑minute, color‑coded questionnaire evaluates seven core safety elements, from leadership to continuous improvement. By highlighting...

The FDA’s crackdown on non‑approved or “copycat” GLP‑1 drugs is expected to push patients toward FDA‑approved therapies, elevating the role of retail pharmacies as primary access points. GoodRx’s head of retail network, David Graziano, says the company’s cost‑plus pricing model...

The episode discusses the alarming rate of medical errors—estimated at 800,000 to a million deaths annually—and explores how AI can help reduce these mistakes. The host highlights personal experience using AI tools like OpenEvidence, GPT, and Gemini to obtain specialist-level...

SteinCares has signed a strategic licensing agreement with Shilpa Biologicals to commercialize a biosimilar across Latin America. Under the deal, SteinCares receives exclusive rights to register, market and distribute the product throughout the region, while Shilpa Biologicals will complete development...

Medical device maker Stryker has launched Synchfix EVT, a flexible syndesmotic fixation system designed to simplify ankle stabilization procedures. The all‑in‑one, single‑use device integrates suture tensioning handles and a low‑profile titanium medial implant, reducing procedural complexity for both adult and adolescent...

Mevion Medical Systems announced that its S250‑FIT proton therapy system has received CE marking under EU MDR, allowing marketing and clinical use across the European Union. The compact, LINAC‑vault‑ready platform, already cleared by the U.S. FDA, fits into standard radiation...

Vico Therapeutics has initiated patient dosing in an expanded European cohort for its Phase I/IIa VO659 trial, targeting Huntington's disease, spinocerebellar ataxia type 3 and type 1. The study employs a twice‑annual intrathecal regimen and will monitor safety, tolerability, pharmacodynamics and pharmacokinetics...

NorthX Biologics and Demeetra have formed a strategic alliance to provide an end‑to‑end solution that couples Demeetra’s CleanCut CHO cell line development platform with NorthX’s GMP manufacturing capabilities. The partnership leverages a high‑titre GS‑knockout CHO cell line that carries no...

The Evolution Summit, organized by Marcus Evans, will take place May 7‑8, 2026 at the Ritz‑Carlton in Fort Lauderdale. The invitation‑only event convenes senior clinical‑trial executives with innovative suppliers to discuss pressing challenges in clinical operations and drug development. Featured speakers include...

Labcorp announced an expanded partnership with PathAI to deploy the AISight Dx digital pathology platform across its U.S. anatomic pathology labs and hospital sites. The AI‑enabled software will allow pathologists to scan, annotate and diagnose slides digitally, aiming to cut turnaround...
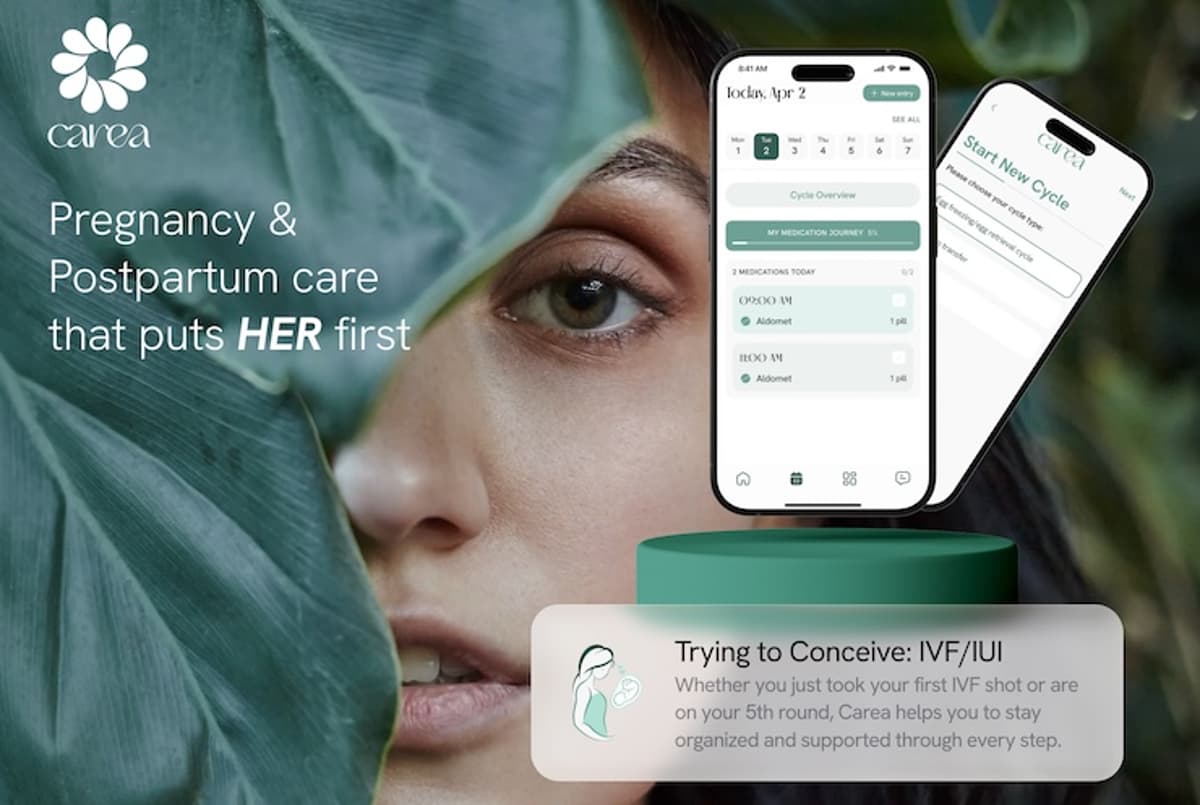
Carea has introduced a new "Trying to Conceive: IVF/IUI" mode within its pregnancy and postnatal wellbeing app, offering a day‑to‑day support system for women undergoing fertility treatment. The feature includes a personalised medication tracker, video guides, reminders, and a visual...

Sleep Cycle and Carnegie Mellon University's Delphi Group have launched a five‑year research collaboration to test whether privacy‑preserved sleep data—particularly nightly cough intensity captured by Sleep Cycle’s Cough Radar—can augment traditional respiratory disease surveillance. The partnership will share de‑identified data...

Galderma announced that regulatory agencies in the European Union, United States and Canada have approved a new state‑of‑the‑art Restylane® syringe for use with its NASHA® lidocaine filler line. The device is cleared for a broad set of facial areas—including cheeks,...

Roger Whitney concludes his four-part series on pre‑Medicare health care by introducing the OODA Loop—observe, orient, decide, act—as a practical decision framework. He walks listeners through building a five‑year cash‑flow plan, estimating MAGI, and assessing ACA subsidy eligibility alongside COBRA...

The Science and Technology Facilities Council’s Hartree Centre has partnered with Alder Hey Children’s NHS Foundation Trust to pilot an AI‑driven staff scheduling platform. The system automatically creates balanced on‑call rotas by factoring in leave, skills and working patterns, replacing...

In this episode, Dr. Aaron Vinnie shares his journey from a leukemia survivor to a Columbia University hematology‑oncology researcher, advocating a shift from traditional cytotoxic chemotherapy to precision strategies that rewire malignant blood cells. He explains how hematologic cancers stem...

Long‑term data from the Phase 3 ULTIMATE I and II trials show that continuous treatment with Briumvi (ublituximab) provides sustained clinical benefits for relapsing multiple sclerosis over five years. More than 80 % of patients who started Briumvi remained free of relapses and...

Gut Clinic, a gastro‑metabolic health platform founded in 2024, secured $1 million (≈₹9 crore) in a seed round backed by more than 15 investors. The capital will fund new outpatient centres, upgraded clinical systems, and expanded diagnostic services across India’s $197 billion preventive...

Veg ER for Pets has opened a 5,000‑sq‑ft, 24/7 veterinary emergency hospital in Etobicoke, marking its first Canadian location and expanding its North American network to 121 facilities. The open‑concept design lets pet owners stay with their animals during treatment, including surgery and...

Johnson & Johnson has filed a supplemental Biologics License Application (sBLA) with the U.S. FDA for Imaavy (nipocalimab‑aahu), a monoclonal antibody intended to treat warm autoimmune hemolytic anemia (wAIHA). The filing is supported by the Phase II/III ENERGY trial, which demonstrated...

A new NHS England GP contract, backed by £485 million, will guarantee same‑day appointments for urgent patients. It mandates that online consultation platforms remain available throughout core hours and redirects £292 million to recruit roughly 1,600 additional full‑time‑equivalent GPs. The contract expands...

President Donald Trump used a brief four‑minute segment of his State of the Union address to push Congress to codify his Most‑Favoured Nations (MFN) drug‑pricing program. He claimed the voluntary agreements would give Americans the lowest prescription prices worldwide, though...

In July 2025 the FDA ordered a complete halt of Sarepta’s gene‑therapy Elevidys after two non‑ambulatory Duchenne muscular dystrophy (DMD) patients died, sparking a market plunge and patient access concerns. Sarepta argues that ambulatory patients, who had been receiving Elevidys...

The FDA reauthorized its rare pediatric disease priority review voucher program, bolstering a wave of regulatory incentives that include new guidance on plausible mechanisms and a Rare Disease Evidence Principles framework. Yet ultra‑rare indications still struggle to attract traditional venture...

HekaBio K.K. and Alpha Tau Medical have secured Japanese regulatory approval for Alpha DaRT, a diffusing alpha‑radiation therapy for unresectable locally advanced or recurrent head‑and‑neck cancer. The approval makes Japan the first country outside Israel to clear the device, positioning HekaBio...

President Trump negotiated lower prices for some drugs on TrumpRx, but discounts only apply to cash paying patients not using their insurance. Many drugs already had big discounts through manufacturers or coupons, and some have generic equivalents that are already...

President Trump has called for his deals with drug companies to be enacted into legislation. It's hard to know what that means since the deals are not public.

Oak HC/FT partner Vig Chandramouli highlighted four health‑tech trends that startups must address to win investor confidence. First, labor management for nurses and allied professionals—who comprise 70% of the workforce—is a largely untapped market. Second, investors are scrutinizing margins, finding...

With Vivtex deal, Novo gains a chance at better oral obesity drugs https://t.co/DhIelbC9jI by @gwendolynawu $NVO #obesity

Stand Up! with Pete Dominick: 1538 Dr Peter Hotez on the state of Vaccination in America. My latest with @PeteDominick https://t.co/LsuvmglNBx

At the ViVE health‑tech conference, HFMA CEO Ann Jordan led a panel that warned the industry that healthcare affordability is an urgent, systemic problem. The discussion highlighted the Vitalic Health initiative, which seeks to marry financial sustainability with better outcomes,...
A HIMSS fireside chat highlighted how voice AI is transitioning from pilot projects to production in hearing‑care clinics. PolyAI’s Alex Brown and Audibel’s Brianna Warner discussed using conversational agents to expand patient access while preserving trust and empathy. The session...
The HIMSS‑hosted session introduced agentic AI as a transformative operating system for healthcare, capable of ingesting the 99% of hospital data that remains invisible to traditional predictive models. Speakers from Philips, AWS, MIT, and Mass General explained how continuous AI...
NHS England has signed a three‑year, £33.6 million contract with London‑based TPXimpact to deliver digital services for maternity and child vaccinations. This agreement is the third digital‑prevention deal signed in the last six months, bringing the total spend on such services...