Today's Healthcare Pulse

FDA greenlights durvalumab combo for high‑risk bladder cancer
The FDA approved durvalumab (Imfinzi) combined with Bacillus Calmette‑Guerin for BCG‑naïve, high‑risk non‑muscle invasive bladder cancer. The POTOMAC trial enrolled 1,018 patients and showed a 32% reduction in disease recurrence risk (hazard ratio 0.68, p=0.015). Durvalumab is given at 1,500 mg IV every four weeks for up to 13 cycles.
Also developing:
By the numbers: Apogee Therapeutics raises $1.3B royalty financing
Diffusion MRI as A Biomarker for Monitoring Recovery After Surgical Repair of Traumatic Peripheral Nerve Injuries: A Longitudinal Case Series
A longitudinal case series evaluated diffusion tensor imaging (DTI) as a biomarker for monitoring recovery after surgical repair of severe ulnar and median nerve transections. Researchers tracked fractional anisotropy (FA) values alongside Medical Research Council sensory and motor grades for twelve months post‑operation. The FA trajectories closely mirrored clinical scores, and a Gompertz‑function model automated the analysis, minimizing user‑dependent variability. The findings support FA‑based DTI as a non‑invasive tool to gauge nerve regeneration and guide timely interventions.

Profluent and Lilly: The Next Gene Editor Will Be Designed by AI
Profluent, an Air Street Capital portfolio company, announced a multi‑program partnership with Eli Lilly to create AI‑designed recombinases for kilobase‑scale gene editing. The deal includes an upfront cash payment, committed R&D funding, and up to $2.25 billion in development and commercial milestones...

China's Brain‑Gut Health Initiative Links Microbiome Shifts to Psychiatric Disorders
The Brain‑Gut Health Initiative (BIGHI), a prospective Chinese cohort of more than 1,200 adults, has identified distinct gut‑microbiome and brain‑imaging signatures that correlate with symptom severity across schizophrenia, depression and bipolar disorder. The multi‑omics findings suggest new, objective biomarkers for...
FDA Clears Eli Lilly’s Foundayo, First New Oral Anti‑Obesity Drug in Years
The U.S. Food and Drug Administration approved Eli Lilly’s once‑daily oral weight‑loss pill, Foundayo, making it the first new anti‑obesity medication in years and only the second oral GLP‑1 option. Priced between $149 and $349 a month, the drug promises a...
WHO Webinar Reveals Ongoing Gaps in Childhood Immunization Across 92 Nations
The World Health Organization convened a global webinar to unveil a new study that maps stark within‑country disparities in childhood immunization across up to 92 nations. The analysis, covering nine vaccine indicators and five socioeconomic dimensions, underscores that gaps tied...

FDA Grants RMAT Status to Orca-Q in High-Risk Hematologic Malignancies
The U.S. Food and Drug Administration has awarded Regenerative Medicine Advanced Therapy (RMAT) designation to Orca‑Q, an investigational allogeneic T‑cell immunotherapy targeting high‑risk hematologic malignancies. The designation follows early phase 1 data that hint at improved overall survival, lower graft‑versus‑host disease...

Chinese Cohort Finds U‑Shaped Cholesterol‑Mortality Link, Upending Biohacker Dogma
Researchers analyzing a Chinese cohort of 163,115 adults uncovered a U‑shaped relationship between total, LDL and non‑HDL cholesterol and all‑cause mortality. Levels below 120 mg/dL (TC) or 70 mg/dL (LDL‑C) were linked to higher cancer and hemorrhagic stroke deaths, while higher levels...

Centene Hikes 2026 Profit Guidance After Buoyant Q1
Centene posted Q1 adjusted earnings of $3.37 per share, far surpassing analysts’ $2.25‑$2.30 expectations, driven by rate hikes that offset a 2‑million drop in ACA enrollment. Net profit rose 18% to $1.5 billion on revenue of $49.9 billion, prompting the company to...

Pfizer Deals Extend Patent Life for a Top-Selling Rare Disease Drug
Pfizer announced settlements with Dexcel Pharma, Hikma Pharmaceuticals and Cipla that push the patent expiry of its rare‑disease drug Vyndamax to June 1, 2031. The deals delay generic tafamidis entry, keeping U.S. sales stable through the early 2030s after the company warned...

FDA Grants Priority Review and Breakthrough Designation to GSK‑Ionis Hepatitis B Drug Bepirovirsen
The U.S. FDA has accepted Ionis Pharmaceuticals’ bepirovirsen for priority review and awarded it breakthrough therapy designation, setting a PDUFA target date of Oct. 26, 2026. The antisense drug, developed with GSK, could become the first therapy to achieve meaningful functional cures...

U.S. Government Will Stop Paying for Test Strips to Detect Deadly Drugs
The Trump administration announced that SAMHSA will stop paying for fentanyl test strips, reversing a policy that began in 2021. The agency says the strips encourage drug use, prompting a letter to states and grant recipients. The change unsettles programs...

The Persistent Misleading Claim That Vaccines Aren’t Properly Tested for Safety
FactCheck.org debunks the persistent anti‑vaccine claim that childhood vaccines lack proper safety testing. The article explains that vaccine trials employ active comparators and extensive phase III studies, gathering safety data from millions of doses before approval. It also highlights ongoing post‑marketing...
Medtronic Secures CE Mark for Stealth AXiS Surgical System in Europe
Medtronic announced that its Stealth AXiS™ surgical system has earned a CE mark, allowing the integrated planning, navigation and robotics platform to be marketed throughout the European Union. The clearance follows earlier FDA approvals and positions the company to offer...

VSRF LIVE TONIGHT: Episode 225: Vaccine Injured: A Path to Recovery
VSRF Live’s Episode 225 aired three personal accounts of alleged COVID‑19 vaccine injuries and highlighted emerging recovery paths, including self‑directed research, experimental therapies, and overseas plasma filtration. The broadcast also promoted a petition to repeal the 1986 vaccine liability shield, urging...

Asian American Communities Report Delaying Care Due to Cost, Recent Immigration Policies
A nationwide electronic survey of 1,027 Asian American, Native Hawaiian and Pacific Islander adults revealed major gaps in screening and treatment for hepatitis B, diabetes, and tuberculosis. While 90% reported a primary‑care physician, only 40% had been screened for hepatitis B and...

Restylane, Sculptra Combo Improves Skin Dryness, Collagen Loss for Menopausal Women
Interim data from two investigator‑initiated trials show that a sequential regimen of Restylane Skinboosters followed by Sculptra markedly improves skin hydration, firmness and collagen density in menopausal women. The 9‑month study reported peak patient satisfaction at month six, with measurable...

Northwestern Engineers Print Artificial Neurons That Talk to Real Brain Cells
Engineers at Northwestern University, led by Mark C. Hersam and Vinod K. Sangwan, printed flexible artificial neurons that generated realistic electrical signals and successfully activated living mouse brain cells. The study, published in Nature Nanotechnology, demonstrates a new level of...
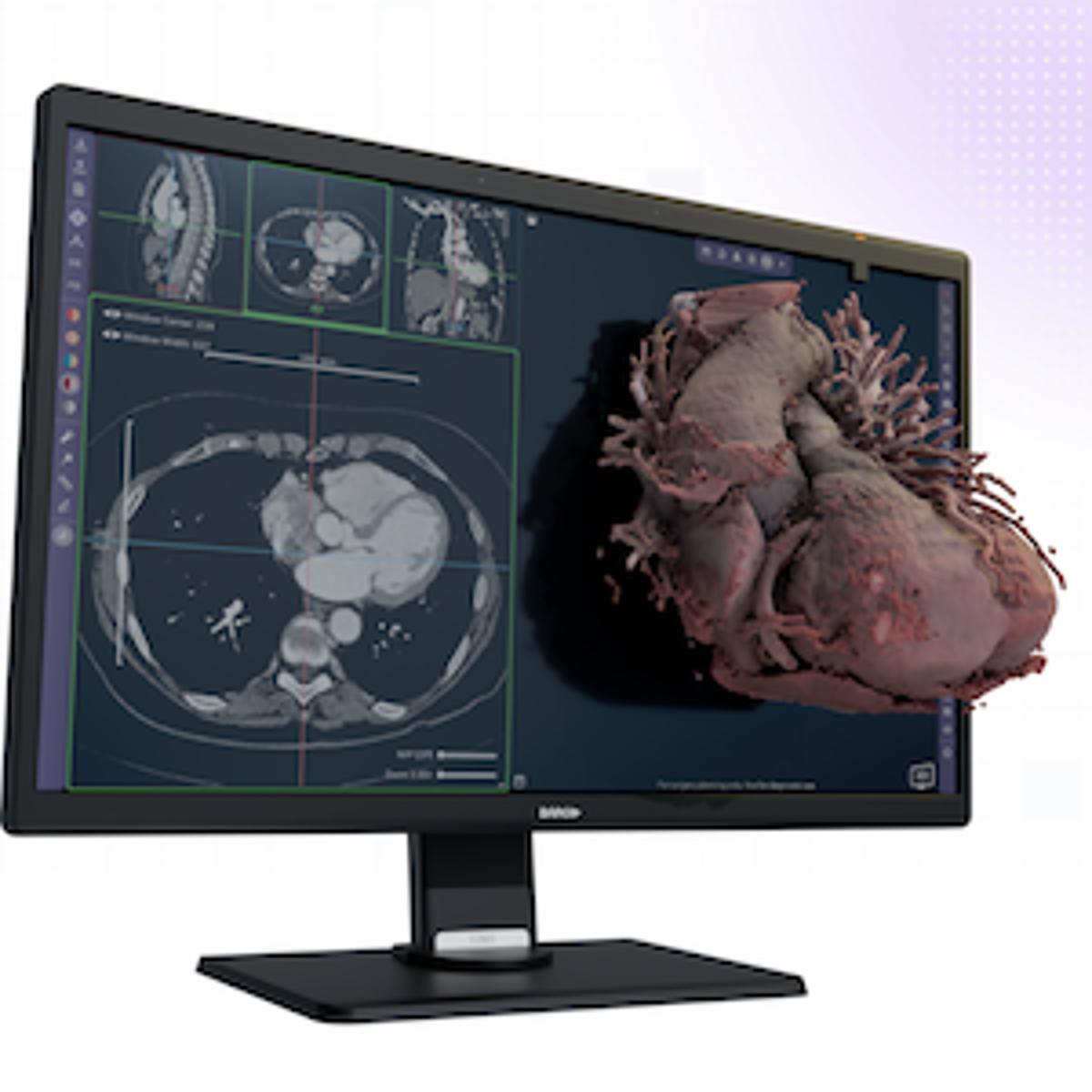
FDA Grants Clearance for Avatar Medical Vision
Avatar Medical has received FDA 510(k) clearance for its Avatar Medical Vision platform, enabling instant 3D processing of CT and MR images for surgical planning. The software supports glasses‑free 3D displays, virtual reality and integrates with Barco’s Eonis monitor, allowing...

Scientists May Have Finally Created a Male Birth Control Pill—So Far, It Shows Very Few Side Effects
YourChoice Therapeutics reported that its experimental male contraceptive pill YCT‑529 was well‑tolerated in a Phase 1 trial of 16 healthy, vasectomized men. The non‑hormonal compound halted sperm production without altering testosterone, heart rate, mood, or sexual desire. Pre‑clinical work in mice...

Frequency of Medical Liability Lawsuits Is Declining, but Risk Remains for Doctors: AMA
The American Medical Association’s latest Physician Practice Benchmark Survey shows that the proportion of doctors sued at least once dropped to 28.7% in 2024 from 31.2% in 2022, indicating a modest decline in medical‑liability lawsuits. However, litigation risk remains concentrated...

Obesity’s Effects on the Immune System May Linger for Years
A European study shows that helper CD4+ T cells retain a pro‑inflammatory effector‑memory phenotype long after mice regain normal weight following obesity. While adipose mass normalizes, the inflammatory T‑cell response persists for weeks and only resolves after extended weight‑maintenance periods....

New York Proposes an Additional Layer of Legal Protection to Those Profiting From Injecting Vaccines
New York is introducing Senate Bill A9140, which would grant blanket legal immunity to doctors, pharmacists and other entities that prescribe, dispense, order, furnish, or administer vaccines to minors. The bill bars any civil lawsuit alleging injury, including residual effects,...

Charting FDA‑Guided Trial Pathways for Merkel Cell Carcinoma
Really enjoyed my fireside chat at MCC4 with Harpreet Singh, MD, the CMO at Precision Medicine, focused on establishing pathways for clinical trials in Merkel cell carcinoma. The physician-scientists in the audience were clearly excited about the prospect of working...
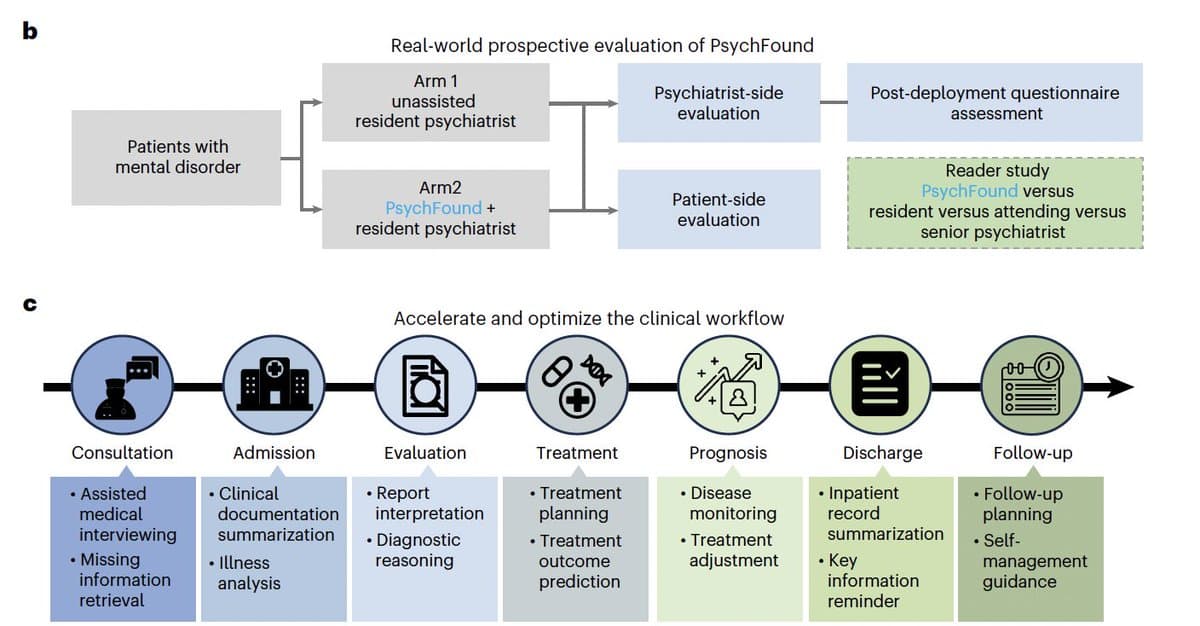
AI‑assisted Psychiatrists Match Attending Quality, Boost Accuracy
Some AI progress for mental health support seen in a real-world prospective study (<-there are few of these) "In a real-world, two-arm prospective study, resident psychiatrists assisted by PsychFound demonstrated higher consultation quality, higher diagnostic accuracy, more appropriate medication selection...
Iowa FD's Whole Blood Pilot Program Proves Worth with Stabbing Victim
The Des Moines Fire Department launched a pilot that equips ambulances with whole‑blood units and transfusion kits, allowing paramedics to start life‑saving transfusions before hospital arrival. In February, a paramedic used the system on a stabbing victim, delivering blood on...

5 Costly IP Mistakes to Avoid
The article outlines five costly intellectual‑property mistakes that medical‑device innovators frequently make, from relying on a single patent to neglecting global filings and broader brand protection. It stresses building a “picket fence” of patents that cover the core device, components,...

Mental Health Chatbots Raise Serious Ethical Concerns, Review Warns
A new peer‑reviewed study in Digital Health warns that mental‑health chatbots are outpacing ethical oversight. The authors identify four major risks: limited human involvement, a weak evidence base, extensive data‑privacy concerns, and the potential for undisclosed criminal activity. They argue...

STAT+: Supreme Court to Hear Case About ‘Skinny Labeling’ and Generic Access
The U.S. Supreme Court will hear oral arguments on a case involving “skinny labeling,” a practice where generic manufacturers seek approval to market a drug for only a subset of its approved uses. By limiting the label, generics aim to...

Healthcare Needs Early Intelligence, Not Reactive Treatment
Healthcare’s version of “prevention” is often hilarious. “We noticed the problem after it became expensive. Good news: we have a brochure.” The body was dropping hints for years. Sleep. Stress. Food. Movement. Air. Labs. Wearables. Microbiome. Mood. But we built a system that waits...

CEOs Face Congressional Hearing on High Hospital Prices
Four hospital CEOs testified before the House Ways and Means Committee about soaring hospital prices, which have risen roughly 300% over the past two decades. Committee Chair Jason Smith highlighted that 2,000 of the nation’s 4,500 hospitals have merged, giving...

ACA Market Dynamics Cost HCA $150M in Q1
HCA Healthcare posted a modest 0.6% rise in Q1 net income to $1.6 billion and a 4.3% revenue increase to $19.1 billion. A milder-than‑expected respiratory season slashed related admissions by 42% and ER visits by 32%, curbing volume growth. Storms disrupted markets...
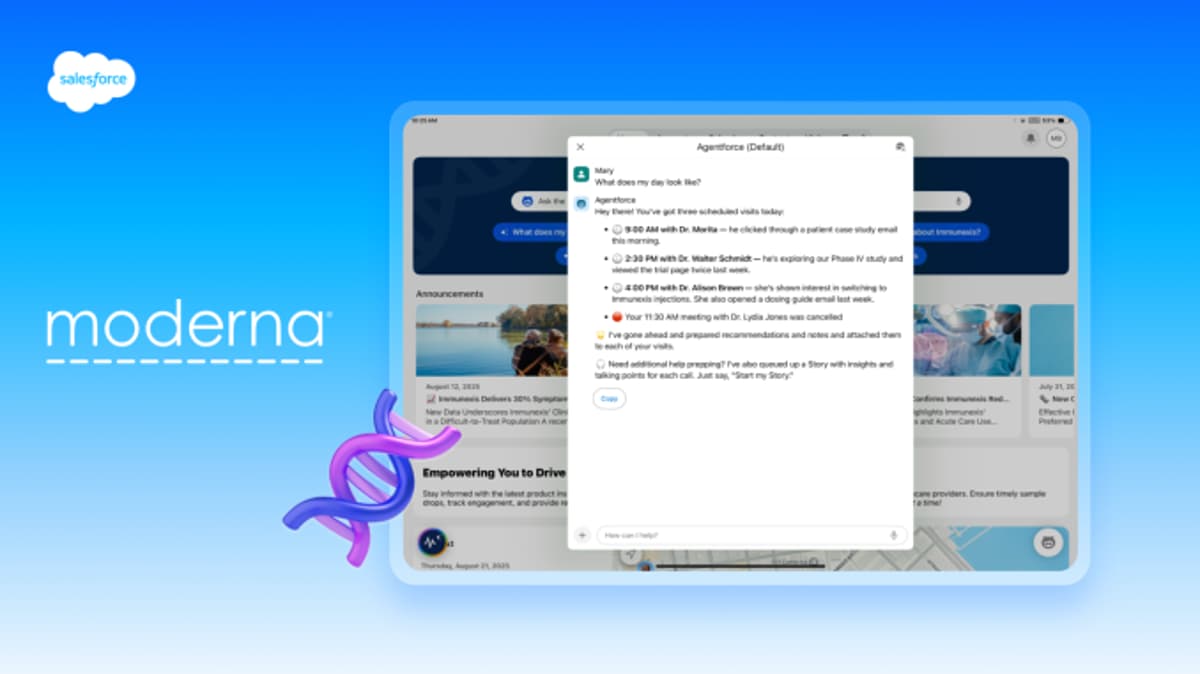
Moderna Deploys Salesforce Agentforce Life Sciences to Unify Global Commercial Operations
Moderna has chosen Salesforce’s Agentforce Life Sciences platform to unify its global commercial operations. The solution will merge data from SAP, e‑commerce and regional systems into a single Customer 360 view, powered by IQVIA OneKey. AI‑driven agents will automate cycle planning,...
$739 Million Acquisition of XOMA Strengthens Ligand’s Biopharma Portfolio
Ligand Pharmaceuticals announced a $739 million acquisition of XOMA Royalty, expanding its royalty‑financing portfolio to over 200 assets. The deal adds more than 120 new royalty streams, including seven marketed drugs such as Vabysmo and Ojemda, and deepens exposure to oncology,...

Zimmer Biomet CFO Departs for New Job
Zimmer Biomet’s chief financial officer, Suketu Upadhyay, is leaving after seven years to become CFO of biotech firm Incyte. The orthopedics maker named controller Paul Stellato as interim CFO while it searches for a permanent replacement. In its first quarter,...

How High Pressure Destroys Relational Care in Nursing
Megan Diaz, a veteran RN, argues that relentless pressure in hospitals narrows nurses' perceptual range, eroding relational care. As bed turnover and discharge speed become primary metrics, clinicians resort to labeling patients rather than exploring underlying context. This shift sacrifices nuanced...

Ludi Announces Ludi Boost™: New AI-Powered Operating Model & Strategic Services Offering for Provider Workforce Management
Ludi, Inc. unveiled Ludi Boost™, an AI‑enabled compensation platform that bundles advanced technology with a strategic services team of more than 80 experts. The solution consolidates compensation data, governance and administration into a single system of record, promising faster program execution,...
Ashvattha Therapeutics Announces Presentations Highlighting Mechanism of Action for Migaldendranib in Diabetic Macular Edema and Neovascular Age-Related Macular Degeneration
Ashvattha Therapeutics presented Phase 2 data on its subcutaneous nanomedicine migaldendranib (MGB) for diabetic macular edema and neovascular age‑related macular degeneration at ARVO. The two‑stage trial showed stable central subfield thickness for up to 12 weeks and maintained visual acuity without...
AtriCure to Participate in the 2026 Bank of America Securities Health Care Conference
AtriCure announced it will take part in the 2026 Bank of America Securities Health Care Conference, with senior management slated for a fireside chat on May 12, 2026. The session will be streamed live via the company’s investor website, giving...

Cellares and Cabaletta Bio Sign 10-Year Commercial Supply Agreement to Scale Rese-Cel
Cellares has entered a 10‑year commercial supply agreement with Cabaletta Bio to manufacture rese‑cel, the company’s autologous CAR‑T therapy for autoimmune diseases, using its fully automated Cell Shuttle and Cell Q platforms. The deal secures long‑term capacity to produce thousands of...
BPL-003
Great piece from @Shea_ARK "AtaiBeckley is advancing BPL-003—an intra-nasal formulation derived from 5-MeO-DMT—in treatment-resistant depression, with positive Phase 2b results supporting a Phase 3 initiation in the second quarter of 2026.[11] With FDA Breakthrough Therapy designation, BPL-003 has the potential...

RFK Jr’s Vaccine Lies Fuel Anti‑vax Agenda
RFK Jr lied about supporting vaccines last week, just as he lied about it many times before. Whatever anti-vax bullshit Bobby is peddling, holding up GAVI’s budget at the demonstrable expense of thousands of kids lives’ globally is unconscionable and...

Using AI to Excuse Patient Denials Damages AI’s Reputation
Letting companies use AI as a smokescreen for why patients are getting denied healthcare is going to be terrible for AI's brand https://t.co/eltFYOkih6

Ambient AI Revives Paper-Chart Conversation Flow, Boosts NPS
Juniper Health says ambient AI is helping bring conversations in the exam room back to something closer to the paper chart days. And patients are responding—Net Promoter Scores are at an all-time high. 🔗https://t.co/doV7kX4Zg3 @NextGen @WHClinics #JuniperHealth #HITSM https://t.co/sThLZIEPIl

Tax Nonprofit Hospitals Like For‑profit Businesses, Says W&M
Then the solution is to tax nonprofit hospitals (or hedge funds with beds) like the for-profit business they are. What is W&M waiting for?

Introducing Peanuts at 4‑6 Months Cuts Allergies 77%
"Peanut allergies plummet by 77% if they're added to babies' diets at 4-6 months of age." This statistic traces back to a 2023 modeling study: Early introduction of peanut reduces peanut allergy across risk groups in pooled and causal inference analyses🥜https://t.co/SHULN2n2X5 https://t.co/CCFCt6Uguu
Treat AI as Strategy, Not a Tool, to Transform Healthcare
Organizations that approach AI as a strategic capability—rather than a standalone tool—will be positioned to unlock its full potential, improving care, efficiency, and patient experience. https://t.co/R0j6YngDUq

AI Protein Design Advances, Yet Developability Remains Overlooked
Some new protein design competition results from Rio, showing the field keeps tackling harder and harder targets w/ AI methods Might be time to factor in other properties (developability, manufacturability)? https://t.co/h1niVwjnSS
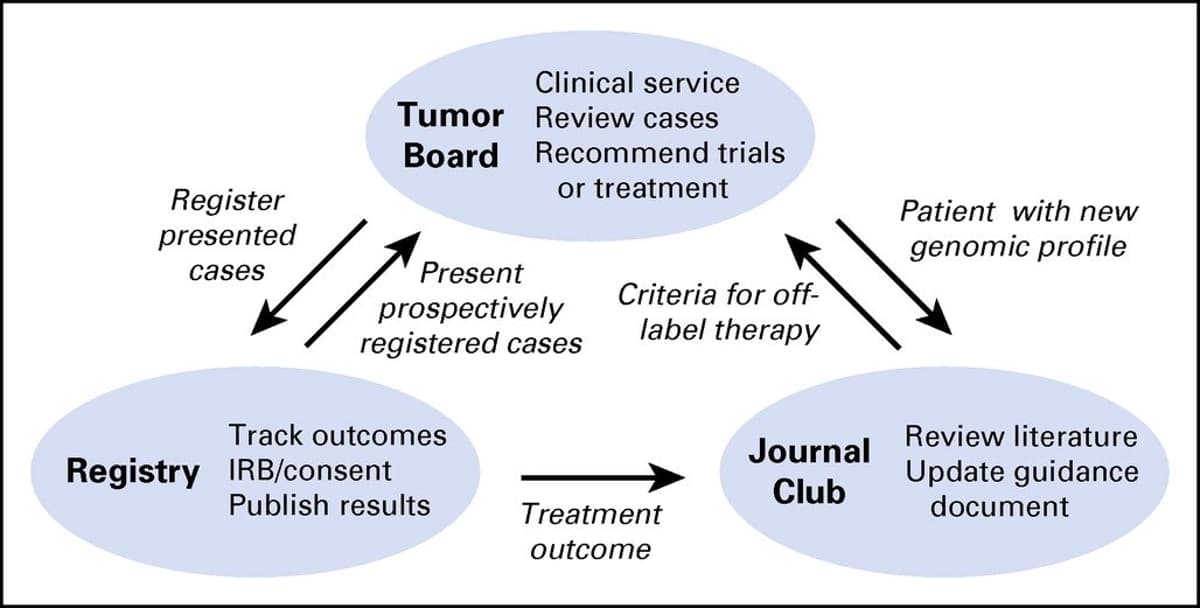
Integrated Academic-Community Tumor Board Enhances Precision Medicine
Implementation & Utility of an Integrated Academic-Community #PrecisionMedicine Molecular Tumor Board [MTB] [Jul 5, 2017] @meburkard et al. @JCOPO_ASCO https://t.co/HOWIx826Lm https://t.co/1JmBdW6K5c

First-Year Lessons Coordinating an Oncology Precision Medicine Clinic
Coordinating an Oncology #PrecisionMedicine Clinic w/in an Integrated Health System: Lessons Learned in Year 1 [1/28/19] @mtmdphd Godden, Wham @antonyruggeri @mullane_michael Wilson @shamsvirani Weissman, Ramczyk, Vanderwall @JimW9200 @Aurora_Cancer @JPCRR https://t.co/bxgjOfLhwk https://t.co/4pR7nFLzGW
Overcoming Barriers to Community Oncology Precision Medicine
Implementing #PrecisionMedicine Programs and #ClinicalTrials in the Community-Based Oncology: Practice: Barriers and Best Practices - @JLErsek @lorablk @mtmdphd & @EdKimMDLCI #ASCO18 Education Book https://t.co/ahsmfePatk #NCORP