Today's Healthcare Pulse

FDA greenlights durvalumab combo for high‑risk bladder cancer
The FDA approved durvalumab (Imfinzi) combined with Bacillus Calmette‑Guerin for BCG‑naïve, high‑risk non‑muscle invasive bladder cancer. The POTOMAC trial enrolled 1,018 patients and showed a 32% reduction in disease recurrence risk (hazard ratio 0.68, p=0.015). Durvalumab is given at 1,500 mg IV every four weeks for up to 13 cycles.
Also developing:
By the numbers: Apogee Therapeutics raises $1.3B royalty financing

UTHealth Houston Names Business Affairs, Strategy Chief
UTHealth Houston announced the appointment of Michael Patriarca as executive vice president and chief business affairs and strategy officer. The newly created role is designed to steer long‑range strategic planning while overseeing finance, operations, IT, data analytics, and infrastructure. Patriarca joins from Tulane Medicine, where he served as vice president and chief business officer. The move reflects UTHealth’s expanding academic, research, clinical, and administrative footprint.

NVO’s 4.4% Yield Boosted by Oral Semaglutide Surge
$NVO pays a decent 4.4% dividend and oral semaglutide is surging: "First-time anti-obesity medication (AOM) prescribing increased 21.7% from December 2025 to March 2026, with first-time AOM semaglutide prescribing increasing by more than 50%. AOM semaglutide also showed its largest quarter-over-quarter percentage...

Air Pollution Exposure in the Womb Linked to Worse Language and Motor Development
A King's College London study of 498 Greater London infants links first‑trimester air‑pollution exposure to lower language scores at 18 months and, for pre‑term babies, to markedly poorer motor development. Children whose mothers lived in high‑pollution areas scored 5‑7 points lower...
![He Declined Routine X-Rays and Was Denied a Dental Cleaning [PODCAST]](/cdn-cgi/image/width=1200,quality=75,format=auto,fit=cover/https://kevinmd.com/wp-content/uploads/18cacd38-6c3b-419c-8d97-fab1cca74be2.png)
He Declined Routine X-Rays and Was Denied a Dental Cleaning [PODCAST]
Patient advocate Aaron Rosenberg was denied a routine dental cleaning after refusing non‑mandatory bite‑wing X‑rays, despite recent imaging and low risk. The clinic cited a licensure risk and an internal policy, though the ADA does not require such X‑rays for...

Site-Neutral Payment Debate Intensifies in Hospital Affordability Hearing
During a House Ways and Means hearing, hospital CEOs and lawmakers debated site‑neutral payment as a lever to curb soaring hospital costs, which have risen roughly 170% since 2005. Republicans touted proposals that could save $160 billion in taxes and $672 billion...

MultiCare to Open $479M Mary Bridge Children’s Hospital May 16
MultiCare is set to open the new Mary Bridge Children’s Hospital in Tacoma on May 16, a $479 million, six‑story facility that replaces the original 1955 campus. The hospital features eight operating rooms, 29 emergency‑department exam rooms, dedicated play spaces and...

CMS Veteran Blum Warns a Version of the Nursing Home Staffing Rule Could Return After November
Former CMS chief operating officer Jonathan Blum warned that a version of the controversial nursing‑home staffing mandate could be revived after the November midterms if Democrats regain control of the House. He told the LTC 100 Leadership conference that policymakers must...

‘Low-Hanging Fruit’: Nationwide Survey of Nursing Home Staff Shows Easy Fixes for Retention Amid Managed Care, PDPM Burdens
A nationwide AAPACN/LTC 100 survey of 489 skilled‑nursing‑facility nurses shows 65% overall job happiness but highlights documentation duplication across Medicaid, PDPM and managed‑care as a major stressor. Retention is driven by purpose, resident connections and a positive team culture, especially relationships...
AI Model Detects Normally 'Invisible' Tissue Changes of Pancreatic Cancer at Stage 0
Researchers unveiled REDMOD, an AI radiomics framework that identifies stage 0 pancreatic ductal adenocarcinoma on routine CT scans. In a multi‑institutional study of 219 cancer cases and 1,243 controls, REDMOD flagged disease an average of 475 days before clinical diagnosis, achieving 73%...

Bowel and Ovarian Cancer Cases Rising Among Younger Adults in England, Research Reveals
A BMJ Oncology study of England’s cancer registry (2001‑2019) shows bowel and ovarian cancers are the only two types rising among adults under 50, while rates for older adults remain stable or decline. The analysis links excess weight to ten...

How AI, Value Based Care Bundles, Medicare Payment Compression, and IMG-Driven Residency Match Dynamics Will Reshuffle the Wealthiest Physician Specialty...
Four converging forces—AI augmentation, mandatory value‑based care bundles, ongoing Medicare fee‑schedule compression, and tightening IMG‑driven residency pipelines—are set to reshape the hierarchy of physician specialty earnings by 2030‑35. AI tools are boosting productivity in radiology, pathology and anesthesia while modestly...

Peptides / Bioregulators
The AGI House in San Francisco hosted the inaugural California Peptide Club, drawing over 100 invite‑only attendees to discuss self‑optimization peptides. Organiser Julius Ritter highlighted the fragmented information landscape and showcased stacks ranging from growth‑hormone releasers to cognitive enhancers. Days later, HHS...
AI, Face Photos May Predict Cancer Survival: Mass General Brigham Study
Mass General Brigham researchers validated FaceAge, an AI tool that estimates biological age from facial photographs, as a survival predictor for cancer patients. The study examined two routine photos per patient for 2,279 individuals, finding that changes in the Face...
Tonsillectomy Doesn’t Lead To Illness, But Tonsillitis Just Might
A recent UK Biobank analysis of nearly 500,000 people shows that higher rates of inflammatory disorders such as IBS among those who had tonsillectomy are actually tied to earlier tonsil infections, not the surgery itself. This finding overturns a 2018...

Beth Israel Lahey Health Rolls Out AI Scribe Systemwide
Beth Israel Lahey Health, a 14‑hospital system in Massachusetts, has selected Heidi as its systemwide AI scribe vendor. After a six‑month pilot involving 1,000 physicians, the health system began rolling out the technology to all providers. Clinicians reported higher note...

FOXO4-DRI Is Fascinating, but Was Never Intended for Human Use, What Are the Takeaways?
FOXO4‑DRI is an experimental senolytic peptide that selectively eliminates senescent cells by disrupting the FOXO4‑p53 interaction, prompting p53‑mediated apoptosis. Pre‑clinical studies across vascular, reproductive, musculoskeletal and renal models report improved endothelial function, restored testosterone production, chondrocyte rejuvenation, and reduced frailty....

Peptides / Bioregulators
A new study examined 6,441 gray‑market peptide samples covering 14 compounds, measuring purity, dose accuracy, and endotoxin levels. Between 41.6% and 71.1% of the products failed basic pharmaceutical standards, and 2.4% contained no active peptide at all. Endotoxin contamination appeared...

The BioPharm Brief: Expanding Access and Redefining Cancer Treatment
Henlius and Organon received European Commission approval for a pertuzumab biosimilar, expanding HER2‑targeted therapy access across the EU. Pfizer announced Phase 3 data showing its bispecific antibody Elrexfio improves progression‑free survival in relapsed or refractory multiple myeloma. Genexine disclosed pre‑clinical activity...

Living Vessel ATEV Offers New Hope for Dialysis Access
Humacyte’s ATEV could change the game for dialysis patients who run out of fistula options. It’s a living vessel that resists infection and needs fewer repairs. Major unmet need, big potential. Biotech

Hire Passion, Not Experience, to Cut Medicare Costs
"No one is waking up every day thinking about Medicare." They work to save seniors $1,100-plus annually on their Medicare. But Chapter CEO Cobi Blumenfeld-Gantz says he's never hired someone from the healthcare industry. Instead, Chapter hires for people excited about working...

Semiglutide Regenerates Cartilage Loss Through Weight Loss Independent Metabolic Restoration Mechanism
Semaglutide, a GLP‑1 receptor agonist originally developed for diabetes, is showing promise as a cartilage‑regenerating therapy. Preclinical pair‑fed mouse studies demonstrate cartilage protection independent of weight loss, while a 24‑week pilot in humans reported a 17% increase in cartilage thickness....
Real‑World OmegaAI Workflow: Beyond Demo Perfection
Most demos show you the perfect path. But what does the software actually feel like to use? This hands-on look at RamSoft’s OmegaAI + Blume pulls back the curtain—no fluff, just real workflow insights. 🔗 https://t.co/F9IGWOx3zl @Ramsoft #RadiologyWorkflow #healthtech

IV Fluids Recalled over Possible Contamination—Patients Face Risk of Vascular Complications
B. Braun Medical is recalling two lots of its 1‑liter Lactated Ringer’s IV electrolyte solution (lots J4P756 and J4S843) after detecting particulate matter that could trigger pulmonary emboli, vessel occlusions, or phlebitis. The affected bags were distributed across the United States...

Judy Faulkner: Profitability Is a ‘Side Effect,’ Not the Goal
Epic Systems, led by founder‑CEO Judy Faulkner, continues to prioritize patient‑centric software over profit maximization, even as its revenue climbs to about $6.7 billion in 2025. Faulkner describes profitability as a "side effect" of delivering value, a stance enabled by the...

Why Patient Understanding Is the Missing Metric in Medicine
Physician Joseph Rotella argues that patient understanding is the lone unmeasured metric in modern healthcare. He proposes a brief, standardized teach‑back moment at the end of every visit to confirm patients can articulate their care plan. The article highlights how...

Wastewater Surveillance Supports COVID-19 Screening in Hospitals
A retrospective study at University Hospital Basel linked SARS‑CoV‑2 concentrations in municipal wastewater to COVID‑19 positivity among asymptomatic patients screened on admission. The analysis of 75,667 PCR tests showed a 1.2% positivity rate, with stronger correlations during periods of high...
MDaudit Launches “Revenue Integrity Redefined” To Help Healthcare Organizations Reclaim Financial Control
MDaudit unveiled its "Revenue Integrity Redefined" campaign, a suite of AI‑driven tools and a maturity‑assessment resource aimed at moving health‑system revenue cycles from reactive denial handling to proactive risk control. The platform’s Meaningful AI framework embeds intelligence across coding, billing...

Higher Lp(a) Threshold More Closely Linked to Major ASCVD Events
A pooled analysis of three large randomized trials found that lipoprotein(a) levels of ≥ 175 nmol/L independently predict all‑cause mortality, cardiovascular death and stroke over seven years, with risk comparable to current smoking. The association was strongest in secondary‑prevention patients and was not...
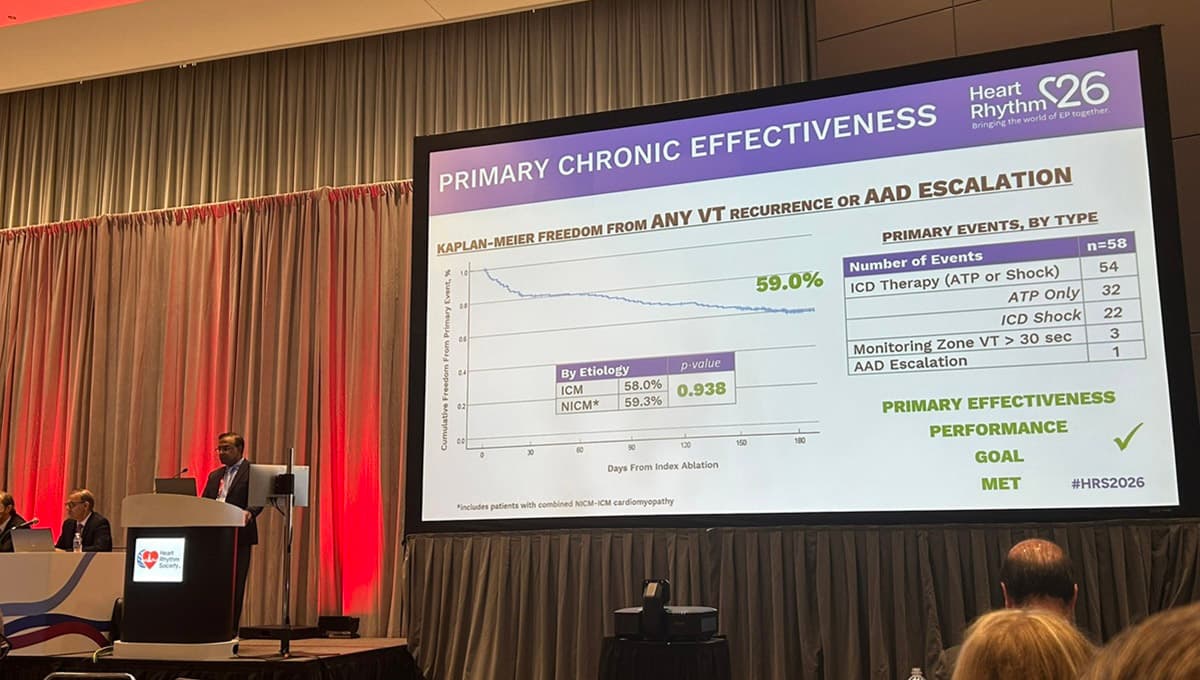
Ultralow-Temperature Cryoablation Shows Promise for VT
The FULCRUM‑VT IDE trial evaluated Adagio Medical’s vCLAS ultralow‑temperature cryoablation system in 209 patients with monomorphic ventricular tachycardia. At six months, 59% of participants remained free of recurrent VT, ICD interventions, and antiarrhythmic escalation, while acute success reached 98% non‑inducibility....

Allina Health Cybersecurity Chief Joins Baylor Scott & White
William Scandrett, former vice president and chief information security officer at Allina Health, has been appointed vice president of information security and chief information security officer at Baylor Scott & White Health. Scandrett announced the move on LinkedIn on April 28, highlighting his...

NPHI Execs: Quality Is Hospices’ Differentiator
At its annual Chicago summit, NPHI CEO Tom Koutsoumpas and President Carole Fisher argued that quality—beyond regulatory metrics—is the key differentiator for nonprofit hospices. NPHI’s Innovation Lab has launched quality dashboards that capture “Measures that Matter,” such as live discharges,...
HIV Drug (Maraviroc) Reverses Muscle Aging by Purging “Zombie Cell” Signals
Researchers are exploring the HIV CCR5 antagonist maraviroc as a senomorphic agent that could blunt muscle aging by dampening chronic SASP signaling. Modeling suggests a 75 mg once‑daily dose achieves high CCR5 occupancy, but human data on sarcopenia are absent. The...
ER Patients Highlight Measles Vaccine Gaps
A University of California, Riverside study of 2,459 emergency‑department patients across ten U.S. hospitals found significant gaps in measles‑mumps‑rubella (MMR) vaccine knowledge, status, and acceptance. Disparities were tied to race, language, insurance coverage, and limited primary‑care access, highlighting systemic barriers....

The IPO Buzz: Biotechs Back in Play
Biotech IPO activity revives as three companies—Avalyn Therapeutics, Hemab Therapeutics, and Seaport Therapeutics—file to raise a combined $601.1 million. Each plans to sell 11.8 million shares within a $16‑$18 price band, targeting roughly $200 million per offering. The deals follow a recent pulmonary‑fibrosis...

Alamar Biosciences IPO Raises $219.9M for Blood-Based Biomarker Tools
Alamar Biosciences closed an upsized IPO, raising $219.9 million by pricing 12.9 million shares at $17 each, and began trading on Nasdaq under the ticker ALMR. The California‑based firm offers the NULISA platform, which detects low‑level protein biomarkers in blood, and its...

STAT+: AIDS Group Sues Trump Administration over Undisclosed Agreement with Gilead
An AIDS activist group has sued the Trump administration for failing to disclose a research and development agreement that underpinned a 2025 settlement with Gilead Sciences over patents on HIV‑prevention drugs Truvada and Descovy. The settlement ended a lawsuit dating...

FDA Warns Public About Vascular Device After 3 Deaths
The FDA issued an early‑alert notice on Terumo Aortic’s Relay Pro Thoracic Stent Graft system after three deaths were linked to a failure of the device to unclasp from its delivery system. The warning applies to the non‑bare stent configuration sized...

HIMSS Chapter Advocacy Leader Issues Call to Action
Lawrence Voyten, chair of the HIMSS Chapter Advocacy Task Force, issued a public appeal urging healthcare professionals to become active members of their local HIMSS chapters. The call emphasizes the need for coordinated advocacy on pressing industry issues such as...

Meta‑analysis Finds 9% of Teens Tried Weight‑loss Supplements, Half Use Protein Products
A meta‑analysis published in JAMA Network Open, led by Harvard T.H. Chan School of Public Health and Australian universities, found that 9% of adolescents have tried nonprescription weight‑loss products and more than half of teen boys use protein supplements. The...

HIMSS Chapters Can Help Educate Policymakers
HIMSS is leveraging its local chapters to meet state legislators and discuss healthcare data security and patient rights. Lawrence Voyten, chair of the HIMSS Chapter Advocacy Task Force, says these meetings amplify HIMSS' policy voice. The initiative aims to translate...

Hospitals, Insurers Clash on 340B Rebate Costs
The Health Resources and Services Administration (HRSA) issued a request for information on reviving a 340B drug‑rebate model after a court‑blocked rollout. Hospital groups, led by the American Hospital Association, warn the model could impose over $1 billion in annual costs,...

FDA Accepts NDA for Zipalertinib, Targeting EGFR Exon‑20 Insertion NSCLC
Taiho Oncology, Taiho Pharmaceutical and Cullinan Therapeutics said the FDA has accepted a New Drug Application for zipalertinib, an oral EGFR inhibitor for patients with EGFR exon‑20 insertion NSCLC who have progressed after platinum chemotherapy. The filing is backed by...

CareTrust REIT Launches $628 Million Cross‑border Acquisition Spree
CareTrust REIT disclosed $628 million in acquisitions across North America and Europe, highlighted by a $380 million purchase of 15 skilled‑nursing facilities in California. The spree, funded through equity and debt, comes as the REIT earns an 8.8% blended yield and receives...

FastFinance: Medicaid Administrative Burdens; Cost Cutting Plans for 2026
HFMA’s FastFinance podcast highlighted growing Medicaid administrative burdens as states rush to upgrade programs ahead of the upcoming OBBBA reforms. A new HFMA report found that 49.5% of revenue‑cycle leaders are most concerned about automation, with 25% fearing errors will...

Cancer Care Costs: Views of Chinese, Asian Indian Patients
A new qualitative study reveals how Chinese and Asian Indian cancer patients and their caregivers experience financial toxicity, intertwining economic strain with cultural expectations of family responsibility. Interviews highlight indirect costs—lost income, transportation, and caregiving time—as major stressors that often...
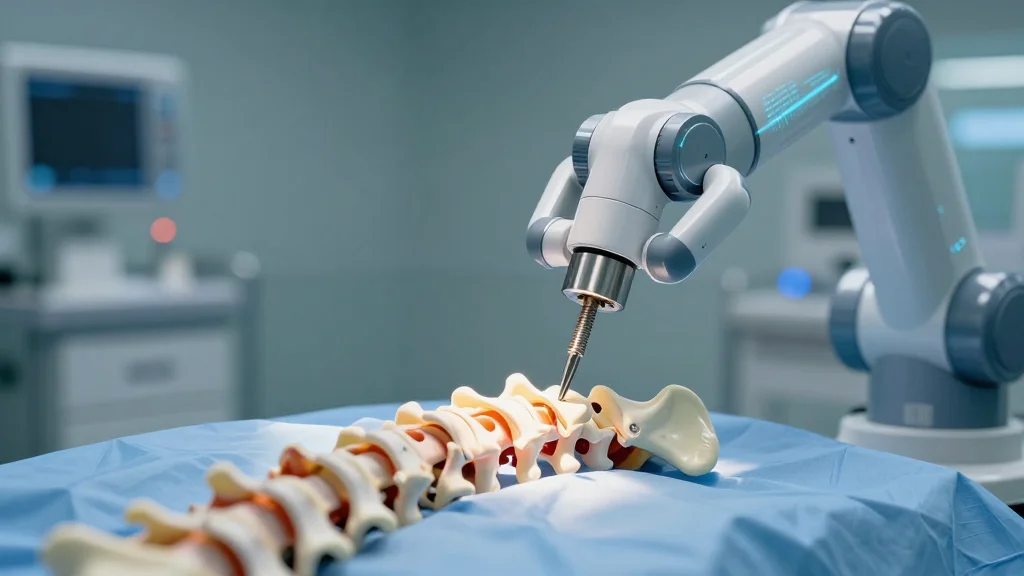
UC San Diego Health Performs First West Coast AI‑Robotic Spine Surgery
UC San Diego Health has completed the first AI‑driven robotic spine fusion on the West Coast, using a system that merges artificial intelligence, advanced imaging and robotic screw delivery. Surgeons say the platform boosts accuracy, reduces radiation exposure and could...

News 4/29/26
The Rural Health Transformation (RHT) fund announced this week will not close the gap left by recent Medicaid cuts, leaving rural providers scrambling for cash. Meanwhile, Central Maine Healthcare disclosed a layoff of 38 IT staff as it trims operating...

Eli Lilly Signs $2.25 B AI‑driven DNA‑editing Pact with Profluent Bio
Eli Lilly has entered a research agreement with AI‑focused Profluent Bio that could deliver up to $2.25 billion in development and commercial milestones. The deal targets kilobase‑scale DNA editing, a capability the partners say could unlock therapies for complex genetic disorders.

Importing Rapamycin to Save Money (2)
A community member placed a test order for sirolimus tablets from RL Pharma Jagdish in India via IndiaMart.com, paying a $25 wire transfer. The package left on Oct 16 2025 and arrived after 26 days on Nov 10 2025, experiencing delays in Zurich, JFK customs,...

Enhanced Cost Messaging Yields Minimal Change in Primary Care Clinic Selection: Tim McDonald, PhD, MPP, and Bryan E. Dowd, PhD
Researchers Tim McDonald and Bryan Dowd found that reducing information barriers about costs had only a marginal impact on how consumers chose primary‑care clinics within a tiered network. The randomized trial showed that most patients already followed the tiered benefit...