Know What's Happening in Healthcare
Know What's Happening in Healthcare

FDA approves KRESLADI, first CIRM‑backed gene‑editing therapy
The FDA cleared KRESLADI, a gene‑editing treatment from Rocket Pharmaceuticals for severe leukocyte adhesion deficiency‑I in children, eliminating the need for a bone‑marrow donor. It is the first product directly funded by California’s Institute for Regenerative Medicine to receive approval.
Also developing:
By the numbers: Syneron Bio raises $150M Series B

Medline is revising usage instructions for its home‑care adjustable beds after the FDA documented safety incidents involving hand‑control pendants that sparked, melted or ignited, and entrapment hazards from third‑party accessories. The agency recorded 12 injuries and two deaths—one fire‑related and one asphyxiation‑related—linked to five models in the Basic Bed franchise. Medline now urges customers to observe weight limits, keep components clear, hang pendants when idle, unplug beds unless adjustments are needed, and use only Medline‑branded side rails and accessories. The FDA also recommends leaving affected beds unplugged when not in use.

The FDA’s updated list of Section 503B outsourcing facilities details registration dates, annual fee requirements, and inspection status as of February 18, 2026. Facilities must complete initial registration and then re‑register each year between October 1 and December 31, otherwise they are delisted on January 1....

The National University of Singapore Academy for Healthy Longevity is hosting the Geromedicine Conference on February 26-27, 2026, bringing together geroscience researchers, clinicians, and industry leaders. The event emphasizes clinical translation of molecules such as NAD+ precursors, urolithin A, and ergothioneine,...

Identy is partnering with the volunteer NGO HumanCoop to roll out offline facial‑recognition tools for undocumented patients in northern Mauritania, creating portable digital medical IDs that work without internet. The initiative will initially cover the Bir Mogrein community of over 2,500...

Eli Lilly has agreed to pay CSL Ltd. $100 million to license the monoclonal antibody clazakizumab, which targets interleukin‑6. Under the agreement, CSL will keep development rights for the drug’s use in preventing cardiovascular complications in end‑stage kidney disease patients, currently...

Rapid functional testing is turning ambiguous genetic variants into actionable medical decisions. A zebrafish model proved a newborn‑identified SMN1 mutation benign, allowing clinicians to defer costly SMA therapy. This proof‑of‑concept shows whole‑genome sequencing can move from data to diagnosis within...

The FDA has escalated its enforcement against health‑fraud scams, targeting products ranging from 7‑OH opioid derivatives and tianeptine‑laden supplements to adulterated honey and toxic cosmetics. Recent public notifications and warning letters detail seizures, recalls, and advisories aimed at protecting consumers...

The FDA’s Center for Drug Evaluation and Research created the Office of Pharmaceutical Quality (OPQ) to standardize drug quality oversight across all manufacturing sites, both domestic and foreign. OPQ’s mandate spans new drugs, biologics, generics, biosimilars, over‑the‑counter products, and certain...

The FDA has accepted Bristol Myers Squibb’s protein‑degrading multiple myeloma candidate iberdomide for review, with a decision expected by Aug. 17 after a Phase 3 trial showed higher minimal residual disease‑negative rates. In contrast, the agency rejected Disc Medicine’s accelerated‑approval bid for...

Leonard Calabrese argues that ambient artificial intelligence—low‑cost, always‑on tools integrated into the exam room—can free clinicians from the constant EMR distraction and restore genuine face‑to‑face interaction. By handling routine documentation and patient‑message drafting, AI lets physicians focus on non‑verbal cues...

The College of American Pathologists issued the 2025 accreditation checklist on Dec. 9, adding a new “Digital Pathology Including Remote Data Assessment” section. The revision requires laboratories to validate digital pathology systems, document remote review locations, and treat remote CLIA‑certified sites...

This week on Lifers, Dr. Zeke Emanuel thinks you can eat the ice cream. Timestamps: (00:00) Preview (00:59) Intro (02:25) The inspiration behind the book (04:20) The importance of socialization and shared meals (07:33) Sustainability versus the short-term hustle (08:30) The truth about supplements and regulation (12:04) Sponsor:...

AI-driven clinical tools are rapidly improving diagnosis and treatment, but they leave patients exposed to hidden financial risks. A case of an autonomous AI ER physician resulted in a $37,000 out‑of‑network bill, highlighting systemic navigation failures. While clinical AI promises...

Innovaccer released the whitepaper "Autonomous Healthcare" outlining AI’s role in alleviating clinicians’ administrative load. The report highlights AI‑powered documentation, scheduling, and data integration tools that can reclaim up to 30% of physicians’ time. CEO Abhinav Shashank argues that these efficiencies...

Rox Heart Radio released a new episode titled “Why AI?” featuring host Roxana Mehran in conversation with cardiology researchers Ami Bhatt and Rohan Khera. The trio explores how artificial intelligence is poised to reshape cardiovascular care, from imaging interpretation to risk prediction....

Pharma launches are missing the mark 58% of the time, a failure rate driven by poor market insight and fragmented decision‑making. Accelerating market‑access planning from pre‑Phase 2 and integrating real‑world data can give companies a strategic north star. Purpose‑built AI tools...

Sarah Bridge, an emergency physician, recounts four years of frontline care in rural New Mexico’s Indian Health Service facilities, where chronic ICU bed shortages, equipment failures and staffing cuts force dangerous patient transfers and improvised treatments. She highlights how historical...

Vision loss remains a leading disability, prompting biotech firms to shift from symptom‑management to disease‑modifying therapies. Gene‑replacement approaches using AAV vectors dominate the inherited retinal disease pipeline, highlighted by Luxturna’s success and multiple late‑stage candidates such as Beacon’s laru‑zova and...

On November 20, 2020, Verb and Johnson & Johnson unveiled OTTAVA, a six‑armed robotic platform designed for minimally invasive surgery. The system integrates AI‑driven vision, haptic feedback, and modular tooling to automate complex suturing and tissue manipulation. Early trials report a 30%...

Livi, Europe’s leading digital‑first healthcare provider, has partnered with AI‑driven holistic platform CloudFit to launch an integrated preventative health solution. The collaboration lets Livi GPs refer patients to CloudFit’s fitness, nutrition and lifestyle coaching, creating a continuous care loop between...

The 2021 Consolidated Appropriations Act introduced mandatory broker compensation disclosures and broader pricing data for health plans, pushing plan sponsors to adopt fiduciary rigor similar to retirement plans. Yet, translating transparency into cost control remains difficult as procedure prices can...

Washington University in St. Louis deployed a clinical decision‑support workflow to curb over‑ordering of inpatient magnesium tests, a high‑volume assay often ordered by default on daily order sets. Analysis of Epic data showed half of the orders were routine "AM...

A wave of clinical trials is testing stem‑cell, gene‑editing and bionic‑eye technologies to treat blindness, especially retinitis pigmentosa and dry age‑related macular degeneration. Early‑stage studies from UC Davis, BlueRock Therapeutics and the University of Michigan report measurable vision gains, such as...

Ipca Laboratories posted a robust 19% year‑on‑year EBITDA increase to ₹530 crore, surpassing broker estimates, while revenue rose 6.6% to ₹2,400 crore. Domestic formulation sales grew 12% and export formulation climbed 17%, offset by flat API sales overall but a 26% rebound...

Recent high‑profile sexual‑abuse scandals, such as the Jeffrey Epstein case, are prompting a wave of trauma reactivation among survivors who had previously kept their experiences hidden. Patients often present with insomnia, irritability, increased alcohol use, or vague anxiety that they...

Medilink North of England provides a structured market‑access offering that helps MedTech and digital‑health innovators move beyond regulatory clearance to achieve commercial adoption in overseas health systems. The service is built around five pillars—market segmentation, global strategy, regulatory documentation, reimbursement...

Professor Segun Fatumo highlights the stark under‑representation of African genomes in global research, where over 86% of GWAS participants are of European ancestry despite Africa housing the greatest genetic diversity. He explains how this gap limits the accuracy of polygenic...

Healthcare providers are drowning in ever‑growing patient records, leading to missed follow‑ups, delayed appointments, and staff burnout. MVSPlus offers a dedicated virtual assistant that cleans, updates, and organizes charts in real time, integrating seamlessly with existing workflows. By handling routine...

Researchers identified interferon regulatory factor 7 (IRF7) as a master transcriptional driver that pushes vascular smooth muscle cells (SMCs) into a pro‑inflammatory, macrophage‑like state, a key step in plaque destabilisation. Single‑cell RNA sequencing and trajectory analysis uncovered an intermediate stem‑endothelial‑monocyte...
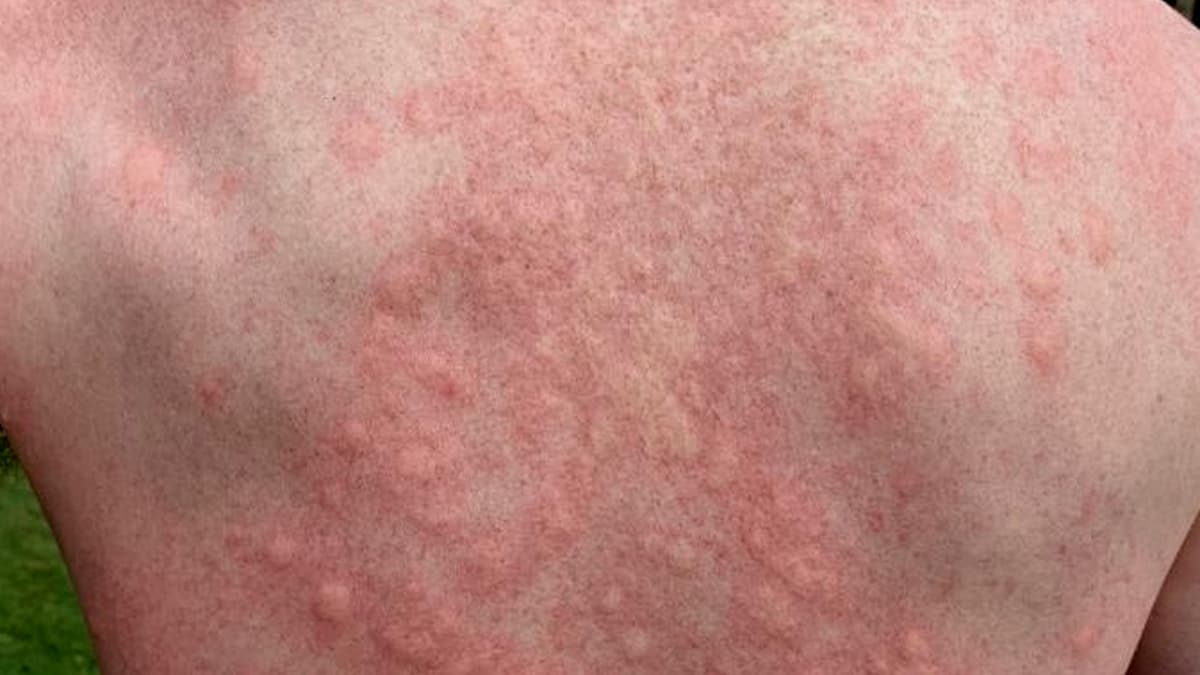
Novartis announced that its oral BTK inhibitor Rhapsido (remibrutinib) achieved significantly higher complete response rates than placebo in the phase 3 RemIND trial for the three most common forms of chronic inducible urticaria (CINDU). The drug, already approved for chronic spontaneous...

A “Variant of Unknown Significance” is a genetic finding that can’t yet be interpreted, leaving families with uncertainty instead of answers. Now, AI agents can help connect sequencing data with scientific literature and biological context to turn VUS into traceable, evidence-backed...

Legacy systems are piling up—and so is the risk. Great insights from @QuestDX on why ECM is the smartest, most scalable way to archive and protect healthcare data. ➡️ https://t.co/NcgrjGUq3D #HealthECM #HealthIT #HITSM

The Association for Diagnostics & Laboratory Medicine (ADLM) has issued a position statement urging Congress and federal regulators to update the Clinical Laboratory Improvement Amendments (CLIA) to explicitly govern AI tools used in clinical laboratories. ADLM warns that without modernized...
The pharmaceutical industry needs stability & clear guidance from #FDA as it attempts to bring through licensure new drugs & vaccines. Vinay Prasad, FDA's head of biologics, has brought drama to a setting where it's not welcome, @matthewherper.bsky.social writes. https://t.co/Qaw1QmYNpL

This book explains how disruptive technologies and emerging trends such as robotics, artificial intelligence, 3D printing, precision medicine or patient design will impact the manufacturing and distribution of pharmaceuticals in order to prepare successfully for a better future of healthcare. It's...

In this brief clip, host David Williams talks with Dr. Sarah Matt about the critical role of explainability in building trust for AI-driven healthcare. Dr. Matt emphasizes that patients and providers must understand how AI models make decisions to feel...

The FDA’s Moderna $MRNA pirouette is one more sign of chaos at the agency https://t.co/zThJqhT2BS via @matthewherper

#FDA backs down on #Moderna: The agency had refused to review the company's licensure application for an mRNA flu vaccine, but after significant pushback has relented. https://t.co/omo7bEBQGW
Korsana Biosciences announced a $175 million financing round led by Fairmount, Venrock, Wellington Management and TCGX to develop its Alzheimer’s antibody candidate KRSA‑028. The startup positions KRSA‑028 as a next‑generation “brain shuttle” designed to clear amyloid plaques more efficiently than existing...

$MRNA says FDA will now review its flu vaccine. PDUFA date is Aug. 5. https://t.co/npZzeEa5nk

FDA agrees to review Moderna flu vaccine application, splitting review into standard approval for those under 65 and accelerated approval for those 65 and above. $mrna https://t.co/HMMlPm0PGh
![[Clinical Rounds] A Multidisciplinary Management Approach to Cervical Cancer During Pregnancy](/cdn-cgi/image/width=1200,quality=75,format=auto,fit=cover/https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/5281f8873aca1a3c24d65c4c50a98a5f.webp)
A 39‑year‑old gravida 3 woman presented at 10 weeks gestation with vaginal bleeding and a prior diagnosis of high‑grade squamous intraepithelial lesion (HSIL) that had not been followed up. Cervical cancer testing confirmed HSIL while the intrauterine pregnancy remained viable. The...

Epetōme, the UK‑based gut‑health brand founded by nutritionist Emily English, has secured a multi‑million‑pound investment led by Active Partners and Redrice Ventures. The funding will accelerate its international rollout and fund expanded clinical trials on probiotics and prebiotics. Since its...

Carterra unveiled Vega, the industry’s first 48‑channel high‑throughput SPR platform, delivering roughly 12‑fold higher screening capacity for small‑ and large‑molecule drug candidates. Covaris introduced the truCOVER® Total RNA Library Prep Kit, enabling robust RNA‑seq libraries from as little as 10 ng...

AI medical advice: Not ready for prime time. A study by the Icahn School of Medicine at Mount Sinai, published in The Lancet Digital Health, reveals vulnerabilities in medical AI systems. https://t.co/ArD323yhzL #research #AI #healthcare #medicine

The FDA removed a consumer webpage that warned against chlorine dioxide, raw camel milk, chelation and hyperbaric oxygen as ineffective autism treatments, citing a routine cleanup of outdated content. The page, unchanged since 2019, remains only via the Internet Archive,...

Immedica’s pegzilarginase (Loargys), the first enzyme replacement therapy for arginase‑1 (ARG1) deficiency, has received NICE endorsement for NHS use in the UK. The weekly IV or subcutaneous treatment cuts blood arginine levels by roughly 80% and is recommended for patients...

The UK government, via Innovate UK, has launched a £20 million funding programme to accelerate development of medicines, medical devices, wearables, virtual‑reality therapies and AI‑enabled tools for drug and alcohol addiction. Grants of up to £10 million for late‑stage projects and up to...

Portal Diabetes has secured FDA breakthrough device designation for its implantable Portal Pump, an insulin delivery system that pairs an abdominal pump with continuous glucose monitoring and concentrated insulin. The company also launched a Phase I study of temperature‑stable Portal Insulin...

Janux Therapeutics has dosed the first participant in a Phase I, open‑label, dose‑escalation trial of JANX011, its inaugural candidate from the adaptive immune response modulator (ARM) platform. The bispecific antibody targets CD19‑expressing B cells to achieve a deep, lasting immune reset...