Healthcare News and Headlines
Healthcare News and Headlines
Healthcare Pulse
EMAIL DIGESTS
Daily
Every morning
Weekly
Tuesday recap

Employers are feeling heightened pressure from medical inflation, specialty‑drug spend and regulatory complexity, prompting a shift toward cost control as the top benefits priority, according to MetLife’s 2026 U.S. Employee Benefit Trends Study. Controlling benefit costs has become the number‑one objective for the first time since 2022, reflecting its direct impact on profitability and talent attraction. Benefit leaders are therefore seeking data‑driven strategies that address cost drivers at their source rather than simply raising employee contributions. Dependent verification auditing emerges as a concrete, measurable solution.

The Consolidated Appropriations Act of 2026 bans pharmacy benefit managers (PBMs) from receiving compensation tied to drug list prices and mandates 100% rebate pass‑through to plan sponsors by August 2028. The law also allocates roughly $190 million to the Centers for...

Private equity firms are intensifying focus on hospice care platforms, with five notable deals reported this quarter. Kinderhook, Renovus and Revelstoke are among the investors leading the charge, targeting fragmented providers to build scalable platforms. The surge reflects broader demographic...

The FDA’s Drug Trials Snapshots program publishes concise, consumer‑focused summaries of the demographic makeup and outcomes of pivotal clinical trials for newly approved drugs. Launched in 2015, the tool covers only New Molecular Entities and original biologics approved after that...

Ionis Pharmaceuticals received FDA acceptance of its supplemental NDA for olezarsen and a priority‑review designation, with a PDUFA action date of June 30, 2026. The decision is backed by two Phase III CORE studies—CORE (n=617) and CORE2 (n=446)—that evaluated once‑monthly subcutaneous...

The American Heart Association’s new scientific statement projects that nearly six in ten U.S. women will live with cardiovascular disease (CVD) by 2050, driven by rising hypertension, obesity, and diabetes. By 2050, high blood pressure could affect almost 60% of...

GSK’s depemokimab, marketed as EXDENSUR, received FDA approval on December 16, 2025 as a six‑month subcutaneous add‑on for severe eosinophilic asthma in patients aged 12 and older. Approval was based on two 52‑week, double‑blind, placebo‑controlled trials (SWIFT‑1 and SWIFT‑2) that...

BrightSpring Health Services reported a strong fourth‑quarter and full‑year 2025, with net revenue climbing 28% to $12.9 billion and adjusted EBITDA rising 34% to $618 million. The company turned a net loss in 2024 into a $104.8 million profit for the year and...

UW Medicine and Seattle Children’s Hospital received $328,133 from the Sudden Unexplained Death in Childhood (SUDC) Foundation to launch a four‑year study using PacBio HiFi long‑read sequencing as a first‑line test. The project will sequence 200 child‑parent trios to uncover...

The FDA issued a suite of new guidance documents in early 2026 aimed at easing development of therapies for rare diseases, including the Rare Disease Evidence Pathway, Plausible Mechanism Pathway, and innovative trial designs for cellular and gene therapies. The...

Quotient Sciences has extended its commercial partnership with Ipsen to manufacture Sohonos (Palovarotene), the approved therapy for fibrodysplasia ossificans progressiva (FOP), an ultra‑rare disease affecting fewer than 1,000 people worldwide. The deal includes Ipsen's investment in a pneumatic closed‑transfer system...

ENDRA Life Sciences announced that its Taeus Liver device achieved high measurement consistency in a MASLD study, recording an intraclass correlation coefficient of 0.89 and a standard error of measurement of 3.3%. The trial involved 14 participants and 56 data...

Eli Lilly disclosed Phase III ACHIEVE‑3 results showing its oral GLP‑1 agonist orforglipron outperformed oral semaglutide in lowering A1C and inducing weight loss. The 52‑week, open‑label trial enrolled 1,698 patients across six countries and compared two doses of each drug. Orforglipron also...

The American College of Radiology (ACR) has urged the Department of Health and Human Services (HHS) to overhaul its AI reimbursement framework, labeling the current policy unsustainable. In a February 19 letter, ACR highlighted that without payment models tied to...
PocDoc, the UK’s leading digital diagnostics firm, has teamed up with the E4 Primary Care Network to launch a mobile health‑check service for over 80,000 residents in Chingford’s underserved areas. The initiative uses PocDoc’s Neighbourhood Bus to deliver a finger‑prick...

RadNet’s TechLive remote‑scanning platform earned European CE certification, extending its vendor‑agnostic, multimodality control to EU hospitals. The solution, already cleared in the U.S., lets technologists operate MR, CT, PET/CT and ultrasound machines from off‑site consoles. RadNet reports a 42% drop...

Bayer announced that its phase‑III PEACE‑3 trial demonstrated a statistically significant overall‑survival benefit for the combination of Xofigo (radium‑223) and enzalutamide versus enzalutamide alone in patients with metastatic castration‑resistant prostate cancer (mCRPC) and bone metastases. Median overall survival extended to...

Heidi AI has partnered with Streamliners to embed HealthPathways into its AI‑driven clinical documentation platform, giving clinicians instant access to trusted care pathways. The Australian Digital Health Agency rolled out a digital‑health teaching toolkit for universities, addressing a gap where...

Europe’s MedTech landscape this week is defined by heightened public funding, regulatory simplification, and a selective capital shift toward high‑impact digital health. Ireland unveiled a €34.3 m ARC Hub, while Horizon Europe health calls target AI‑driven projects such as digital‑mental‑health and...

Healthcare CIOs are moving AI from experimental pilots to core infrastructure, emphasizing measurable outcomes over hype. At Vive 2026 and HIMSS 2026, leaders highlighted AI’s role in streamlining documentation, revenue‑cycle tasks, and patient‑portal interactions. Simultaneously, state‑level AI regulations—such as Texas’...

Prime exits® and American HealthCare Capital are offering a fully licensed behavioral health platform in Northern Colorado, comprising two ready‑to‑operate outpatient facilities. The business currently generates no revenue but retains all licenses, accreditation, EMR, billing systems and payer contracts, dramatically...

A national healthcare staffing firm generated roughly $16.4 million in 2025 revenue and $3.7 million gross profit while operating two divisions—Health Information Technology (HIT) and travel nursing/allied health. The company leverages a proprietary database of over 174,000 clinicians and HIT professionals and...

A Texas‑based orthotics and prosthetics provider reports $1.22 million in 2025 revenue, marking a rebound after a dip in 2024. The family‑run firm delivers a full suite of services, from microprocessor‑controlled prostheses to diabetic foot care, and operates multiple clinics across...

Taiwan’s Academia Sinica and National Taiwan University Hospital have launched PanMETAI, an AI‑integrated metabolomics platform that analyzes roughly 260,000 metabolic signals from a 500‑microliter blood‑serum sample. Using deep‑learning on nuclear magnetic resonance data, the model achieved a 99% area‑under‑curve for distinguishing...

PACS Group reported FY 2025 revenue of $5.29 billion, up 29% year‑over‑year, and net income of $191.5 million, driven by strong occupancy and disciplined acquisitions. Occupancy averaged 89.1% overall and 94.9% at mature facilities, well above the industry average. More than 73%...

Recent warnings highlight that many over‑the‑counter pain and arthritis products are contaminated with undisclosed pharmaceutical ingredients. These items are often marketed as dietary supplements or all‑natural remedies, sold both online and in brick‑and‑mortar stores. The hidden substances can cause severe...

Third Way Health announced an oversubscribed $15 million Series A round led by Health Velocity Capital, with Kristin Torres Mowat as the lead investor. The new capital lifts the company’s total funding to $22.5 million, earmarked for product development and scaling operations. The...

The FDA has warned consumers against buying ULTRA ADVANC3 on Amazon and ULTRA ADVANC3 GOLD on NaturistaRex after laboratory tests revealed hidden prescription drugs. The supplements contain undeclared dexamethasone, diclofenac, and methocarbamol, each carrying serious health risks. The agency advises immediate discontinuation and...

Elevance Health, formerly Anthem, is deploying artificial intelligence across its operations while drawing a firm line against using AI for claim denials. The insurer leverages AI to flag incomplete claims, accelerate prior‑authorization approvals, and power virtual assistants that guide members...

In a recent informal briefing, Dr. Jayne was thanked for a public mention that highlighted her contributions. The discussion veered into a grammatical debate over the present perfect indicative, underscoring the audience’s attention to language precision. Participants also touched on...

Fulgent Genetics reported Q4 2025 revenue of $84.1 million, a sequential increase and strong year‑over‑year growth across its Precision Diagnostics and Biopharma segments. Non‑GAAP gross margin improved to 44.3% while operating expenses fell, delivering a $0.7 million adjusted EBITDA and a $4.5 million...

An expression of concern has been issued for a 2003 Nature paper that reported the gene product Murr1 restricts HIV‑1 replication in resting CD4⁺ lymphocytes. The editors identified duplicated control panels in Figure 3b, which were meant to demonstrate siRNA activity,...

Treace Medical Concepts reported third‑quarter 2025 revenue of $50.2 million, an 11% year‑over‑year increase, while adjusted EBITDA loss narrowed 49% to $2.6 million. The mix shifted toward lower‑ASP bunion correction systems, pulling gross margin down to 79.1% from 80.1% a year earlier....

Senator Ron Johnson (R‑WI) is urging Congress to draft a “Right to Try 2.0” bill that would force the FDA to grant broader, faster access to experimental therapies for rare diseases. FDA Commissioner Martin Makary pushed back, defending the agency’s...

Catalyst OrthoScience announced FDA 510(k) clearance for its Archer® Patient‑Specific Instrumentation (PSI), a suite of humeral and glenoid guides designed to translate 3D pre‑operative plans into the operating room. The system pairs with Archer® 3D Planning Software, supporting both anatomic...
![[Comment] Language in Rare Disease: A Call for Systemic and Empathetic Action](/cdn-cgi/image/width=1200,quality=75,format=auto,fit=cover/https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/82158fe01b2f637b44373e1d263a5ee3.webp)
Globally, roughly 300 million individuals live with a rare disease, confronting isolation, diagnostic delays, and fragmented care. The Rare Diseases International–Lancet Commission argues that the language used by clinicians perpetuates stigma and hampers coordinated treatment. It calls for a systemic overhaul...
Researchers have adapted lymphovenous bypass, a microsurgical technique that reroutes lymphatic fluid into the venous system, as a potential treatment for Alzheimer’s disease. In a Phase I pilot study of 20 patients, the procedure reduced cerebral amyloid‑β levels by roughly 30%...

TrueScan, a whole‑body MRI center in St. Peterburg, Florida, has begun offering AI‑powered coronary CT angiography (CCTA) from Heartflow, available as a standalone test or bundled with its MRI packages. The move addresses growing patient and physician demand for coronary imaging...

NASA conducted its first-ever medical evacuation from the International Space Station in January 2026 when astronaut Mike Fincke experienced a microgravity‑related health event. The entire Crew‑11 returned early aboard a SpaceX capsule because no spare crew‑ready vehicle was available. The...
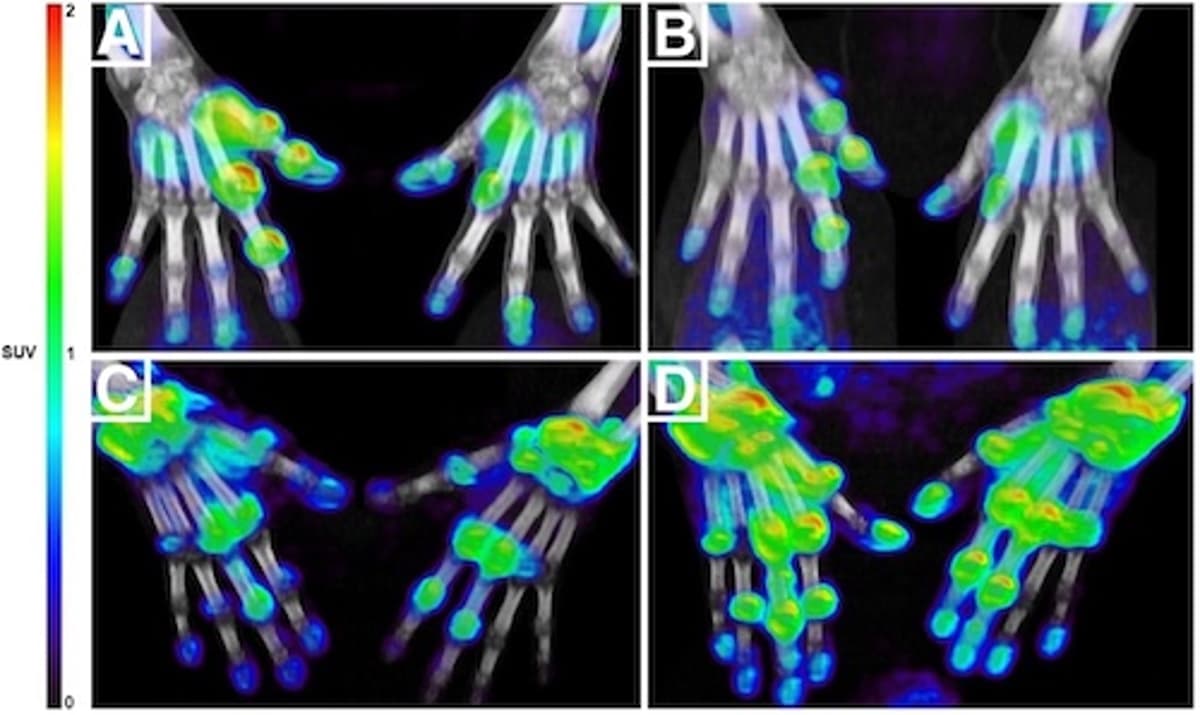
A new macrophage‑targeted PET/CT tracer (11C‑DPA‑713) can identify rheumatoid arthritis patients who will respond to anti‑TNF therapy within four weeks, far earlier than the traditional three‑to‑six‑month window. The study of 20 patients showed that standardized uptake values (SUVs) at baseline...

The U.S. Department of Justice filed an amicus brief supporting AbbVie in its effort to overturn a Colorado statute that prohibits pharmaceutical manufacturers from limiting discounts under the federal 340B drug pricing program when hospitals use contract pharmacies. The Colorado...

The American Hospital Association and Epic have launched the Safer Births Post‑Partum Hemorrhage Collaborative, a seven‑month program aimed at cutting postpartum hemorrhage rates. The initiative combines monthly expert‑led webinars, peer‑to‑peer forums, and dedicated office‑hours for hands‑on EHR guidance. Participants gain...

Collective Health has unveiled Collective AI, an AI‑powered benefits assistant built with Google Cloud. The system answers member queries, supplies real‑time data to customer‑service reps, and offers employers plan‑design insights to streamline enrollment. Leveraging Google Cloud’s privacy‑by‑design infrastructure, the tool...

Alignment Healthcare posted a fourth‑quarter loss of $11 million, but revenue rose 44 percent to just over $1 billion and membership jumped 25 percent to 236,300. The loss narrowed sharply from $31 million a year earlier, and the full‑year loss shrank to $724 000 versus $128 million...
Long COVID remains clinically fragmented as the CDC, WHO and the National Academy of Sciences each use slightly different definitions, creating gaps in diagnosis and treatment equity. High‑risk groups—women, older adults, smokers and those with obesity—continue to experience disproportionate symptom...

The Senate Health, Education and Labor Committee approved the bipartisan Health Care Cybersecurity and Resiliency Act by a 22‑1 vote, with only Sen. Rand Paul dissenting. The legislation obliges the Department of Health and Human Services to craft a cybersecurity...

GE HealthCare and UCSF Health have signed a 10‑year Care Alliance to embed advanced imaging technology throughout UCSF’s expanding health system. The partnership will support the construction of two new hospitals—an adult facility at Parnassus Heights and a pediatric center...

The FDA granted accelerated approval to zongertinib (Hernexeos) for adults with unresectable or metastatic non‑squamous NSCLC that carry activating HER2 TKD mutations, extending its use to treatment‑naive patients. The decision rests on the Beamion LUNG‑1 trial, which reported a 76%...

ARUP Laboratories has unveiled the Innovation Central Laboratory, a dedicated hub for co‑development with pharmaceutical and biotech partners. The facility is designed to validate technologies and move next‑generation diagnostics from concept through to commercial readiness. It will host projects ranging...

The FDA approved KOMZIFTI (ziftomenib) on November 13 2025 as an oral 600 mg capsule for adult patients with relapsed or refractory acute myeloid leukemia (AML) harboring an NPM1 mutation. Approval rests on the single‑arm KO‑MEN‑001 trial, which enrolled 112 participants across seven...