
Interventional Radiologists Launch New Chain of Standalone Outpatient IR Centers
Two California interventional radiologists have launched Elmnt IR, a chain of standalone outpatient interventional radiology centers, beginning with a flagship location in Los Angeles. The venture targets high‑impact conditions such as benign prostatic hyperplasia, uterine fibroids, knee osteoarthritis, and select oncology procedures, aiming to deliver minimally invasive care outside traditional hospital settings. Co‑founders Jonathan Steinberger and Gabriel Lipshutz emphasize seamless referral pathways that keep referring physicians central to patient management. The company is backed by New Dimensions Health Fund, which invests $50‑$250 million in healthcare services.

AstraZeneca Reports the P-III (I CAN) Trial Data on Ultomiris for IgA Nephropathy
AstraZeneca announced interim Phase III (I CAN) data for Ultomiris in IgA nephropathy, enrolling about 510 high‑risk adults. The trial met its primary endpoint, showing a roughly 30% reduction in 24‑hour proteinuria at 34 weeks, with benefits evident as early as 10...

Voice Therapy App Improving Outcomes for Parkinson’s Patients at University Hospitals of Derby and Burton
University Hospitals of Derby and Burton introduced a voice‑therapy app for Parkinson’s patients, enabling home‑based exercises after an initial in‑person assessment. The pilot saved 14‑17 hours of clinical time each month, equivalent to six extra therapy slots per week, and...

Mainetti Launches MAMI – A Non-Profit Initiative Supporting Premature Infants in Neonatal Intensive Care Units Across Italy
Mainetti Italia and the Cuore di Maglia Association have launched MAMI, a non‑profit initiative that supplies CE‑certified therapeutic fabric gloves to premature infants in neonatal intensive care units (NICUs) across Italy. The gloves, made from fire‑proof fabric and polypropylene granules,...

Trelleborg Costa Rica Site Earns ISO 13485 Certification
Trelleborg Medical Solutions' Costa Rica manufacturing site earned ISO 13485:2016 certification, confirming its quality management for medical devices. The 107,600‑sq‑ft facility, opened in December 2025 in the Evolution Free Zone, is the company’s first Central American plant and supports nearshoring and supply‑chain...

ICR Bolsters Healthcare Unit
ICR announced two senior hires to strengthen its healthcare communications franchise: John Capodanno was named head of U.S. healthcare corporate communications, and Matthew Willey was appointed managing director of the medical‑technology and diagnostics practice. Capodanno brings over 25 years of...

Utepreva Launches Utepreva Endometrial Sampler to Support Early Diagnosis of Endometrial Cancer
Utepreva has introduced the Utepreva Endometrial Sampler, a single‑use device aimed at improving tissue capture for early endometrial cancer diagnosis, slated for market release in October 2026. The sampler integrates a sponge tip, plunger‑driven suction, and a collection chamber to...

Fraunhofer IAP and NMI Achieve Biomimetic Tissue Mechanics
Fraunhofer IAP and NMI have created a patent‑pending biomimetic tissue substitute that combines a polyurethane acrylate base, a 3D‑printed wavy metastructure, and an electrospun collagen surface. The metastructure reproduces the nonlinear stiffness of pericardial tissue, while the collagen layer supports...

ViewsML Closes Funding Round to Accelerate Commercialization of AI-Driven Virtual Biomarker Staining Platform
ViewsML announced an oversubscribed $4.9 million seed round led by Wittington Ventures, with new investors Mayo Clinic and Continuum Health Ventures joining repeat backers. The capital will fast‑track commercialization of its AI‑driven virtual biomarker staining platform, which extracts per‑cell biomarker insights...

Iran War Pushing up Till Price of some Drugs by up to 30%, Pharmacies Warn
The Iran‑U.S. conflict has driven up fuel and air‑freight costs, pushing wholesale drug prices in England up 40‑50%. Community pharmacies now charge 20‑30% more for over‑the‑counter painkillers and hay‑fever tablets, with a 100‑tablet pack of paracetamol rising from £0.41 ($0.52)...
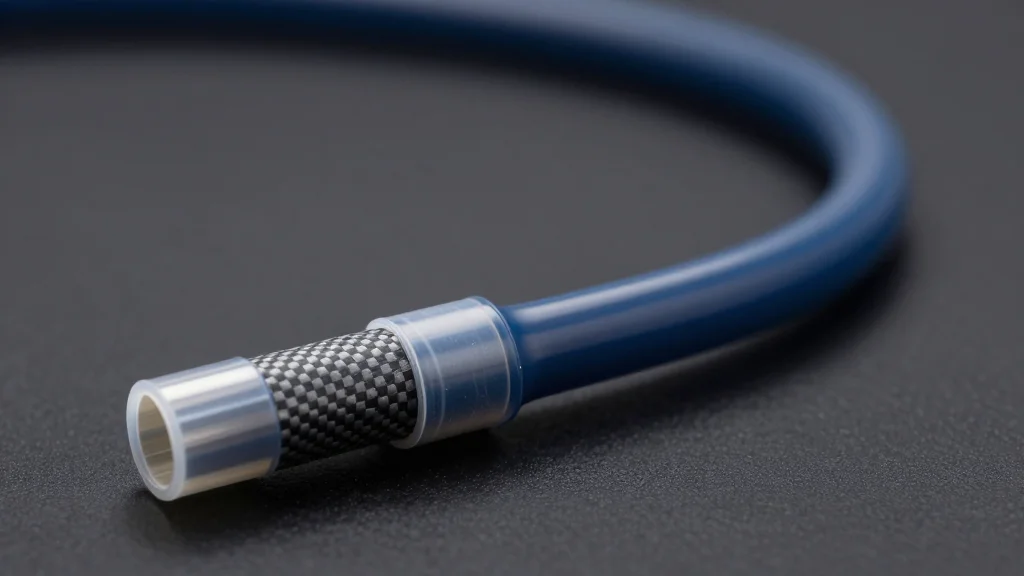
Engineering Insights: Polyimide, PTFE & Reinforced Tubing for Next-Gen Devices
MicroLumen’s white paper spotlights polyimide and PTFE‑polyimide composite tubing as pivotal for next‑generation catheters. The thermoset polyimide delivers unmatched dimensional stability under mechanical and thermal loads, while PTFE adds low‑friction surfaces. Engineers can tailor torque, pushability, and flexibility through braided...

How States Can Solve Mental Health Workforce Shortages
Emergency department visits for suicide attempts more than tripled between 2015 and 2020, highlighting a critical need for robust suicide screening in hospitals. Yet about half of accredited hospitals report staffing shortages that impede consistent, evidence‑based care. Roughly 40% of...
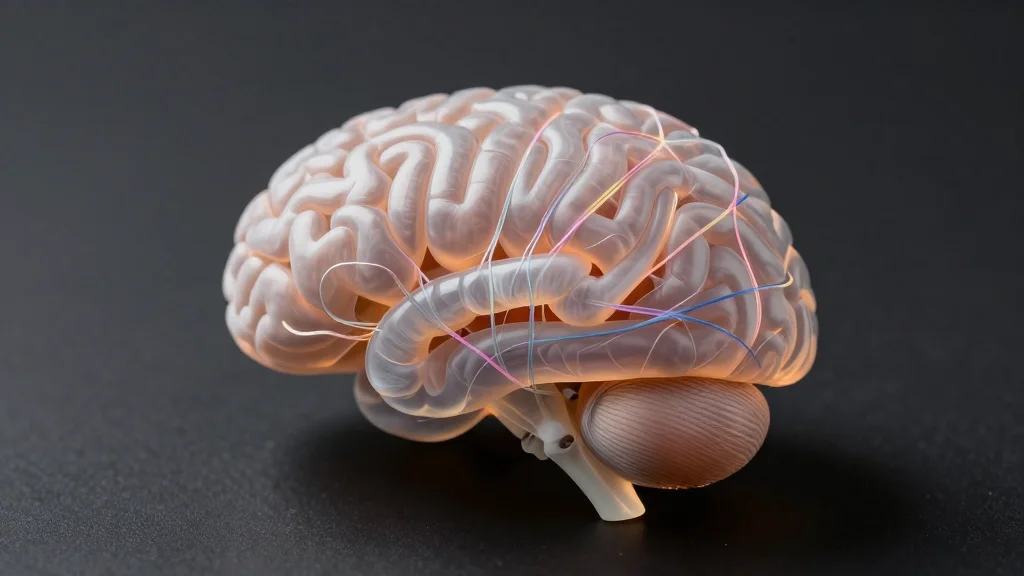
FMRI-Based Mega-Study of Psychedelics Reveals Patterns of Brain Signaling Reorganization
An international consortium analyzed resting‑state fMRI scans from over 250 healthy volunteers who received psilocybin, LSD, DMT, mescaline or ayahuasca, creating the largest pooled dataset of psychedelic brain imaging to date. Using a unified processing pipeline and Bayesian hierarchical modeling,...

State High Court Weighs in on Woman Taken for Organ Donation But Was Still Alive
A Mississippi woman declared brain‑dead was taken to the state organ recovery agency, only to revive with signs of life, prompting lawsuits alleging negligence and unauthorized sedation. The daughter’s bystander claim was dismissed, while the broader question of whether the...

How to Scale a Private Medical or Dental Practice From One to 100 Locations
Paul Vigario, CEO of SurfCT, argues that scaling a private medical or dental practice from one to 100 locations hinges on building a flawless first office rather than perfecting clinical skills or splurging on equipment. He warns that perfectionism and...

Strata 2026 Healthcare Financial Outlook Report: Medicaid Cuts and Labor Costs Top CFO Concerns
Strata Decision Technology’s 2026 Healthcare Financial Outlook finds Medicaid reimbursement cuts and rising labor expenses now dominate CFO agendas, with 66% naming Medicaid cuts as their top concern. While 43% anticipate modest margin growth, 25% expect declines, underscoring a focus...

Muons, Massive Waves and Restored Sight: The Winners at the ‘Oscars of Science’ – Podcast
The Breakthrough Prize, dubbed the "Oscars of science," handed out $3 million awards in physics, mathematics and life sciences at a high‑profile ceremony in Los Angeles. Jean Bennett was among the laureates, receiving the life‑sciences prize for co‑creating the first FDA‑approved gene‑augmented...

InteliCare to Enter New Sector with Warrigal Care Contract for 65 Retirement Villas
InteliCare Holdings signed a three‑year, $200,000 Master Subscription Agreement with Warrigal Care to install its AI‑driven sensor platform in 65 retirement villas, marking the company’s first foray into the retirement‑village market. The deal opens a pathway to expand across Warrigal’s...
Before Vaccines, Diphtheria Used to Kill Hundreds Each Year. Now It’s Back in Australia
Australia is witnessing a resurgence of diphtheria, with the Northern Territory reporting 17 respiratory cases and 60 cutaneous cases in the past year, and Western Australia’s Kimberley region logging 27 cases in the last month. The outbreak extends to Queensland...
New Study Offers Insight Into the Type of AI Radiologists Prefer
Researchers at Moffitt Cancer Center evaluated 200 CT‑scan reports generated by a general‑purpose large language model (LLM) and a domain‑specific AI fine‑tuned on radiology and oncology data. Radiologists consistently preferred the specialized model, citing greater completeness, accuracy and conciseness, and...

US Health Secretary Kennedy Defends Trump’s Mathematically Impossible Drug Discount Claims
U.S. Health Secretary Robert F. Kennedy Jr. defended President Donald Trump’s mathematically impossible drug‑discount claims during a Senate Finance Committee hearing, insisting a drop from $600 (≈ $566 USD) to $10 represented a 600% reduction. The assertion contradicts basic percentage math, as the actual discount...

Surgeon Calls PowerPort Catheter Infection Warnings Inadequate in Federal Trial
An expert radiologist testified that Bard’s PowerPort catheter instructions downplay infection risk, classifying it as a “possible complication” without stating the roughly 14% infection rate. The plaintiff, Robert Cook, suffered sepsis after his implanted port became infected, delaying his colorectal...

NSW Opens $96M RNA Research and Manufacturing Facility
New South Wales has launched a $96 million (≈$63 million USD) RNA research and manufacturing hub at Macquarie University’s Innovation Precinct. The state‑run facility, operated by Aurora Biosynthetics, will produce RNA‑based vaccines and therapeutics from a single site. NSW pledged an additional...

This Routine Heart Scan Sees the Danger Coming Long Before Symptoms Strike
Researchers at Kumamoto University demonstrated that adding a delayed imaging phase to a standard cardiac CT scan enables measurement of Late Iodine Enhancement (LIE) and Extracellular Volume (ECV) fractions. In a cohort of 1,207 patients tracked for an average of...

Chemed Corp (CHE) Q1 2026 Earnings Call Transcript
Chemed Corp reported mixed Q1 2026 results, with VITAS hospice admissions rising 6% to 17,419 but revenue growth and EBITDA margin lagging due to a shift toward higher‑margin hospital short‑stay patients. Roto‑Rooter saw residential revenue decline 3.1% and a 21%...

West Pharmaceutical Services Inc (WST) Q1 2026 Earnings Call Transcript
West Pharmaceutical reported net sales exceeding $3 billion with over 4% organic growth, driven by high‑value product (HVP) components and a 50% surge in GLP‑1 revenue. Fourth‑quarter revenue reached $850 million, up 7.5% reported and 3.3% organically, while adjusted EPS rose 12%...

HHS Still Developing Long COVID Biomarkers, Online Patient Resource Hub
U.S. Health and Human Services Secretary Robert F. Kennedy Jr. announced that HHS is still working to identify biomarkers for Long COVID and is creating an online patient‑physician resource hub. The effort follows criticism that the previous administration dismantled the...

RFK Jr. Defends Vaccine Guidance Changes and Health Spending Cuts
Health Secretary Robert F. Kennedy Jr. testified before the Senate, defending recent vaccine‑policy shifts and a sweeping health‑spending reduction. He highlighted a rapid response to the nation’s worst measles outbreak in decades and pledged transparency while asserting that the incoming...

How the DOJ Is Tackling Fraud in the ACA Marketplace
At a Las Vegas conference, the DOJ highlighted a growing fraud scheme in the ACA Marketplace that preys on homeless, unemployed and mentally‑ill Medicaid recipients. Street marketers falsify denials, open special enrollment periods, and become agents of record, repeatedly switching...
Can Doctors Discuss a President's Mental Health? Why Ethics and Public Risk Collide
The BMJ opinion piece by David Nicholl and Trisha Greenhalgh examines whether doctors can publicly discuss a U.S. president’s mental health. It reiterates that professional standards prohibit diagnosing without a direct clinical exam, especially for complex conditions like behavioral variant...

Paragon: Reforming Policies That Drive Hospital Costs Key To Tackling Health Spending
The Paragon Health Institute released a new report, "The Hospital Cost Crisis," identifying government policies as a primary driver of soaring hospital prices and consolidation. The think‑tank proposes 12 policy reforms, notably site‑neutral payment adjustments and changes to the 340B...

2027 Medicare Advantage – Part D Final Rule Summary
The Centers for Medicare & Medicaid Services issued a final rule on April 6, 2026 that overhauls Medicare Advantage (Part C) and the Medicare Prescription Drug Benefit (Part D) for coverage starting January 1, 2027. The rule implements changes mandated by the Inflation Reduction Act and...

‘They’ve Turned the Dial up on Denying Preauthorizations’: CHS Blames Payers for Volume Drop
Community Health Systems (CHS) reported a first‑quarter drop in same‑store inpatient admissions (‑1.3% YoY) and surgeries (‑2.2%), attributing the decline to aggressive payer pre‑authorization denials and macro‑economic anxiety among patients with high‑deductible plans. The pressure was most pronounced among commercial...
Interventional Radiology Procedure Offers Relief From Painful Blood-Clot Side Effect
A new NIH‑sponsored trial, C‑TRACT, evaluated catheter‑directed stent placement for patients with post‑thrombotic syndrome (PTS) after deep‑vein thrombosis. The study enrolled 225 participants across 29 U.S. centers and compared stenting plus standard care to anticoagulation and compression alone. Six months...

Vermont Hospital Partners with OB-GYN Group to Sustain Birthing Services
Northwestern Medical Center in St. Albans, Vermont, has teamed up with independent OB‑GYN practice Maitri Health Care to preserve local obstetric and gynecologic services. Birth volumes at the hospital have dropped from over 500 to roughly 300 deliveries per year,...

FDA Notification Regarding Dehydrated Alcohol
The FDA has issued a formal notice telling companies to stop distributing any dehydrated alcohol products that are not the FDA‑approved Ablysinol from Belcher Pharmaceuticals. Ablysinol, approved on June 21, 2018, is the only legally marketed dehydrated alcohol in 1 mL and 5 mL...

Hospital Margins Decline in 2026 as Expenses Outpace Revenue
Hospital operating margins slipped into negative territory in early 2026, with the median margin at –0.3% year‑to‑date after a –0.6% reading in January. Expenses outpaced revenue growth, driven by a 7.6% rise in drug costs and a 7.8% increase in...

E&C Comprehensive Data Privacy Bill Includes HIPAA Carveout
The House Energy & Commerce Committee released a draft national data privacy bill that explicitly carves out HIPAA‑covered entities from its preemptive framework. The exemption aligns with recommendations health groups submitted to the committee last year. By preserving state‑level health...

Cleveland Clinic’s Plan to ‘Flip’ the Medicare Advantage Model
Cleveland Clinic is reversing the trend of hospitals exiting Medicare Advantage by moving to the payer side, taking delegated premium risk to manage total cost of care. The health system entered three value‑based risk agreements and targets $1.4 billion in Medicare...

NDIS Cuts Threaten Everyday Support
Australia's federal government announced plans to cut at least 160,000 participants from the National Disability Insurance Scheme (NDIS) by 2030, aiming to halve the scheme's annual growth rate from 10% to 5%. The NDIS, costing about $50 billion AUD (≈ $33 billion USD)...
DoD Seeks to Split Defense Health Program Into Two Accounts in Fiscal 2027
The Pentagon’s FY2027 budget proposes ending the single Defense Health Program and replacing it with two distinct accounts: Combat Operational and Medical Readiness (COMP) and a Private Sector Care Program (PSCP). The request totals $42.5 billion in discretionary spending—$20.3 billion for COMP...

At-Home Care Providers Prioritize Talent, Culture-First Acquisitions
Home‑based care providers are increasingly making cultural fit and talent alignment the cornerstone of acquisition strategies. Executives at Nova Leap, LifeCare Home Health and Arosa say that mis‑aligned culture can destroy value, while retaining strong leadership teams enhances post‑deal performance....

Raise a Glass: ‘Grandparents Happy Hour’ Law Passed In Minnesota
Minnesota Gov. Tim Walz signed the “Grandparents’ Happy Hour” law, removing the requirement that nursing homes and assisted‑living facilities hold a liquor license to serve alcohol. The change permits organized happy‑hour events for residents, with safeguards that staff serving drinks...
Hope Dims for Trump’s Surgeon General Nominee: 4 Details
President Donald Trump’s nominee for surgeon general, Dr. Casey Means, a wellness influencer and entrepreneur, faces an increasingly uncertain path to confirmation after a contentious Senate HELP Committee hearing. The hearing highlighted her lack of an active medical license, incomplete...

Dr. Oz Announces a 50-State Audit of Medicaid Program Oversight
Dr. Mehmet Oz, CMS administrator, announced a nationwide audit requiring all 50 states to revalidate Medicaid providers within 30 days, expanding the federal anti‑fraud push beyond the states previously targeted. The audit focuses on high‑risk areas and follows recent errors in...

How Women Executives Are Rewriting Healthcare Leadership — 4 Takeaways
At Becker’s Hospital Review’s 16th Annual Meeting, four senior women leaders in healthcare discussed how they are reshaping leadership norms. They emphasized that clarity is a structural skill that resolves the “decisive but accommodating” paradox, and that sponsorship—providing access and...

Highest-Paid Payer CEOs in 2025
UnitedHealth Group chief Stephen Hemsley topped the 2025 list of highest‑paid payer CEOs, earning a total of $60.94 million, largely driven by a one‑time $60 million equity award. Former UnitedHealth CEO Andrew Witty placed second with $24.6 million, while Cigna, Elevance Health, CVS...

CMS Delays Part D Portion of BALANCE Model on Expansion of GLP-1 Access
On April 21, the Centers for Medicare & Medicaid Services announced that the Medicare Part D component of the Better Approaches to Lifestyle and Nutrition for Comprehensive Health (BALANCE) Model will be delayed until 2027 pending additional evaluation and data collection....

3 Female CFOs on Moments that Defined Their Careers
At Becker’s 16th Annual Meeting, three senior healthcare CFOs—Shelly Schorer of CommonSpirit Health, Judy Peek‑Lee of the University of Vermont Medical Center, and Monica Price of Texas Health Resources—shared the pivotal moments that shaped their careers. They highlighted how female...

Cognitive Decline May Begin Up to 8 Years Before CVD Events in Older Adults
A large nested case‑control analysis of the ASPREE trial found that older adults who later suffered a cardiovascular event experienced accelerated declines in global cognition, memory, processing speed and verbal fluency. The cognitive deterioration began three to eight years before...