Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.
Biogen-Ionis Tau Antisense Drug Shows Cognitive Slow‑down in Early Alzheimer’s Phase 2
Biogen and Ionis announced that diranersen, an antisense therapy targeting tau protein, slowed cognitive decline and cut brain tau biomarkers in a 416‑patient Phase 2 study of early Alzheimer’s disease. The dual signal of biological impact and clinical benefit could reshape longevity‑focused biohacking strategies.

IL-15 Super‑Agonist ANKTIVA: Longevity’s Hidden Cancer Breakthrough
In 2024 the FDA approved a drug for bladder cancer. It is now the most interesting molecule in longevity medicine that no one is talking about. The drug is ANKTIVA - an IL-15 super-agonist. IL-15 is the body's signal for keeping...

Vanda’s Imsidolimab Wins Orphan‑Drug Designation in Japan for Rare Psoriasis
Vanda Pharmaceuticals announced that Japan’s Ministry of Health, Labour and Welfare has granted orphan‑drug designation to its IL‑36 inhibitor imsidolimab for generalized pustular psoriasis (GPP). The designation unlocks development subsidies and up to ten years of market exclusivity, a pivotal...

Open Source Therapeutics Divulges New PARP-1 Inhibitors
Open Source Therapeutics announced the discovery of two first‑in‑class PARP‑1 inhibitors aimed at treating DNA‑repair‑deficient cancers. The compounds demonstrated up to 80% tumor growth inhibition in mouse xenograft models and exhibit oral bioavailability with a half‑life suitable for once‑daily dosing....
Traws Pharma Announces New Antiviral Program
Traws Pharma announced a new antiviral development program aimed at emerging RNA viruses and other high‑risk pathogens. The initiative earmarks roughly $200 million over the next five years to fund pre‑clinical research, platform technology, and early‑stage clinical trials. The company is...

Use of Ultra-Rapid Insulin Plus MiniMed 780G Raises Time in Range
A single‑arm trial of 211 participants with type 1 diabetes showed that pairing Eli Lilly’s ultra‑rapid insulin lispro‑aabc (Lyumjev) with Medtronic’s MiniMed 780G hybrid closed‑loop system significantly boosted time‑in‑range. Children achieved a mean TIR of 68.6% (up from 51.2%) and adults reached...

Vertex’s Journavx Made History, but Left a Void in the Pain Space
Vertex Pharmaceuticals secured FDA approval for Journavx, the first novel non‑opioid pain drug in decades, amid the ongoing opioid crisis and Purdue Pharma’s recent multi‑billion‑dollar settlement. The medication targets the NaV1.8 sodium channel and quickly reached one million prescriptions, but...

Ironwood Pharmaceuticals Reports the US FDA Approval of Linzess to Treat Pediatric Patients (≥2yrs.) With Functional Constipation
U.S. FDA has approved an expanded label for Ironwood Pharmaceuticals’ Linzess (linaclotide) for children aged two years and older with functional constipation. The decision follows a 12‑week Phase III trial in 2‑ to 5‑year‑old patients, where the 72 µg dose significantly increased...

Apogee Therapeutics Reports Part B P-II (APEX) Data on Zumilokibart in Atopic Dermatitis (AD)
Apogee Therapeutics announced Phase II Part B (APEX) results for its IL‑4/13 inhibitor zumilokibart in moderate‑to‑severe atopic dermatitis. In a 346‑patient study, the mid‑ and high‑dose arms met the primary endpoint at week 16, with 65.9% and 61.6% of patients achieving EASI‑75 versus...

One Infusion. A Permanent Gene Edit. A Lifetime of LDL Lowering. The VERVE-102 NEJM Data, the Lilly Acquisition Thesis, and...
Verve Therapeutics reported Phase 1b Heart‑2 data for its gene‑editing LDL therapy VERVE‑102 in the New England Journal of Medicine. A single intravenous infusion achieved up to 88% PCSK9 knock‑down and a 62% reduction in LDL‑C that persisted for 18...

FDA Approves Ironwood's LINZESS for Constipation in Kids 2‑5
Ironwood Pharmaceuticals announced that the U.S. Food and Drug Administration has approved LINZESS for functional constipation in children aged 2 to 5 years. The decision adds a new pediatric indication to a drug already used by more than 5.5 million patients...

How ASGCT and OTXL Are Working to Revive Shelved Cell and Gene Therapies
The American Society of Gene & Cell Therapy (ASGCT) and Orphan Therapeutics Accelerator (OTXL) have launched CGTxchange, an AI‑driven matchmaking platform designed to revive cell and gene therapies that were shelved for ultra‑rare diseases. By aggregating confidential and public data...

Meta Biohub Unveils Protein‑Design AI Tackling Cancer
Mark Zuckerberg and Priscilla Chan’s Biohub just released new Evolutionary Scale Models, to map, predict, and design proteins, with system showing results against cancer and immune disease targets https://t.co/GcRGpatDrL #ArtificialIntelligence #Innovation #Technology #Tech #TechNews

Basilea Secures $13.3 Million BARDA Grant to Push Novel Urinary‑Tract Antibiotic
Basilea Pharmaceutica Ltd. announced that the U.S. Biomedical Advanced Research and Development Authority (BARDA) awarded the company a $13.3 million grant to advance its novel antibiotic ceftibuten‑ledaborbactam etzadroxil. The funding lifts BARDA’s total commitment to $25 million and opens the door to...

Wedbush Boosts Apogee Therapeutics to Outperform, Sets $135 Target Amid $1.3 B Blackstone Deal
Wedbush reaffirmed an Outperform rating on Apogee Therapeutics and lifted its twelve‑month price target to $135, up from $120. The upgrade comes as Apogee announced a financing package with Blackstone Life Sciences that could deliver up to $1.3 billion in non‑dilutive...

The Evolving Biopharma Regulatory Landscape: Q&A with Harpreet Singh, MD
Harpreet Singh, former FDA oncology division director and now chief medical officer at Precision for Medicine, explains how the FDA is reshaping biopharma regulation through a shift to single pivotal trials for high‑risk cancers, the expanding but opaque National Priority...

STAT+: Trump’s Drug-Pricing Deals Set to Be Tested by New Product Launches
President Trump’s most‑favored‑nation (MFN) agreements with 17 drugmakers require new U.S. launches to be priced at net levels comparable to other wealthy nations. The policy, whose terms remain secret, is slated for its first public test as three drugs—AstraZeneca’s hypertension...

Sanofi’s Venglustat Secures the US FDA Priority Review for Type 3 Gaucher Disease
Sanofi’s oral glucosylceramide synthase inhibitor venglustat has received FDA acceptance of its New Drug Application and a priority‑review designation for treating the neurological manifestations of type 3 Gaucher disease (GD3). The decision follows the phase‑III LEAP2MONO trial, which enrolled 43 patients...

AbbVie Reports the US FDA Approval for Decnupaz to Treat Blastic Plasmacytoid Dendritic Cell Neoplasm (BPDCN)
AbbVie’s Decnupaz (pivekimab sunirine‑pvzy) received U.S. FDA approval for treating adult patients with blastic plasmacytoid dendritic cell neoplasm (BPDCN), an ultra‑rare aggressive blood cancer. The decision was based on the global Phase I/II CADENZA trial that enrolled 84 CD123‑positive hematologic malignancy...
New Drug ‘Functionally Cures’ Many Hepatitis B Virus Infections
GSK’s antisense drug bepirovirsen (bepi) added to standard antivirals produced a functional cure in 19% of chronic hepatitis B patients in two phase 3 trials, rising to 26% among those with the lowest surface‑antigen levels. The cure, defined as undetectable HBV DNA...
Stanford Researchers Block Aging Enzyme to Regrow Knee Cartilage, Launch Oral Trials
Stanford University scientists led by Helen Blau and Nidhi Bhutani have demonstrated that an injection blocking the aging enzyme 15-PGDH can regenerate knee cartilage in aged mice and human tissue samples. The breakthrough, which also boosted muscle mass and endurance,...
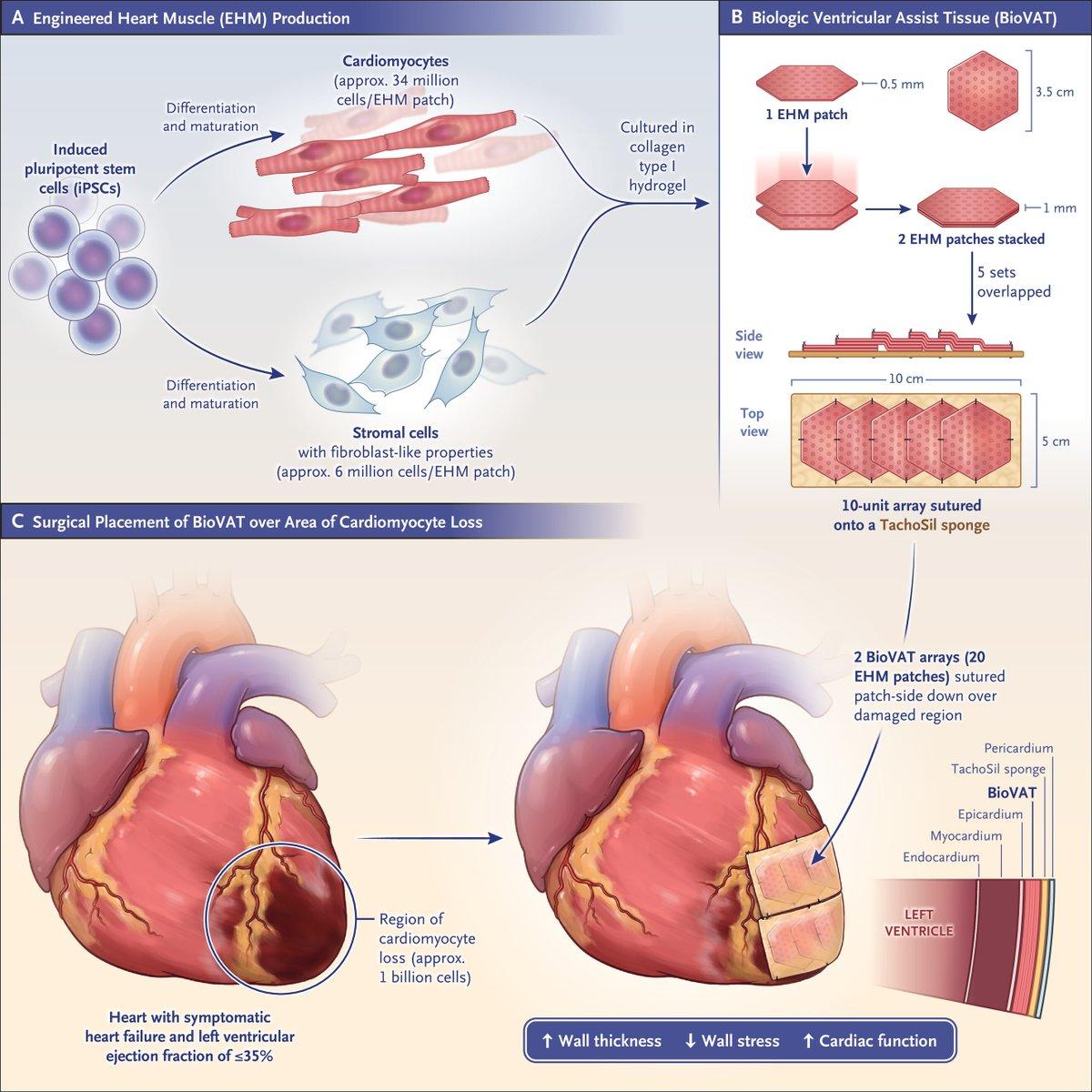
Stem‑cell Heart Patches Improve Outcomes in 12 of 20 Patients
Cardiac "remuscularization" for treating severe heart failure with patched heart muscle derived from stem cells (a biological ventricular assist device) successful in 12 of 20 patients @NEJM https://t.co/mAwgj2rAwE https://t.co/3ogxaJFDE0 https://t.co/J1ozMsS4Oh

Outlook Therapeutics Secures FDA Appeal Win for nAMD Therapy, Stock Jumps
Outlook Therapeutics Inc. (OTLK) announced that the FDA granted its appeal for the ONS-5010/LYTENAVA neovascular age‑related macular degeneration therapy, prompting a rise in its share price. The decision follows positive data from the pivotal NORSE TWO trial and confirmatory evidence...
BioRestorative Completes Phase 2 Dosing of BRTX-100, Shares Jump 20%
BioRestorative Therapies Inc. finished dosing its Phase 2 trial of BRTX‑100 for chronic lumbar disc disease, prompting a 20% surge in its stock. The autologous stem‑cell therapy, already granted FDA Fast Track status, was administered to 99 patients over a 52‑week...

Merck and Mastercard Are Seeing Real Agentic AI Results. Both Say the Plumbing Came First.
Merck is leveraging AI agents to accelerate drug discovery and marketing, cutting research cycles by a third and delivering compliant marketing drafts up to 80% faster. The gains stem from a "plumbing‑first" strategy that now supports 2,500 AWS accounts, multiple...

How Real-World Evidence Is Shaping Inclusive Clinical Trials with Takeda CMO Dr. Awny Farajallah — Episode 257
In the latest Xtalks Life Science Podcast, Takeda CMO Dr. Awny Farajallah explains how real‑world evidence (RWE) is reshaping clinical trial design to be more inclusive and diverse. He highlights the use of RWE in rare‑disease programs such as narcolepsy...
FDA Accepts BridgeBio’s Application for Potential First Limb-Girdle Muscular Dystrophy Drug
BridgeBio’s oral therapy BBP‑418 has received FDA priority review, with a target action date of Nov. 27, 2025, positioning it for a potential launch in late 2026 or early 2027. The Phase 3 FORTIFY trial met all primary and secondary endpoints, showing...

Congo Seeks US Monoclonal Antibody for Bundibugyo Ebola Strain
Democratic Republic of Congo officials have formally requested 500 doses of the experimental US‑developed monoclonal antibody MBP‑134 to treat patients infected with the Bundibugyo strain of Ebola. The antibody, which targets Zaire, Sudan and Bundibugyo variants, was previously used in...
Dyne Therapeutics Files BLA for Z‑Rostudirsen, Seeks Priority Review for DMD Treatment
Dyne Therapeutics announced the filing of a Biologics License Application with the FDA for Z‑rostudirsen, an exon 51‑skipping therapy for Duchenne muscular dystrophy, and has asked for priority review. The move follows a Phase 1/2 DELIVER trial that met its...
Hansa Biopharma’s Idefirix Hits 90% Graft Survival in EU Post‑Authorization Study
Hansa Biopharma announced that its gene‑therapy desensitization product Idefirix met the primary endpoint of a European post‑authorization study, delivering 90% one‑year graft failure‑free survival in highly sensitized kidney‑transplant patients. The results also showed 92% graft survival and 98% patient survival,...

Jade Biosciences Initiates First-in-Human Phase 1 Trial of BAFF-R Antibody JADE201
Jade Biosciences has dosed the first participant in a first‑in‑human Phase 1 trial of JADE201, an investigational half‑life‑extended afucosylated anti‑BAFF‑R monoclonal antibody. The randomized, placebo‑controlled study evaluates single ascending doses in rheumatoid arthritis patients, focusing on safety, tolerability, pharmacokinetics and pharmacodynamics....

Junevity to Present Breakthrough Research on siRNA Therapeutics at American Aging Association’s 2026 Annual Meeting
Junevity will present pioneering in‑vivo data showing an siRNA therapeutic can restore global gene networks to a healthier state. Co‑founder Dr. Janine Sengstack will share results from diabetic mouse studies and safety data in rats and non‑human primates, marking the...

STAT+: Kailera’s Own ‘Triple-G’ Drug Also Looks Very Powerful
The FDA postponed its decision on AstraZeneca’s experimental breast‑cancer therapy camizestrant after advisers criticized the SERENA‑6 trial design, giving the company extra time for additional analyses. Meanwhile, Blackstone Life Sciences pledged up to $1.3 billion to Apogee Therapeutics to fund Phase 3...

Chinese Scientists Use Supercomputer to Cut New Drug Screening Time From Years to Seconds
Chinese researchers have launched GalaxyVS, an AI‑driven drug‑discovery platform that leverages the Tianhe supercomputer to screen up to 100 billion chemical compounds in seconds. The system achieves a daily throughput of 16 trillion molecular dockings, a million‑fold speed increase over the previous...

Zuckerberg, Chan's Biohub Launches Protein 'World Model'
The Chan Zuckerberg Biohub has unveiled an AI‑driven "world model" for protein biology, built on an atlas of 6.8 billion proteins and 1.1 billion structures. The open‑source platform combines the ESMC language model with the ESMFold2 design engine to predict structures, design...

Lilly Just Bought Renowned Regulator Peter Marks a Pipeline to Play With
Eli Lilly has spent $3.8 billion acquiring Curevo, LimmaTech Biologics and Vaccine Company, giving the company a fledgling vaccine pipeline. The deals were driven in part by the hiring of former FDA CBER chief Peter Marks, who left the agency amid concerns over...

Psilocybin Shows Promise for Suicidal Ideation, Depression and Nerve Pain
A single 25‑mg dose of psilocybin cut chronic suicidal ideation in 70% of participants in a Sheppard Pratt trial, while separate studies reported three‑month depression relief in humans and month‑long nerve‑pain reduction in mice. Researchers say the findings could broaden...

FDA Extends Review of AstraZeneca’s Camizestrant NDA for ESR1‑Mutated Advanced Breast Cancer
The U.S. Food and Drug Administration has extended the PDUFA review date for AstraZeneca’s New Drug Application for camizestrant, an oral selective estrogen receptor degrader combined with a CDK4/6 inhibitor, after requesting more data to support its use in hormone‑receptor‑positive,...

Eli Lilly to Acquire Curevo for up to $1.5 Billion, Boosting Shingles Vaccine Portfolio
Eli Lilly announced a definitive agreement to acquire Curevo Vaccine for up to $1.5 billion in cash. The deal gives Lilly access to Amezosvatein, a Phase‑3‑ready shingles vaccine that matched the standard of care in Phase 2 while halving side‑effects. The transaction...

MediciNova Completes Phase 2 Trial of MN-001 for Diabetic NAFLD
MediciNova Inc. announced the last‑patient, last‑visit milestone for its Phase 2 MN-001‑NATG‑202 trial, evaluating tipelukast in hypertriglyceridemia and non‑alcoholic fatty liver disease (NAFLD) linked to type‑2 diabetes. The trial’s completion clears the path for data analysis and potential Phase 3...

China’s Drug Regulator Clears Wave of Home-Grown Innovative Medicines Amid Biotech Boom
China’s National Medical Products Administration has approved 19 innovative drugs this year, with domestic firms accounting for 15 of them, highlighting a rapid biotech surge. The regulator recently overhauled its approval pathway to speed market entry for home‑grown medicines. Leading...

The Most Expensive Drugs on the Market in 2026 — and Why They Cost So Much
In 2026 the United States market is dominated by ultra‑high‑priced one‑time gene and cell therapies, with Lenmeldy leading at $4.25 million per patient. Other top‑tier products include Hemgenix ($3.5 M), Elevidys ($3.2 M), Lyfgenia ($3.1 M) and Skysona ($3 M). Reuters notes the median list...

FDA Removes Boxed Warning About Risk of Leg and Foot Amputations for the Diabetes Medicine Canagliflozin (Invokana, Invokamet, Invokamet XR)
The FDA has eliminated the boxed warning on canagliflozin (Invokana, Invokamet, Invokamet XR) that highlighted a heightened risk of leg and foot amputations. The decision follows new data from three clinical trials showing the amputation risk is lower than originally...
Popular Anti-Aging Drug Combo Caused Severe Brain Damage in Mice
Researchers at the University of Connecticut discovered that the popular anti‑aging drug combo dasatinib plus quercetin (D+Q) causes severe myelin damage in mice, affecting both young and old subjects. The study, published in PNAS, showed dramatic loss of the protective...
Kura’s Encouraging Cancer Drug Data Could Unlock $2B Market Opportunity
Kura Oncology reported that its experimental farnesyltransferase inhibitor darlifarnib, combined with Bristol Myers Squibb’s KRAS inhibitor Krazati, shrank tumors in 77% of the 26‑patient FIT‑001 cohort. Objective response rates reached 67% in pancreatic cancer, 50% in non‑small cell lung cancer,...

D&D Pharmatech Reports P-II Trial Results on Zabopegdutide for Metabolic Dysfunction-Associated Steatohepatitis (MASH)
D&D Pharmatech disclosed topline Phase II data for its GLP‑1‑like agent zabopegdutide in 67 overweight or obese patients with metabolic dysfunction‑associated steatohepatitis (MASH). After a 2‑week titration to a 40 mg weekly maintenance dose, 48‑week histology showed a 50% fibrosis‑stage improvement versus...

Junshi Biosciences Achieves Phase 3 Success with Toripalimab Combo in NSCLC
Junshi Biosciences announced that its phase 3 NEOTORCH trial met primary endpoints, showing toripalimab plus chemotherapy reduced recurrence risk by 60% in resectable stage II‑III non‑small cell lung cancer. The data pave the way for a supplemental new drug application...
Precision BioSciences Reports First cccDNA Elimination in Hepatitis B Patients
Precision BioSciences announced that its ELIMINATE-B study demonstrated the first clinical evidence of complete cccDNA elimination in hepatitis B patients after two doses of PBGENE-HBV, achieving a 10‑fold reduction in viral transcripts. The result could reshape curative strategies for chronic...

Cartesian Therapeutics Secures $150 Million Credit Facility to Push Autoimmune Cell Therapy
Cartesian Therapeutics announced a $150 million credit facility with K2 HealthVentures, including an initial $50 million tranche that will fund operations through 2028. The financing backs the upcoming Phase 3 AURORA trial readout for Descartes‑08, its lead cell‑based therapy for myasthenia gravis and...

Jade Biosciences Doses First Patient in JADE‑101 Phase 2 Trial for IgA Nephropathy
Jade Biosciences announced the first dose of JADE‑101 in its JUNIPER Phase 2 trial for immunoglobulin A nephropathy, enrolling about 30 patients. The monoclonal antibody targets APRIL, a driver of pathogenic IgA, and interim results are slated for 2027.