Today's Pharma Pulse

FDA greenlights Merck’s tenofovir‑free HIV combo Idvynso
The FDA approved Idvynso, a once‑daily two‑drug regimen of doravirine and islatravir, marking the first tenofovir‑free, non‑INSTI HIV therapy. Phase III trials showed viral suppression comparable to Gilead’s three‑drug Biktarvy in patients without prior treatment failure or doravirine resistance.
Also developing:

Gene‑Therapy Pioneers Win $3 Million Breakthrough Prize for Vision and Sickle‑Cell Treatments
The Breakthrough Prize Foundation awarded a $3 million Life Sciences prize to three University of Pennsylvania scientists and two collaborators for pioneering gene‑therapy treatments that restore sight in Leber congenital amaurosis and target sickle‑cell disease. The honor spotlights Luxturna’s market entry and a parallel blood‑disorder therapy, underscoring the commercial and clinical momentum of gene‑editing platforms.

FDA Approves Breakthrough Therapy for Critically Ill Children
FDA says ‘yes’ to transformative therapy for sick children — and more media coverage of UCLA https://t.co/N6AJsRwCyX

Eli Lilly to Spend $6.3 B on Centessa Buy, with up to $1.5 B in Milestone Payouts
Eli Lilly agreed to acquire UK‑based Centessa Pharmaceuticals for an upfront $6.3 billion, plus a contingent value right that could add $1.5 billion if the lead sleep‑wake disorder drug meets regulatory milestones. The deal, priced at a 40% premium, follows Lilly’s cash‑rich...

NTU Unveils AI‑Powered Biochip That Detects Disease Biomarkers in 20 Minutes
Scientists at Singapore’s Nanyang Technological University have built an AI‑driven nanophotonic biochip that can identify disease‑related microRNA biomarkers in roughly 20 minutes. The prototype pairs a camera and mobile app for real‑time analysis, aiming to cut diagnostic turnaround times dramatically.
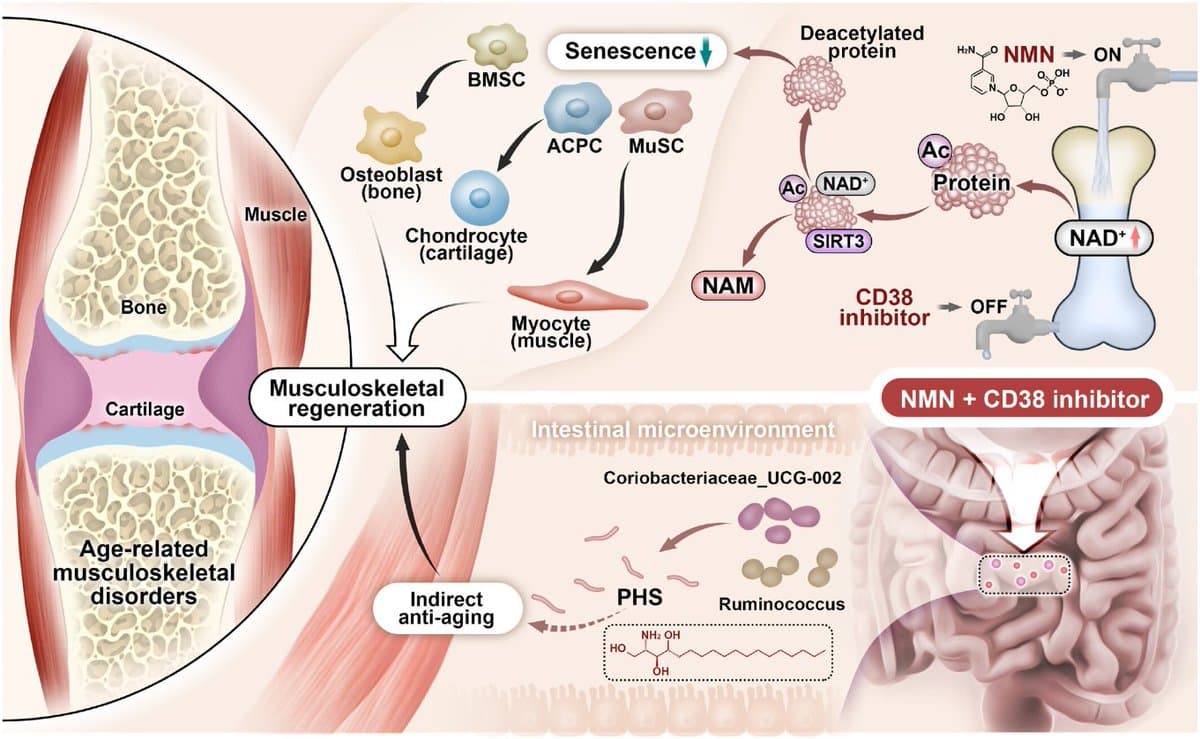
Combined NMN and CD38 Inhibitor Rejuvenates Aging Muscles
Double-Pronged NAD Preservation: Delaying Cellular Senescence and Initiating Musculoskeletal Regeneration https://t.co/bnkkn0xblj Scheme 1: A novel synergistic drug combination (N + A) consisting of an NAD+ precursor (NMN) and an NAD+ consumption (CD38) inhibitor (API) promotes musculoskeletal regeneration in aging.

FDA Grants Breakthrough Therapy Designation to Plixorafenib for Rare CNS Tumors
The U.S. FDA has granted Breakthrough Therapy Designation to plixorafenib, an investigational BRAF inhibitor for adults with rare, high‑grade gliomas harboring the BRAF V600E mutation. Early‑stage data showed a 67% overall response rate, positioning the drug as a potential game‑changer...

AI-Optimized Molecules Drive Clinical Interest and Expansion
#AACR2026 is in full swing and our posters are very popular. And some pharma companies realized that we have clinical data now and the molecules were AI-optimized to avoid the common liabilities. Finally, some genuine interest and we will have...

Here’s Simulations Plus Inc (SLP)’s Vision for AI in Drug Development
Simulations Plus Inc (NASDAQ:SLP) announced on March 26 that three major pharmaceutical firms have joined its AI‑enabled modeling collaboration programs, embedding the company’s AI agents directly into their drug‑development workflows. The firm emphasized a fully integrated AI deployment rather than...
Why Recursion Pharmaceuticals Inc (RXRX) Expanded Citeline Partnership
Recursion Pharmaceuticals announced an expanded partnership with Citeline on March 31, adding the data‑provider’s real‑world evidence capabilities to its AI‑driven drug discovery platform. The integration is designed to improve clinical‑trial design and accelerate decision‑making for its pipeline of cancer and...

Here’s What TD Cowen Thinks About Tempus AI Inc (TEM) Stock
TD Cowen upgraded Tempus AI Inc (NASDAQ:TEM) to a Buy rating, trimming its price target to $65 from $70. The company reported an 83.4% revenue jump to $1.3 billion in 2025 and projects $1.59 billion for 2026, a 25% increase. Strategic partnerships...

Leaked Images Show Monkeys Forced Into Weight‑Loss Drug Tests in UK Labs
A former laboratory technician leaked graphic footage of long‑tailed macaques being restrained and fed experimental anti‑obesity drugs via oral gavage in two UK facilities. The images, obtained by The Mail on Sunday, have ignited calls for faster phasing out of...

Cubical Bipyramid Nanoparticles Enable Systemic Magnetic Hyperthermia for Ovarian Cancer
Researchers at Oregon State University have created a cubical bipyramid magnetic nanoparticle that can be delivered intravenously and heated to 42‑46 °C under an alternating magnetic field, overcoming the need for direct tumor injection. The breakthrough, published in Advanced Functional Materials,...

FDA Expands Vertex's Alyftrek, Trikafta Labels to Reach 95% of U.S. Cystic Fibrosis Patients
The U.S. Food and Drug Administration approved label extensions for Vertex Pharmaceuticals' CF drugs Alyftrek and Trikafta, now covering roughly 95% of cystic fibrosis patients in the United States. The expansion adds eligibility for about 800 more patients, based on...

Novel Diabetic Wound Treatment Turns Cells Into Manufacturers
Researchers at Texas A&M have created a novel wound dressing for diabetic foot ulcers that leverages an interwoven extracellular matrix produced by human cells, then strips the cells away, leaving a purely biological scaffold. The approach sidesteps the immune‑rejection and...

Rogan Tipped the Scales on Psychedelic Research in the US
Joe Rogan appeared at the White House as President Trump signed an executive order to fast‑track psychedelic research and clinical trials. The directive directs federal agencies to accelerate approval pathways for psychedelic therapeutics aimed at PTSD, veteran mental health, and...

StockWatch: Revolution’s Phase III Pancreatic Cancer Data Dazzles Investors, Analysts
Revolution Medicines reported Phase III RASolute 302 results showing its oral RAS inhibitor daraxonrasib extended median overall survival to 13.2 months in previously treated metastatic pancreatic ductal adenocarcinoma, versus 6.7 months for standard chemotherapy (HR 0.40, p < 0.0001). The data sparked a...

What to Know Before Buying GLP-1 Drugs Online
The FDA warns that many online ads for GLP‑1 weight‑loss drugs promote compounded versions that lack the rigorous testing of brand‑name products like Ozempic, Wegovy, and Mounjaro. These compounded formulations can vary in concentration, contain unapproved ingredients, and have been...

Searches up 900%: Google Faces Influx of Illegal Peptide Ads
Google is facing a surge in advertisements for illegal synthetic peptides, with searches for terms like BPC‑157, GHK‑CU and Retatrutide jumping up to 900% over the past year. The ads violate Google’s own policies and Australian law, prompting the company...

Can a Common Parasite Medication Calm the Brain’s Stress Circuitry During Alcohol Withdrawal?
Researchers at UC San Diego discovered that rodents with high P2rx4 gene expression exhibit markedly increased alcohol consumption during withdrawal. Administering the antiparasitic drug ivermectin produced a dose‑dependent reduction in lever‑pressing for alcohol, especially in animals that responded behaviorally. Electrophysiological...

Lung‑resident Memory B Cells Drive Lasting Flu Protection
Long-term flu protection may depend on lung-resident memory B cells, whose persistence is shaped by the strength of B cell receptor signaling, offering new directions for vaccines targeting immune defenses within lung tissue. immunology

Nrf2 Activation Offers Therapeutic Promise, Yet Requires Caution
Nrf2 Activation in Inflammatory Diseases: A Review of Natural and Synthetic Modulators 🔑 Mechanistic insights and preclinical and clinical evidence on the role of Nrf2 in inflammatory diseases and evaluates the therapeutic potential of its key activators. “ The nuclear factor...

China Expands Insurance to Cover Over Half of Cancer Treatment Costs
China's national basic medical insurance now reimburses more than half of cancer treatment costs after adding 36 new antitumor drugs to its reimbursement list. The move lowers out‑of‑pocket spending for patients, including a cut of over 8,000 yuan per vial for...

Joe Rogan Reveals Trump IMMEDIATELY Offered Him FDA Approval for Unbelievable New Treatment...
President Donald Trump issued an executive order accelerating federal research on ibogaine, a Schedule I psychedelic, to create FDA pathways for veteran mental‑health treatment. The order follows claims that ibogaine can address depression, PTSD and substance abuse, with the FDA expected...

Dasatinib and Quercetin as Senolytic May Cause Brain Damage
A March 2026 PNAS study shows that the senolytic combo dasatinib and quercetin (D+Q) triggers demyelination in the corpus callosum of aged mice. The researchers used intermittent oral doses of 5 mg/kg dasatinib and 50 mg/kg quercetin, identical to regimens linked to...

USC Compound Halts Alzheimer-Linked Brain Inflammation
As a medical school professor at USC, this one is personal. My colleagues at Keck School of Medicine developed a compound that stops brain inflammation linked to Alzheimer's -- while preserving normal brain function. The target: cPLA2, an enzyme that drives inflammation...

Unexpected Insight Among 11 Lessons From AACR26
11 things I learned at #AACR26 Industry Partnering event – number 7 may well surprise you: https://t.co/T0ePbaIU1K https://t.co/CMh15HfnnG

MoonLake Immunotherapeutics CEO Sells $2.8 Million of Stock, Cutting Ownership to 4.9%
MoonLake Immunotherapeutics CEO Jorge Santos da Silva sold 150,000 shares for $2.8 million on April 10 and 13, 2026, trimming his direct ownership to 4.9%. The sale, the second‑largest since 2023, comes as the company navigates mixed clinical data and a...

KRAS Targeting Fuels $30B Valuation for RVMD
The race to catch KRAS, pancreatic cancer’s ‘greasy ball,’ and create the most promising drug in decades Or, how $RVMD is now worth $30B https://t.co/iFilUpezjn via @angRchen #AACR26

Order Set to Speed Psychedelic Drug Research Access
the order will dramatically accelerate access to new medical research and treatments based on psychedelic drugs

New Synthetic Opioid Shows Lower Addiction Risk Than Morphine in Rodent Study
Researchers at the National Institute on Drug Abuse reported that DFNZ, a novel nitazene‑derived opioid, produced weaker withdrawal symptoms and less compulsive self‑administration than morphine in rats, hinting at a safer pain‑relief alternative.

HHS Partners to Boost Psychedelic Trial Data, Prioritize Breakthroughs
HHS will work with the Department of Veterans Affairs, as well as the private sector, to increase clinical trial participation, data sharing, and real-world evidence generation regarding psychedelic drugs, and shall prioritize drugs that have received Breakthrough Therapy status

10x Genomics Unveils Atera Platform for Whole‑Transcriptome Spatial Biology
10x Genomics announced Atera, a new in situ spatial biology platform that delivers whole‑transcriptome analysis at single‑cell resolution and scale. Debuted at the AACR 2026 meeting, the technology aims to reshape disease research and clinical diagnostics by removing trade‑offs between...

20 Years Of Priority Review Vouchers, A Tool For Spurring Needed Drugs
The U.S. priority review voucher (PRV) program, created in 2007 to spur drug development for neglected tropical diseases (NTDs), has turned into a lucrative market incentive, with vouchers fetching nine‑figure sums. A landmark case is MDGH’s moxidectin, the first new...

Trump Signs Order to Fast‑track Ibogaine and Other Psychedelics
President Donald Trump signed an executive order to accelerate the FDA’s review of ibogaine and other psychedelic drugs, citing potential relief for veterans with PTSD and opioid addiction. The move includes priority vouchers that could cut review times from months...

A Closer Look at Body Composition in Obesity Drug Trials
A new systematic review of 36 randomized controlled trials, published in Annals of Internal Medicine, reveals that obesity medications often cause a sizable loss of lean mass. In 65% of drug trials, more than a quarter of total weight loss...

Major Antineoplastic Mechanisms of Combination Ivermectin-Mebendazole
In this episode of American Sunrise, chief scientific officer Dr. Peter McCullough discusses a new wellness‑company study showing that the antiparasitic drugs ivermectin and mebendazole, when used alongside standard cancer therapies, yielded an 84% positive net clinical benefit in over 200...

Weekly Reads: Prasad Successor, Sammy Hagar Stem Cells, More on OSK Trial, Finnish Speaker Needed
Vinay Prasad is set to leave his role as head of the FDA's Center for Biologics Evaluation and Research (CBER within two weeks, prompting speculation about the agency’s future direction. A new flexible oversight framework for rare‑disease gene and cell therapies...

STAT+: The Race to Catch KRAS, Pancreatic Cancer’s ‘Greasy Ball,’ and Create the Most Promising Drug in Decades
A new wave of KRAS‑targeted therapies is reshaping pancreatic cancer treatment after decades of failure. Revolution Medicines’ daraxonrasib, a next‑generation KRAS inhibitor, delivered dramatic survival benefits for patient Leanna Stokes, who enrolled in a clinical trial. The drug’s success has...

The Next Chemical Cage Has a Beautiful Door
President Biden signed an executive order fast‑tracking psychedelic drugs, granting the FDA priority vouchers for serotonin‑2A agonists. The move follows a public ceremony with Joe Rogan, RFK Jr., and FDA Commissioner Marty Makary, signaling a shift from traditional plant‑based use to pharmaceutical...

Chinese Biotech's Global Surge May Hinge on AI
China’s drug makers are speeding up – will AI be their secret weapon? Chinese biotech firms are striking big global deals as drug makers, but could artificial intelligence take them to the next level? No "overcapacity" here.... https://t.co/sktFORaVey via @scmpnews

TG Therapeutics: Flawless Execution And $500 Million Non-Dilutive Capital Bolster Outlook
TG Therapeutics was downgraded from Strong Buy to Buy after a 17% rally that brought its valuation closer to fundamentals. The company’s flagship MS drug, Briumvi IV, continues to capture market share, while a fully enrolled sub‑cutaneous trial could double...

Forbion Crowned Europe’s Largest Independent VC, Targets Asian Expansion
Forbion has been named Europe’s largest independent venture capital firm and, in the same announcement, unveiled a plan to seek new life‑science investments across Asia. Managing Partner Sander Slootweg said the move aims to offset funding constraints in Europe and...

HSS Studies Contribute Early Evidence to Help Guide Emerging Perioperative Considerations for Patients Using GLP‑1 Medications
At the ASRA annual meeting, Hospital for Special Surgery researchers presented two studies on peri‑operative management of patients taking GLP‑1 agonists. A multicenter ultrasound assessment of 354 elective surgery patients found no statistically significant difference in full‑stomach incidence between GLP‑1...

Teva's $40 Target: Olanzapine, Specialty Drugs and Biosimilars Fuel Upside
Teva Pharmaceutical Industries' stock has surged 130% in the past year, and analysts see three catalysts—FDA approval of a long‑acting olanzapine, accelerating specialty‑drug sales, and a wave of biosimilar launches—that could push the share price past $40. The outlook rests...

Quality Metrics for Drug Manufacturing
The FDA is advancing a Quality Metrics Reporting Program to collect objective manufacturing data from drug producers. By analyzing these metrics, the agency aims to enhance risk‑based surveillance, predict shortages, and streamline inspections. The initiative builds on a decade of...

Definium Therapeutics Applauds White House Executive Order to Accelerate Mental Health Innovation and Expand Access to Psychedelic Medical Treatments
Definium Therapeutics welcomed the White House’s new executive order that aims to speed research, regulatory review, and access to innovative mental‑health treatments, including psychedelics. The order directs federal agencies to streamline pathways and boost cross‑agency collaboration. Definium highlighted its DT120...

Three Gene Therapy Pioneers Just Won the Breakthrough Prize. This Is Their Story
Three pioneering scientists—Jean Bennett, Albert Maguire, and David J. Wilson—have been honored with the 2026 Breakthrough Prize in Life Sciences for their work on Luxturna, the first gene‑therapy approved by the U.S. Food and Drug Administration. Luxturna treats a rare...

Compass Pathways Commends White House Executive Order to Accelerate Research and Access for Psychedelic Treatments
Compass Pathways welcomed the White House Executive Order aimed at speeding up research and access to psychedelic therapies for serious mental illness. The biotech highlighted its COMP360 synthetic psilocybin, which has delivered statistically significant results in two Phase 3 trials for...
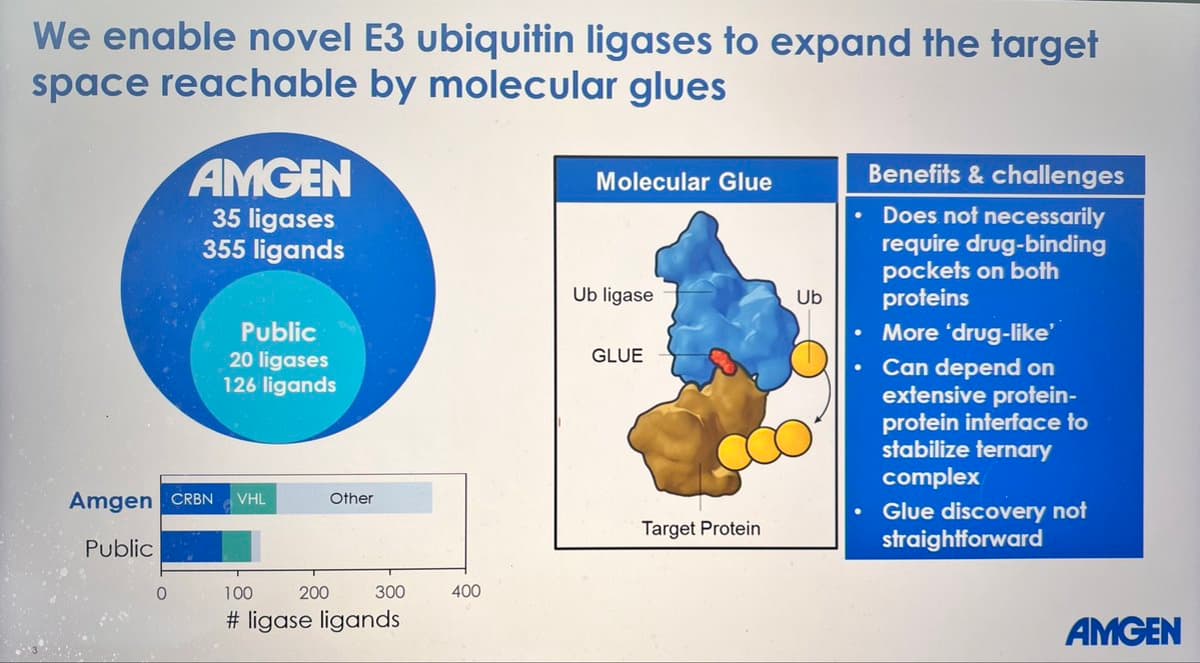
Amgen's Potts Lab Unveils Universal Molecular Glue Platform
Next up #aacr26 is @pottslab from Amgen discussing their broad and diverse molecular glue & degrader platform “any target, every time” https://t.co/SfJ0jGYene

Funding Global Cancer Research to Cure Every Patient
EVERY CANCER. EVERY PATIENT. EVERYWHERE. @ConquerCancerFd of the American Society of Clinical Oncology (@ASCO) raises funds to support the world's leading researchers who are improving treatments & discovering cures for patients around the world. https://t.co/du6r6gdsgV #ASCO26