Today's Pharma Pulse

CMS postpones GLP‑1 Medicare pilot, hitting Lilly and Novo shares
The Centers for Medicare & Medicaid Services indefinitely delayed its BALANCE financing model aimed at expanding Medicare coverage for GLP‑1 obesity drugs. The postponement knocked Eli Lilly’s stock down about 2% and Novo Nordisk’s by roughly 4%, with analysts projecting a near‑term revenue loss of up to $500 million for Lilly that could rise to $3.3 billion.
Also developing:
By the numbers: Asahi Kasei acquires German biotech Aicuris for $920.7M

Viatris Recalls Extended-Release Xanax over Dissolution Test Failure
Viatris has issued a Class II recall of a single batch of its extended‑release Xanax (Xanax XR) after dissolution testing showed the tablets did not meet release specifications. The affected bottles, each containing 60 3‑mg tablets, were manufactured in Ireland and are being withdrawn from the U.S. market. This recall is one of 30 drug recalls listed in the FDA’s latest weekly enforcement report and follows a 2022 recall of 110 bottles for the same product. Viatris reported Xanax XR sales of $145 million in 2024, down from $154.8 million in 2023.

CRISPR Advances Outpace Ethics in Germline Editing Debate
In 2018, He Jiankui edited the genomes of two human embryos, brought them to term, and went to prison for it. The babies are now children. We don't know how they're doing. Seven years later, base editing and prime editing can...

Substack Live | Flagship Pioneering Announcement: Building on the Code of Life
Flagship Pioneering is hosting a Substack Live session on April 21 at 12:30 pm ET featuring origination partner Jake Rubens. The event will unveil a new initiative that leverages the "code of life" to advance synthetic‑biology‑based therapeutics. Rubens, an MIT‑trained synthetic biologist...

Elisrasib Shows Strong Response Rates in Advanced KRAS‑G12C Lung Cancer
Elisrasib, a next‑generation KRAS‑G12C inhibitor, produced clinical benefit in a 165‑patient cohort of advanced non‑small cell lung cancer at the AACR 2026 meeting. The oral drug, given at a 600 mg daily dose, showed durable responses in patients previously treated with...

FDA Accepts Ultragenyx BLA for UX111 Gene Therapy Targeting Sanfilippo A
Ultragenyx announced that the U.S. Food and Drug Administration has accepted its resubmitted Biologics License Application for UX111, an AAV9 gene therapy for Sanfilippo syndrome Type A. The agency set a PDUFA action date of September 19, positioning UX111 as...

STAT+: Eli Lilly’s $3.25 Billion Acquisition of Kelonia Therapeutics Caps Startup’s Tortuous Ride
Eli Lilly announced a $3.25 billion acquisition of Kelonia Therapeutics, a small biotech focused on cell therapies for cancer and autoimmune diseases. The deal may include additional payments if Kelonia meets clinical, regulatory, and commercial milestones. Kelonia, formerly Elcano Therapeutics, survived on...

Merck Partner Reveals Bispecific Data; Biogen Pays $100M for Antibody Rights
Merck’s partner Sino Biopharm disclosed first‑in‑human results for its PD‑1×VEGF bispecific antibody, MK‑2010, reporting that six of 11 lung‑cancer patients achieved a response at a low dose. A second cohort showed four responses among nine patients, indicating activity across dosing...

2018 Study Predicts Psilocybin Should Move to Schedule 4
The psychedelic executive order requires government to "initiate and complete review of any product containing a Schedule I substance that has successfully completed Phase 3 clinical trials ... so that rescheduling ... may proceed as quickly as practicable". My 2018...

Israel's SynBio Surge: CEOs Reveal Scaling Success
Israel has built one of the most concentrated synbio ecosystems outside the US. The country's National Bioconvergence Program, run through the Israel Innovation Authority, is specifically designed to close the gap between lab-scale innovation and commercial biomanufacturing. This session at @SynBioBeta 2026...

Kelonia Investor Venrock Will Clear a 45-Fold Return on Startup's Sale to Lilly
Venture capital firm Venrock is set to pocket roughly $900 million after its modest $20 million seed investment in Kelonia Therapeutics paid off. Kelonia agreed to an all‑cash acquisition by Eli Lilly valued at $3.25 billion, delivering an upfront payment that triggers the windfall....

Biotech Must Prioritize Collaboration, Not Nationalism, for Patients
After a week in Washington DC at one of the best new annual events by @semafor , I came to China for just 2 nights to meet some of my friends from Washington. I even had to skip this year's...
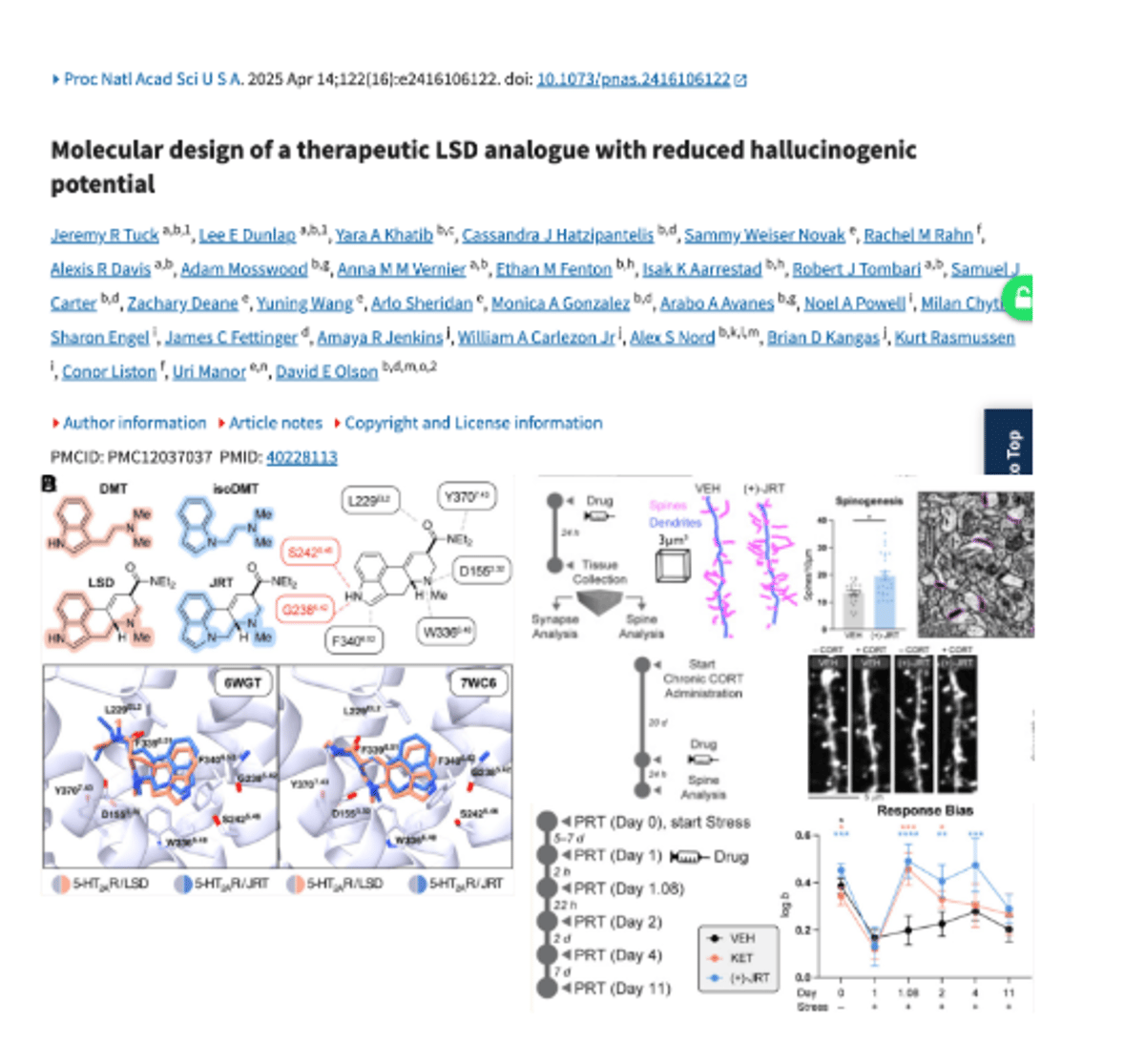
JRT: LSD-Derived Antidepressant Boosts Brain Spines without Hallucinations
One dose: > 46% more dendritic spines in prefrontal cortex > 100x more potent than ketamine as an antidepressant What is it? LSD with a two atom swap. > no trip > no hallucinations > no schizophrenia signature It's called JRT. https://t.co/sozMqlJctc

AACR 2026: Revolution’s Next Prospect, Merck’s Reveal and a Lung Cancer Battle
At AACR 2026, Revolution Medicines reported that its RAS‑G12D inhibitor zoldonrasib produced a 52% response rate and a median 11.1‑month progression‑free survival in heavily pre‑treated non‑small cell lung cancer (NSCLC) patients, hinting at accelerated‑approval potential. Merck presented early data on...

Balaji Predicts Booming Market for Bro Biotech
.@theojaffee asks Balajis for his thoughts on Clavicular, Balajis says he’s cheering for Clavicular because he’s tapping into bro biotech/bro science and that’s set to be a major market. Peptides, etc https://t.co/CgSt6rg1zI

LLY's Second in Vivo CAR‑T Deal via Kelonia Buyout
Kelonia turns #ASH25 late-breaker into buyout, with $LLY's second move into in vivo Car-T. Via @ApexOnco -> https://t.co/NPhdnuhPu3 $AZN $GILD

Gilead Declines Another Arcus Option After Phase 3 TIGIT Fail
Gilead Sciences has declined another development option with Arcus Biosciences following the Phase 3 failure of their joint TIGIT immunotherapy. The setback ends a key pillar of a long‑standing collaboration and signals Gilead’s intent to trim external programs. Both companies will...

Key Overlooked Factors in Clinical Trial Design
New presentation on things we often forget to consider when designing a clinical trial: https://t.co/BVlSCIHdLu #Statistics #clinicaltrials

Inside the $3.25B Eli Lilly Acquisition: Memo and Deck
Read the memo and slide deck that led to th creation of the company Eli Lilly bought for $3.25 billion today https://t.co/cNoTs7M4YM

Lilly Adds Gene Delivery Technology to CAR T in up to $7B Kelonia Deal
Eli Lilly announced an up‑to $7 billion acquisition of Kelonia Therapeutics, securing its Phase 1 lentiviral in‑vivo CAR‑T candidate KLN‑1010 and the iGPS gene‑delivery platform. iGPS uses lentiviral particles to program a patient’s own T‑cells, potentially eliminating ex‑vivo manufacturing and pre‑treatment chemotherapy. The deal...

Nektar Jumps as Alopecia Drug Shows Promise
Nektar surges again as alopecia drug shows new promise in extension study https://t.co/UV0ccY4HiR @ByJonGardner $NKTR + 25% $LLY

All Major Psychedelics Produce Identical Brain Network Effects
@Nature published the most comprehensive brain scan study on psychedelics ever done. Psilocybin, LSD, ayahuasca. Different compounds. Same effect on your brain networks. Every single time.

Agenus Cancer Cocktail Records 0% Response Rate, Missing Midstage Goal
Agenus and its spin‑out MiNK Therapeutics reported a zero percent overall response rate in a Phase 2 trial of a three‑drug cocktail for advanced gastroesophageal adenocarcinoma. The regimen combined two experimental immunotherapies, botensilimab and balstilimab, with MiNK’s allogeneic iNKT cell therapy...

AACR 2026 Unveils Merck Breakthrough and Lung Cancer Hope
AACR 2026: Revolution’s next prospect, Merck’s reveal and a lung cancer battle https://t.co/u2iwCd3DKv #AACR26 $RVMD $MRK $NUVL $NUVB
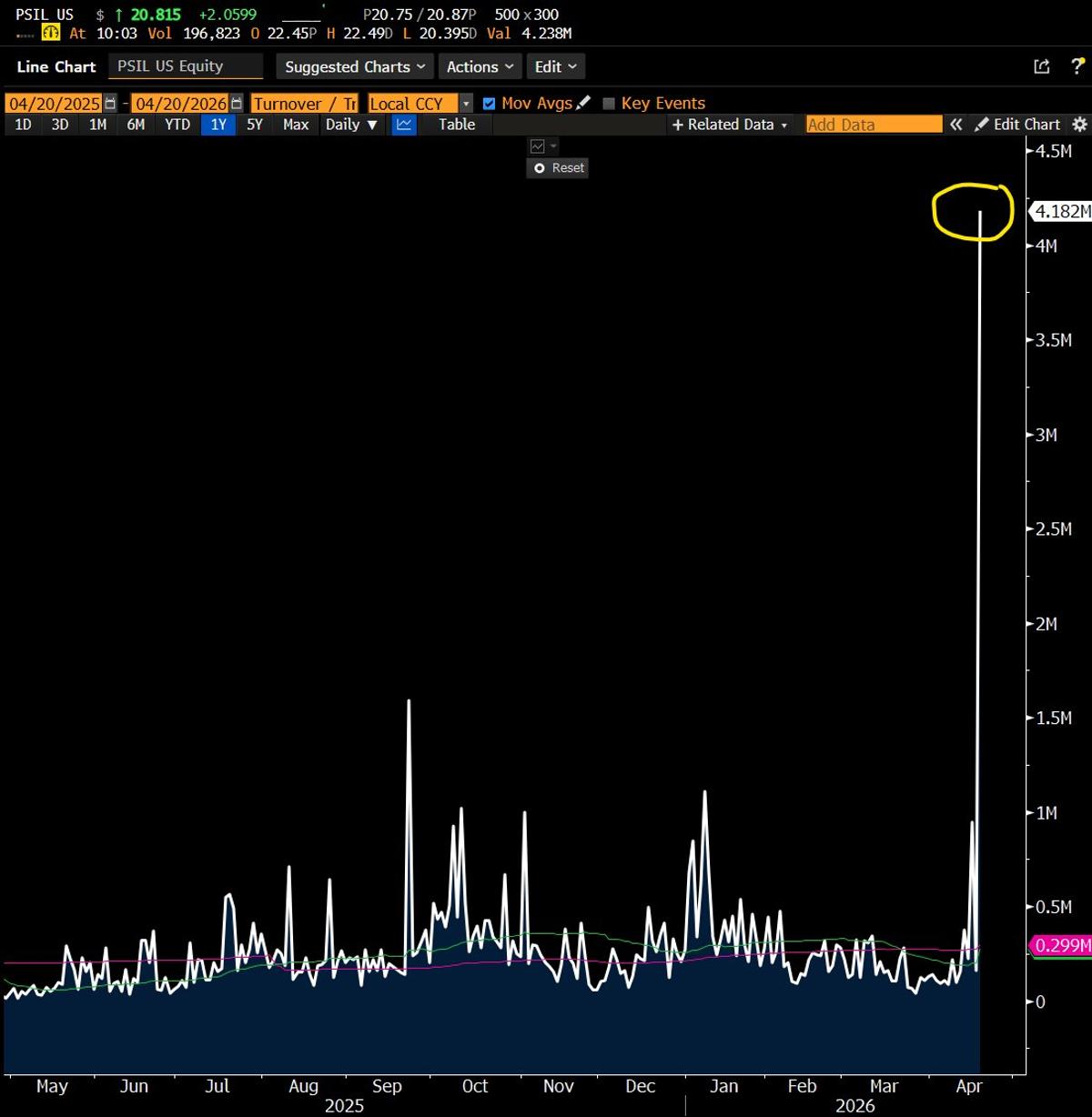
PSIL Surges as Trump Fast‑tracks Psychedelic Research
Far Out Man: The Psychedelics ETF $PSIL has destroyed its daily volume record in first 30min of trading.. Trump just signed ex order expediting research/access to psyche drugs to cure depression. The sector has been in dog house, waiting for...

Novo’s Late-Stage Sickle Cell Win Piles Pressure on Competitors
Novo Nordisk reported that its oral pyruvate kinase‑R activator etavopivat reduced vaso‑occlusive crises by 27% in the Phase 3 HIBISCUS trial and more than doubled hemoglobin response rates versus placebo. The data support a regulatory filing slated for the second half...

Molecular Glue Daraxonrasib Shows Promise Against Pancreatic Cancer
Good summary of the marked benefit of the molecular glue drug (daraxonrasib) vs pancreatic cancer, from Revolution Medicines, and other progress (adds to the neoantigen vaccine with 6-year survival) gift link https://t.co/qk7Ar9dCAQ https://t.co/SMiA51fiwX

Psychedelics Go Mainstream
President Donald Trump issued an executive order to speed up research and access to psychedelic therapies, allocating $50 million in federal funding and instructing regulators to dismantle long‑standing barriers. The move validates a growing investment thesis that the psychedelic sector will...

Beyond GLP-1Rs: Emerging Targets Poised to Gain Share of Obesity Market
The global obesity drug market is projected to reach $172.6 bn by 2031, up 139% from 2026, driven by GLP‑1R giants like Wegovy and Zepbound. Non‑GLP‑1R therapeutics, currently a niche at $310 m, are forecast to surge 50‑fold, hitting $15.5 bn by 2031....
STAT+: Finally Cracking KRAS as a Druggable Target
Revolution Medicines announced that its KRAS inhibitor daraxonrasib produced tumor shrinkage in a Phase 2 trial for pancreatic cancer, marking the first clear efficacy signal for an oral drug targeting the once‑undruggable KRAS protein. The result follows a wave of...

LLY Acquires Kelonia for $3.25bn After ASH25 Insights
It's official: $LLY buying Kelonia for $3.25bn + milestones. @ByMadeleineA take on the #ASH25 dataset that triggered this move: https://t.co/nVUNlq7YlO

Four‑Week Orange Trial Shows Lipid Shifts in Fatty‑Liver Patients
Researchers at Italy’s National Institute of Gastroenterology completed a randomized 4‑week trial in which 60 patients with metabolic dysfunction‑associated steatotic liver disease ate 400 g of Navelina oranges daily. The study documented measurable changes in serum lipid species without affecting weight...

Regeneron Expands Dupixent to Pediatric Immunology and Teams with Telix on Radiopharma
Regeneron announced a label expansion for Dupixent in Europe to cover pediatric chronic inflammatory diseases and disclosed a strategic partnership with Telix Pharmaceuticals to develop radiopharmaceutical oncology agents. The moves broaden the company's biologic footprint and add a new oncology...

STAT+: Pharmalittle: We’re Reading About Trump Boosting Psychedelic Treatment, the Future for Weight-Loss Drugs, and More
Eli Lilly CEO David Ricks warned that GLP‑1 weight‑loss drugs will likely reach only about half of the eligible overweight and obese population due to healthcare system complexities and cost barriers. Currently, just 10% of those who could benefit are...

Oregon Passes Bill to Support Medical Cannabis Among Hospice Patients
Oregon enacted Ryan’s Law, mandating hospice, residential and palliative care providers to permit qualified patients to use medical cannabis and to establish safety policies and staff training. The bill adds hospice and end‑of‑life care as a new qualifying condition for...

CStone Unveils Preclinical Data on Three Novel ADCs at AACR 2026
CStone Pharmaceuticals disclosed preclinical data for three proprietary antibody‑drug conjugates—CS5007, CS5006 and CS5008—at the American Association for Cancer Research annual meeting. The bispecific EGFR/HER3 ADC, CS5007, demonstrated plasma stability better than the DS‑8201 benchmark, with free payload release under 0.5%...

A Vaccine for Lyme Disease Could Be on the Horizon
Pfizer and Valneva reported that their Lyme disease vaccine candidate LB6V reduced cases by about 70% in a Phase 3 trial. The four‑dose regimen targets the OspA protein, preventing bacterial transmission from ticks to humans. If regulators approve it, the...

Decoding Resistance to Targeted Therapy via New Cancer Models
ATCC and the Broad Institute have created a panel of isogenic non‑small cell lung cancer (NSCLC) cell lines that model resistance to osimertinib, the newest EGFR inhibitor. Using CRISPR editing and gene‑overexpression, the team introduced six clinically observed resistance mechanisms,...

Pheast Therapeutics Reports Early P-Ia Data for PHST001 at AACR 2026
Pheast Therapeutics presented initial Phase Ia data for its anti‑CD24 macrophage checkpoint inhibitor PHST001 at the AACR 2026 meeting. The study showed clear target engagement, activation of innate immunity and a favorable safety profile across dose‑escalation cohorts. Early clinical signals...

Boehringer Ingelheim Launches AI Center for Pharma Research in London
Boehringer Ingelheim announced the launch of a new artificial‑intelligence and machine‑learning centre in London, its fourth dedicated AI hub after sites in Austria, Germany and the United States. The company will invest £150 million (about $200 million) over the next ten years...
![[Podcast] From Protocol to Patient: Inside Clinical Supply with Almac Clinical Services](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/render/image/public/images/thumbnails/e66ba866d739cb77f8cd3922f2eb37b8.webp?width=1200&resize=contain&quality=75)
[Podcast] From Protocol to Patient: Inside Clinical Supply with Almac Clinical Services
Almac Clinical Services has launched a six‑part podcast series that walks listeners through the end‑to‑end clinical supply chain, from protocol translation to global regulatory release. The series targets biopharma sponsors, highlighting how early collaboration with supply partners can streamline planning,...

Who Is Dr. Houman Hemmati, Potentially CBER’s Next Leader?
Dr. Houman Hemmati, a board‑certified ophthalmologist and biotech entrepreneur, is the leading candidate to replace Dr. Vinay Prasad as director of the FDA’s Center for Biologics Evaluation and Research (CBER). Hemmati co‑founded Optigo Biotherapeutics, held senior roles at companies such as...

Update: Lilly Makes $7bn Bid for in Vivo CAR-T Firm Kelonia
Eli Lilly has confirmed a $3.25 billion upfront offer for Kelonia Therapeutics, with milestone payments that could lift the total value to $7 billion. Kelonia’s in vivo CAR‑T candidate KLN‑1010 demonstrated MRD‑negative responses in a four‑patient early‑stage study, highlighting the promise of off‑the‑shelf cell...

Merck’s Early PD-1/VEGF Data Competitive in Lung Cancer, but Summit ‘Looms Large’
Merck’s anti‑PD‑1/VEGF bispecific MK‑2010 posted a 55% overall response rate in treatment‑naïve non‑small cell lung cancer (NSCLC) and a 44% response in later‑line patients, figures that rival the current leader, Summit/Akeso’s ivonescimab. The data were presented at the AACR meeting,...

Nektar’s Long-Term Mid-Stage Alopecia Data Fuel Phase 3 Plans
Nektar Therapeutics reported that its IL-2 variant rezpegaldesleukin achieved modest efficacy and safety signals in the long‑term extension of a previously failed Phase 2b alopecia areata trial, barely meeting Wall Street expectations. The data revive confidence in the program and...

STAT+: Extended Use of Nektar Therapeutics Drug Shows Promise in Alopecia
Nektar Therapeutics announced that its experimental oral drug rezpeg produced significant hair regrowth in patients with severe alopecia areata. After a year of treatment, 27% of participants reached a SALT Score 20, meaning at least 80% of the scalp was covered...

Novo Nordisk Reports Positive P-III (HIBISCUS) Study Data for Etavopivat in Sickle Cell Disease
Novo Nordisk announced that its oral pyruvate kinase activator etavopivat met both primary endpoints in the Phase III HIBISCUS trial for sickle cell disease. In 385 patients aged 12 and older, the drug reduced vaso‑occlusive crisis (VOC) rates by 27% and...

AstraZeneca Reports Positive P-III (MIRANDA) Data for Tozorakimab in COPD
AstraZeneca announced that its anti‑IL‑33 antibody tozorakimab achieved its primary endpoint in the Phase III MIRANDA trial, delivering a statistically significant reduction in the annualised rate of moderate‑to‑severe COPD exacerbations. The double‑blind study enrolled 1,454 patients receiving standard inhaled therapy and...
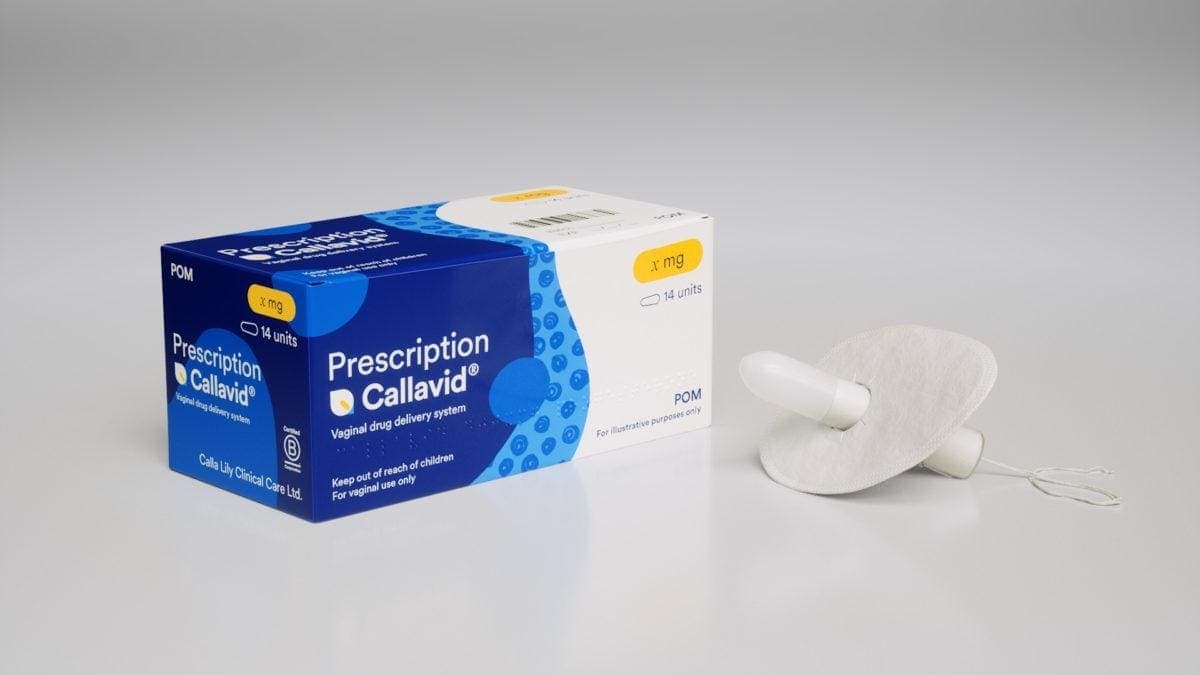
Vaginal Drug Delivery Had A Funding Problem—Merck Changed That
Calla Lily Clinical Care’s tampon‑shaped vaginal drug delivery platform, Callavid, has secured a strategic collaboration with Merck to accelerate clinical development for IVF luteal‑phase support and miscarriage prevention. The device, cleared under FDA 510(k) and patented in 14 countries, promises...

AZ Is Three for Three with COPD Hope Tozorakimab
AstraZeneca’s anti‑IL‑33 antibody tozorakimab has succeeded in all three pivotal Phase 3 COPD trials, showing a statistically significant reduction in moderate‑to‑severe exacerbations. The MIRANDA study confirmed benefit with biweekly dosing, while OBERON and TITANIA validated once‑monthly regimens. These results place AZ...

Novo Nordisk’s Sickle Cell Therapy Hits in Phase 3, but Data Lag Expectations
Novo Nordisk announced that its oral sickle‑cell drug etavopivat met its primary endpoint in a Phase 3 trial, showing a statistically significant reduction in painful vaso‑occlusive crises. The study, however, fell short of the ambitious efficacy and safety benchmarks the...