Today's Pharma Pulse

CMS postpones GLP‑1 Medicare pilot, hitting Lilly and Novo shares
The Centers for Medicare & Medicaid Services indefinitely delayed its BALANCE financing model aimed at expanding Medicare coverage for GLP‑1 obesity drugs. The postponement knocked Eli Lilly’s stock down about 2% and Novo Nordisk’s by roughly 4%, with analysts projecting a near‑term revenue loss of up to $500 million for Lilly that could rise to $3.3 billion.
Also developing:
By the numbers: Asahi Kasei acquires German biotech Aicuris for $920.7M

Omada Health Study Shows 6% Weight Loss and Muscle Preservation on GLP-1 Program
Omada Health released a 12‑week study of 245 adults with obesity showing its GLP‑1 Care Track delivered 6.0% average weight loss, a 3.3‑point drop in body‑fat percentage and a threefold rise in muscle‑mass share compared with a control group. The findings highlight how digital coaching can sharpen the metabolic benefits of GLP‑1 drugs.

Haryana Drugs Regulator Reports Fake Mounjaro Seizure in Gurugram: Eli Lilly
Eli Lilly disclosed that Haryana’s drug regulator seized roughly ₹70 lakh (about $85,000) of counterfeit Mounjaro injections in Gurugram. The company said it is actively supporting the investigation and praised the authorities’ enforcement action. Mounjaro, a GLP‑1 drug used for obesity, has...

UCB to Acquire Neurona Therapeutics for $650M, Adding Regenerative Epilepsy Asset
UCB announced a definitive agreement to acquire Neurona Therapeutics for $650 million upfront and up to $500 million in milestones, adding the regenerative cell therapy NRTX‑1001 to its epilepsy pipeline. The deal, expected to close by Q2 2026, expands UCB’s portfolio into advanced‑therapy...

Innate Pharma to Present P-II (MATISSE) Interim Data of IPH5201 in NSCLC at AACR 2026
Innate Pharma will present interim Phase‑II (MATISSE) data for its anti‑CD39 antibody IPH5201 in resectable non‑small cell lung cancer (NSCLC) at the AACR 2026 meeting. The analysis of 40 patients shows a pathological complete response (pCR) of 35.7% in tumors...
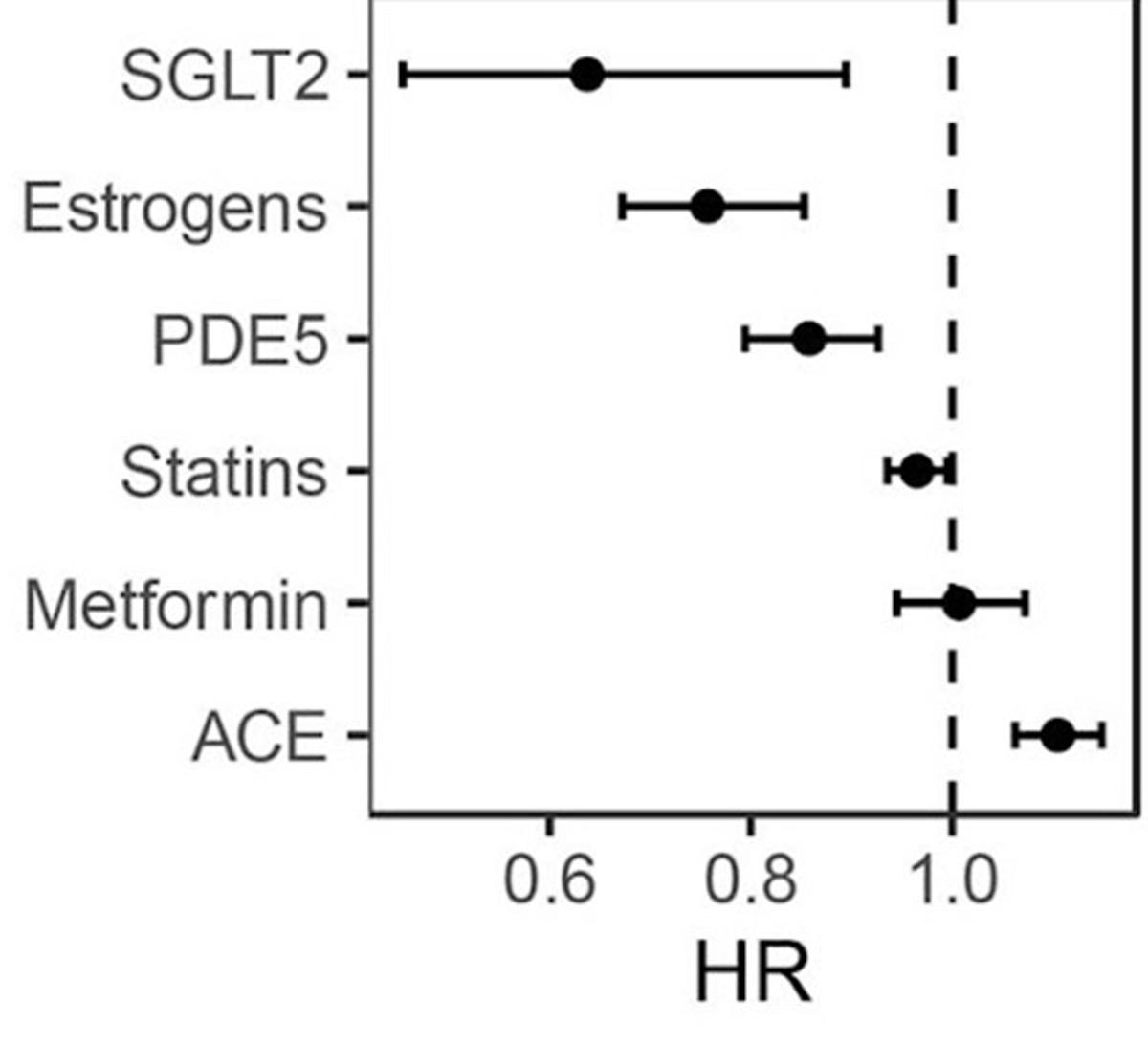
Certain Drugs Like SGLT2 Inhibitors Cut Mortality Risk
A 2024 study on half a million UK individuals analyzed the mortality effects of 406 medications 92% were seen to be associated with increased mortality risk because of the underlying diseases. However, 14 were surprisingly associated with reduced mortality. An overview by...

Mitrix Bio as an Example of the Trend Towards Alternative Paths to Initial Human Data
Mitrix Bio reported preliminary Phase 1 safety results for large‑dose mitochondrial infusions, showing no immediate adverse effects in two older participants. The company simultaneously opened Right‑to‑Try clinics in Dallas, Newport Beach and Palm Beach, offering the experimental therapy under a patient‑driven model. Its...

Eli Lilly in Advanced Talks to Buy Kelonia Therapeutics for over $2 Billion
Eli Lilly is in advanced talks to acquire Kelonia Therapeutics for over $2 billion, according to the Wall Street Journal. The deal, which may include milestone payments, could be announced as early as Monday and would bolster Lilly’s oncology pipeline. Shares...

Nanobiotix's Nanoprimer Boosts LNP‑DNA Immunotherapy Bioavailability, Cuts Toxicity
Nanobiotix unveiled preclinical results that its Nanoprimer platform, given before lipid‑nanoparticle DNA immunotherapies, markedly increases systemic exposure and reduces hepatic toxicity in mice. The data, presented at the 2026 AACR meeting, could unlock more effective LNP‑based cancer treatments.

Contributor: Focus on the Real Causes of the Shortage in Hormone Treatments
U.S. pharmacies have struggled to fill estradiol patch prescriptions, prompting media narratives that blame a surge in menopause hormone demand. In reality, usage of hormone therapy has hovered around 5% of menopausal women, far from unprecedented levels. The shortage is...

GLP-1 May Only Be the Beginning, Not the End of the Story
Researchers led by Richard DiMarchi and Matthias Tschöp published a paper in Molecular Metabolism showing that triple agonist retatrutide can drive weight loss even when GLP‑1 signaling is blocked. Their preclinical work demonstrates that co‑activating GIP and glucagon receptors produces...

Freudenberg Medical Launches ISO Class 5 Cleanroom for Biopharma
Freudenberg Medical announced CleanAssure, an ISO 5 controlled cleanroom that delivers washed, dried and gamma‑sterilized single‑use assemblies for biopharma customers. The service expands the company’s portfolio beyond component manufacturing to include validated cleaning and sterilization under cGMP conditions. By integrating this...

Magazine: Digital Twins Become the New Battleground for CDMOs
The latest issue of Pharmaceutical Technology highlights how knowledge‑graph digital twins are becoming a strategic weapon for contract development and manufacturing organisations (CDMOs) tackling complex biologics, gene therapies and advanced products. By creating a virtual replica of the entire manufacturing...

Georgetown Engineers Pectin-Based Bone Grafts to Replace Metal Implants
Georgetown University researchers have engineered a 3D‑printed bone graft that combines pectin—a food‑grade polysaccharide—with hydroxyapatite layers to mimic natural bone architecture. The pectin matrix can be printed at room temperature, creating a porous scaffold that promotes nutrient flow and cell...

The Future of Quality in CDMOs: The Five-Stage Journey to an Advanced Pharmaceutical Quality System (PQS)
Sharp, a leading contract development and manufacturing organization (CDMO), outlined a five‑stage journey to an advanced Pharmaceutical Quality System (PQS) that shifts quality from basic compliance to a strategic, performance‑driven function. The roadmap progresses through foundational compliance, strategic direction, integrated...

Four Ways to Build a Secure and Scalable CGT Distribution Network
Cell and gene therapies (CGTs) are expanding rapidly, with 34 US products approved and a projected $80 bn global market by 2029, driven largely by oncology. Successful commercialization now hinges on building secure, scalable distribution networks that protect fragile, cryogenic products...

What Actually Drives Speed in Complex Drug Development Programs
Speed in complex drug development is less about pushing harder and more about early, cross‑functional coordination. Traditional sequential handoffs often create hidden delays, forcing teams to revisit decisions under pressure. Thermo Fisher Scientific’s Accelerator™ Drug Development model integrates CDMO and...

Potential Spillover Effects on Diagnostic Delay for Cancer During the NHS-Galleri Trial
A quasi‑experimental study examined whether England’s NHS‑Galleri trial, which tests a cell‑free DNA multi‑cancer early detection (MCED) assay, caused spillover effects on cancer diagnostic timelines. Using a difference‑in‑differences design across 21 cancer‑alliance regions, researchers found that participating regions experienced a...

Pulnovo Raises $100M Led by Medtronic to Advance PADN System for Pulmonary Hypertension
Pulnovo Medical announced an oversubscribed $100 million financing round led by Medtronic, bringing together existing backers such as EQT, Qiming Venture Partners, Gaorong Ventures, OrbiMed and Lilly Asia Ventures. The capital will accelerate clinical development, regulatory work and commercialization of Pulnovo’s...
STAT+: PBMs Warn Trump’s Proposal to Disclose Drug Prices Is Illegal
The U.S. Department of Labor has issued a proposed rule that would require pharmacy benefit managers (PBMs) to disclose detailed drug‑pricing information to employers and simplify audit processes. The rule, released in January, has sparked a flood of comments, with...
Fully Defined 3D Culture Substrate for Cancer Research
AMSBIO announced that its fully defined MatriMix 511 extracellular matrix enables patient‑derived colorectal cancer cells to form robust 3D organoids. In a Kyoto University study, the organoids preserved stage‑specific tumor biology and expressed metastatic markers, outperforming alternative matrices. MatriMix’s composition...

AlphaGen Therapeutics to Present Preclinical Studies of Two Next-Generation Alpha Therapies at AACR 2026
AlphaGen Therapeutics announced it will present preclinical data on two next‑generation alpha radiopharmaceuticals, AG1002 and AG1206, at the AAC 2026 meeting in San Diego. AG1002 is a non‑agonist SSTR2‑targeting agent that achieved a superior tumor‑to‑kidney ratio and robust tumor inhibition in multiple...

EpiBiologics Reports First Patient Dosed in P-I Study of EPI-326 for EGFR-Driven Solid Tumors
EpiBiologics has dosed the first patient in a global Phase‑1 trial of EPI‑326, a tissue‑selective bispecific antibody that targets EGFR in advanced solid tumors. The study will assess safety, tolerability, pharmacokinetics and early anti‑tumor activity in non‑small cell lung cancer...

From Product to Patient in Nuclear Medicine: Why Vertical Integration Is Essential for a Competitive Advantage
Nuclear medicine’s ultra‑short radiopharmaceutical half‑lives make delivery inseparable from production, forcing a single, time‑bound operational system. Curium has built a globally integrated model that combines isotope manufacturing, quality release, and distribution into one coordinated network. The approach proved its resilience...

Using AI to Advance Validated Real-World Evidence
The HealthVerity event highlighted how AI can turn early‑stage life‑science questions into reproducible real‑world evidence (RWE) within minutes. Speakers from HealthVerity, Chiesi USA, Argenx and Medeloop shared practical methods for building transparent, iterative analyses that support feasibility, patient‑journey, regulatory and...

Emyria Launches Empax Global Partnership Platform Targeting International Drug Sponsors
Emyria (ASX:EMD) has launched the Empax Global Partnership Platform, giving international drug sponsors and CROs direct access to its clinical delivery infrastructure for complex psychedelic‑assisted therapies. The service creates a dual‑revenue model that blends existing reimbursed treatment programs with high‑margin,...

Trump Cuts Crucial Health Research Funding, Fueling Crisis
This piece in the WaPo summarizes what I've been say. The feds under Trump are choking off research dollars -- particularly for women's health, cancer and mental health. Young researchers are particularly hard hit, steering scientists into other work. It's...

GSK's Blenrep Approved in China for Multiple Myeloma
#GSK Blenrep (belantamab mafodotin) approved in China with a Bortezomib and Dexamethasone combo for treating Adults with a form of multiple myeloma.

FDA Seeks More Cardiovascular Data on Eli Lilly’s Orforglipron After Safety Concerns
The U.S. FDA has asked Eli Lilly to provide further cardiovascular safety data on its newly approved oral GLP‑1 weight‑loss drug Orforglipron, after the agency’s approval letter flagged “unexpected serious risks.” Lilly says patient safety remains its top priority as it...

AI-Driven Drug Discovery: Current Landscape and Future Promise
Cool review and perspective on the current state and the future of AI-powered drug discovery https://t.co/AgwqOlt4RU

Alembic Pharma Secures US FDA Nod for Generic Methotrexate Injection, Boosting US Generics Portfolio
Alembic Pharmaceuticals announced US FDA approval for its generic methotrexate injection USP on April 16, 2026. The clearance covers 50 mg/2 mL multi‑dose and 1 g/40 mL single‑dose vials, bringing the Indian drugmaker’s total US ANDA approvals to 236. The move strengthens Alembic’s foothold...

Gene‑Therapy Pioneers Win $3 Million Breakthrough Prize for Vision and Sickle‑Cell Treatments
The Breakthrough Prize Foundation awarded a $3 million Life Sciences prize to three University of Pennsylvania scientists and two collaborators for pioneering gene‑therapy treatments that restore sight in Leber congenital amaurosis and target sickle‑cell disease. The honor spotlights Luxturna’s market entry...

FDA Approves Breakthrough Therapy for Critically Ill Children
FDA says ‘yes’ to transformative therapy for sick children — and more media coverage of UCLA https://t.co/N6AJsRwCyX

Eli Lilly to Spend $6.3 B on Centessa Buy, with up to $1.5 B in Milestone Payouts
Eli Lilly agreed to acquire UK‑based Centessa Pharmaceuticals for an upfront $6.3 billion, plus a contingent value right that could add $1.5 billion if the lead sleep‑wake disorder drug meets regulatory milestones. The deal, priced at a 40% premium, follows Lilly’s cash‑rich...

NTU Unveils AI‑Powered Biochip That Detects Disease Biomarkers in 20 Minutes
Scientists at Singapore’s Nanyang Technological University have built an AI‑driven nanophotonic biochip that can identify disease‑related microRNA biomarkers in roughly 20 minutes. The prototype pairs a camera and mobile app for real‑time analysis, aiming to cut diagnostic turnaround times dramatically.
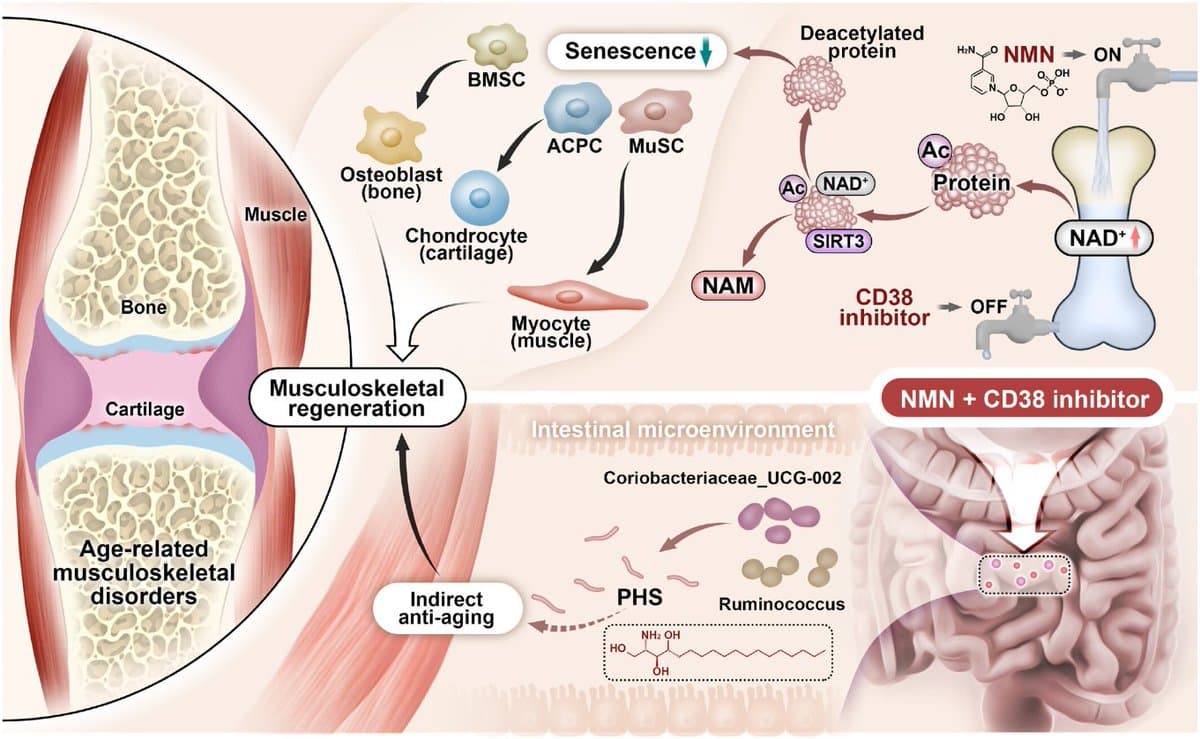
Combined NMN and CD38 Inhibitor Rejuvenates Aging Muscles
Double-Pronged NAD Preservation: Delaying Cellular Senescence and Initiating Musculoskeletal Regeneration https://t.co/bnkkn0xblj Scheme 1: A novel synergistic drug combination (N + A) consisting of an NAD+ precursor (NMN) and an NAD+ consumption (CD38) inhibitor (API) promotes musculoskeletal regeneration in aging.

FDA Grants Breakthrough Therapy Designation to Plixorafenib for Rare CNS Tumors
The U.S. FDA has granted Breakthrough Therapy Designation to plixorafenib, an investigational BRAF inhibitor for adults with rare, high‑grade gliomas harboring the BRAF V600E mutation. Early‑stage data showed a 67% overall response rate, positioning the drug as a potential game‑changer...

AI-Optimized Molecules Drive Clinical Interest and Expansion
#AACR2026 is in full swing and our posters are very popular. And some pharma companies realized that we have clinical data now and the molecules were AI-optimized to avoid the common liabilities. Finally, some genuine interest and we will have...

Unexpected Insight Among 11 Lessons From AACR26
11 things I learned at #AACR26 Industry Partnering event – number 7 may well surprise you: https://t.co/T0ePbaIU1K https://t.co/CMh15HfnnG

Here’s Simulations Plus Inc (SLP)’s Vision for AI in Drug Development
Simulations Plus Inc (NASDAQ:SLP) announced on March 26 that three major pharmaceutical firms have joined its AI‑enabled modeling collaboration programs, embedding the company’s AI agents directly into their drug‑development workflows. The firm emphasized a fully integrated AI deployment rather than...
Why Recursion Pharmaceuticals Inc (RXRX) Expanded Citeline Partnership
Recursion Pharmaceuticals announced an expanded partnership with Citeline on March 31, adding the data‑provider’s real‑world evidence capabilities to its AI‑driven drug discovery platform. The integration is designed to improve clinical‑trial design and accelerate decision‑making for its pipeline of cancer and...

Here’s What TD Cowen Thinks About Tempus AI Inc (TEM) Stock
TD Cowen upgraded Tempus AI Inc (NASDAQ:TEM) to a Buy rating, trimming its price target to $65 from $70. The company reported an 83.4% revenue jump to $1.3 billion in 2025 and projects $1.59 billion for 2026, a 25% increase. Strategic partnerships...

Leaked Images Show Monkeys Forced Into Weight‑Loss Drug Tests in UK Labs
A former laboratory technician leaked graphic footage of long‑tailed macaques being restrained and fed experimental anti‑obesity drugs via oral gavage in two UK facilities. The images, obtained by The Mail on Sunday, have ignited calls for faster phasing out of...

Cubical Bipyramid Nanoparticles Enable Systemic Magnetic Hyperthermia for Ovarian Cancer
Researchers at Oregon State University have created a cubical bipyramid magnetic nanoparticle that can be delivered intravenously and heated to 42‑46 °C under an alternating magnetic field, overcoming the need for direct tumor injection. The breakthrough, published in Advanced Functional Materials,...

FDA Expands Vertex's Alyftrek, Trikafta Labels to Reach 95% of U.S. Cystic Fibrosis Patients
The U.S. Food and Drug Administration approved label extensions for Vertex Pharmaceuticals' CF drugs Alyftrek and Trikafta, now covering roughly 95% of cystic fibrosis patients in the United States. The expansion adds eligibility for about 800 more patients, based on...

Novel Diabetic Wound Treatment Turns Cells Into Manufacturers
Researchers at Texas A&M have created a novel wound dressing for diabetic foot ulcers that leverages an interwoven extracellular matrix produced by human cells, then strips the cells away, leaving a purely biological scaffold. The approach sidesteps the immune‑rejection and...

Rogan Tipped the Scales on Psychedelic Research in the US
Joe Rogan appeared at the White House as President Trump signed an executive order to fast‑track psychedelic research and clinical trials. The directive directs federal agencies to accelerate approval pathways for psychedelic therapeutics aimed at PTSD, veteran mental health, and...

StockWatch: Revolution’s Phase III Pancreatic Cancer Data Dazzles Investors, Analysts
Revolution Medicines reported Phase III RASolute 302 results showing its oral RAS inhibitor daraxonrasib extended median overall survival to 13.2 months in previously treated metastatic pancreatic ductal adenocarcinoma, versus 6.7 months for standard chemotherapy (HR 0.40, p < 0.0001). The data sparked a...

What to Know Before Buying GLP-1 Drugs Online
The FDA warns that many online ads for GLP‑1 weight‑loss drugs promote compounded versions that lack the rigorous testing of brand‑name products like Ozempic, Wegovy, and Mounjaro. These compounded formulations can vary in concentration, contain unapproved ingredients, and have been...

Searches up 900%: Google Faces Influx of Illegal Peptide Ads
Google is facing a surge in advertisements for illegal synthetic peptides, with searches for terms like BPC‑157, GHK‑CU and Retatrutide jumping up to 900% over the past year. The ads violate Google’s own policies and Australian law, prompting the company...

Can a Common Parasite Medication Calm the Brain’s Stress Circuitry During Alcohol Withdrawal?
Researchers at UC San Diego discovered that rodents with high P2rx4 gene expression exhibit markedly increased alcohol consumption during withdrawal. Administering the antiparasitic drug ivermectin produced a dose‑dependent reduction in lever‑pressing for alcohol, especially in animals that responded behaviorally. Electrophysiological...