Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech rounds
Spanish biotech firm Ona announced an $86.6 million Series B financing, ranking among the largest venture rounds in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funding will be used to scale Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical programs.

Eli Lilly’s Mounjaro Gains 10% in India, Defying Generic Competition
Eli Lilly’s diabetes drug Mounjaro grew 10% in India even after a cheaper generic entered the market, boosting its international GLP‑1 share to roughly 53%. Morgan Stanley says the drug’s overseas revenue could outpace consensus, highlighting a broader shift toward obesity treatment.

GT Biopharma Posts $2.8M Q1 Loss, Launches GTB-3650 Phase 1 Leukemia Trial
GT Biopharma announced a net loss of $2.83 million for the first quarter and disclosed that its GTB-3650 program has entered a Phase 1 trial for leukemia. The company ended the quarter with $8.9 million in cash, underscoring a tighter cost structure as...

Citius Oncology Posts $5.6M LYMPHIR Revenue, Secures $41.5M Financing
Citius Oncology reported $5.6 million net revenue from the LYMPHIR launch in the first half of fiscal 2026 and closed a $5 million equity offering plus up to $36.5 million in debt and equity financing. The company says the capital will fund a...

Why J&J Thinks Its New Psoriasis Pill Could Be One of Its Biggest Drugs Ever
Johnson & Johnson’s newly approved oral psoriasis pill Icotyde targets the IL‑23 receptor, making it the first once‑daily tablet in a class dominated by injectables. The drug entered the market in March and logged roughly 1,500 prescriptions within its first...
Dr. Reddy’s Laboratories Launches Its Generic Semaglutide Injection in Canada
Dr. Reddy’s Laboratories received Health Canada’s Notice of Compliance on April 28, 2026 and launched the first generic semaglutide injection in Canada, the inaugural G7 approval for a generic version of the GLP‑1 drug. The product is offered in 2 mg and 4 mg...

Researchers Identify First Suite of Human Antibodies Against Measles Virus
NIH‑funded researchers have isolated and structurally mapped the first comprehensive set of human monoclonal antibodies against measles virus, revealing nine distinct epitopes on the H and F surface proteins. One antibody, 4F09, locked the fusion protein and cleared virus from...
ENHERTU® (Fam-Trastuzumab Deruxtecan-Nxki) Approved in the US for Two New Indications for Patients with HER2-Positive Early Breast Cancer
The FDA has approved AstraZeneca and Daiichi Sankyo’s ENHERTU (fam‑trastuzumab deruxtecan‑nxki) for both neoadjuvant and adjuvant treatment of HER2‑positive early‑stage breast cancer. In the neoadjuvant setting, ENHERTU plus taxane, trastuzumab and pertuzumab achieved a 67.3% pathologic complete response, outperforming standard...

FDA Places Full Hold on Aardvark’s Prader‑Willi Trial; Company to Unblind Data
The FDA has imposed a full clinical hold on Aardvark Therapeutics’ ARD-101 program for Prader‑Willi syndrome. The company will unblind data from its Phase 3 HERO trial and open‑label extension to evaluate safety and efficacy, while maintaining a cash runway of...

FDA Allows De‑Identified Real‑World Evidence in Device Submissions, Opening Path for Drug Developers
The U.S. Food and Drug Administration issued new guidance in December 2025 that permits sponsors to submit de‑identified real‑world evidence for medical device approvals. The change removes a long‑standing data‑access barrier and signals a possible shift toward broader use of...

Protara Therapeutics Shows 55% 12‑Month Complete Response in BCG‑Naïve NMIBC Phase 2 Trial
Protara Therapeutics disclosed that its intravesical cell therapy TARA‑002 achieved a 55% complete response rate at 12 months in a fully enrolled BCG‑naïve cohort of the Phase 2 ADVANCED‑2 trial. The data, presented at the AUA 2026 meeting, underscore durable efficacy...

United Therapeutics Corporation Announces FDA Clearance to Proceed with UHeart Xenotransplantation Clinical Trial
United Therapeutics received FDA clearance to begin the EXPRESS clinical trial of its UHeart xenotransplant, a pig‑derived heart with ten gene edits. The phase‑1/2/3 study will initially enroll up to two end‑stage heart‑failure patients, with safety and efficacy data reviewed...

Isomorphic Labs Secures $2.1 B Series B to Accelerate AI Drug Discovery
Isomorphic Labs, the London‑based AI biotech founded by DeepMind’s Demis Hassabis, closed a $2.1 billion Series B round led by Thrive Capital. The capital will expand its IsoDDE platform and integrate AlphaFold 3, underscoring growing investor belief that AI can reshape pharmaceutical...
FDA Grants First ctDNA-Guided Approval for Genentech's Tecentriq in Bladder Cancer
Genentech, part of Roche, secured FDA approval for Tecentriq (atezolizumab) and Tecentriq Hybreza as adjuvant treatments for muscle‑invasive bladder cancer patients with circulating tumor DNA residual disease. The decision, based on Phase III IMvigor011 data, shows a 36% drop in disease‑free...
![[Comment] Emerging Β-Lactam and Β-Lactamase Inhibitor Strategies for Complicated Urinary Tract Infections](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/9fed89640d11fbd33efb7e76f58daf8c.webp)
[Comment] Emerging Β-Lactam and Β-Lactamase Inhibitor Strategies for Complicated Urinary Tract Infections
Complicated urinary tract infections (cUTIs) and acute pyelonephritis remain leading causes of hospitalisation and antibiotic consumption worldwide. Rising rates of ESBL‑producing and carbapenem‑resistant Gram‑negative bacteria are eroding the efficacy of existing β‑lactam regimens. Recent phase‑3 data show that novel β‑lactam/β‑lactamase...
Agenus Announces Publication of Phase 1b Botensilimab and Balstilimab Data in Post-Immunotherapy Hepatocellular Carcinoma in Liver Cancer
Agenus published Phase 1b data on botensilimab plus balstilimab in 19 patients with treatment‑refractory hepatocellular carcinoma who had progressed after prior immunotherapy. The combination achieved a 17% objective response rate, a 50% clinical benefit rate at 18 weeks, median progression‑free survival...

FDA Approves Two Separate Indications for Fam-Trastuzumab Deruxtecan-Nxki in HER2-Positive Early-Stage Breast Cancer
The FDA has approved fam‑trastuzumab deruxtecan‑nxki (Enhertu) for two new indications in HER2‑positive early‑stage breast cancer: a neoadjuvant regimen followed by taxane, trastuzumab and pertuzumab, and an adjuvant regimen for patients with residual invasive disease. In the DESTINY‑Breast11 neoadjuvant trial,...

The BioPharm Brief: Oncology Momentum, CAR-T Advances, Strategic Expansion
AstraZeneca’s exploratory POTOMAC trial showed that combining its checkpoint inhibitor Imfinzi with BCG lowered early recurrence risk in patients with high‑risk non‑muscle‑invasive bladder cancer. At ASGCT 2026, Imviva presented early remission data from an allogeneic CAR‑T platform targeting lupus, hinting at...

TRI-611
TRI‑611 is a CNS‑penetrant, CRBN‑mediated molecular‑glue degrader targeting ALK fusion proteins in ALK‑positive non‑small cell lung cancer (NSCLC). By recruiting a non‑G‑loop degron distal to the orthosteric site, it degrades ALK independently of the active site, sidestepping common tyrosine‑kinase inhibitor...
Clinical Trial Failures of 2026
Clinical trial success rates remain low, hovering around 5% overall, with Phase II proving especially challenging. Industry‑sponsored Phase II and III terminations have roughly doubled from 2013 to 2023, often driven by commercial strategy shifts. In 2026, several high‑profile late‑stage...

Repeated Psilocybin Doses Show Safety, Promise for OCD
A randomized clinical trial of repeated doses of psilocybin for the treatment of obsessive–compulsive disorder 🍥"Administration of up to eight doses of psilocybin in a clinical research setting appears to be safe and potentially effective for patients with OCD. Larger trials...

Pfizer Secures EU Approval for HYMPAVZI Gene Therapy, Cuts Bleeds 93%
Pfizer has obtained European Commission marketing authorisation for HYMPAVZI (marstacimab) to treat hemophilia A or B patients with inhibitors aged 12 and older. The approval, covering all 27 EU members plus Iceland, Liechtenstein and Norway, is based on a Phase III...

METiS TechBio IPO Raises $270 M, Debuts as First Public AI‑Powered Drug Delivery Firm
METiS TechBio listed on the Hong Kong Stock Exchange, raising HK$2.11 billion ($269.5 million) and becoming the world’s first publicly listed AI‑powered drug‑delivery company. The offering was oversubscribed more than 6,900 times, propelling the stock 173% higher on debut and drawing a...
Antiviral Ensitrelvir Cuts Risk of COVID-19 in Household Contacts by Two-Thirds, Study Finds
A Phase III trial published in the New England Journal of Medicine shows that the oral antiviral ensitrelvir, given within 72 hours of an index case’s symptom onset, cuts the risk of symptomatic COVID‑19 in household contacts by roughly two‑thirds. The study...
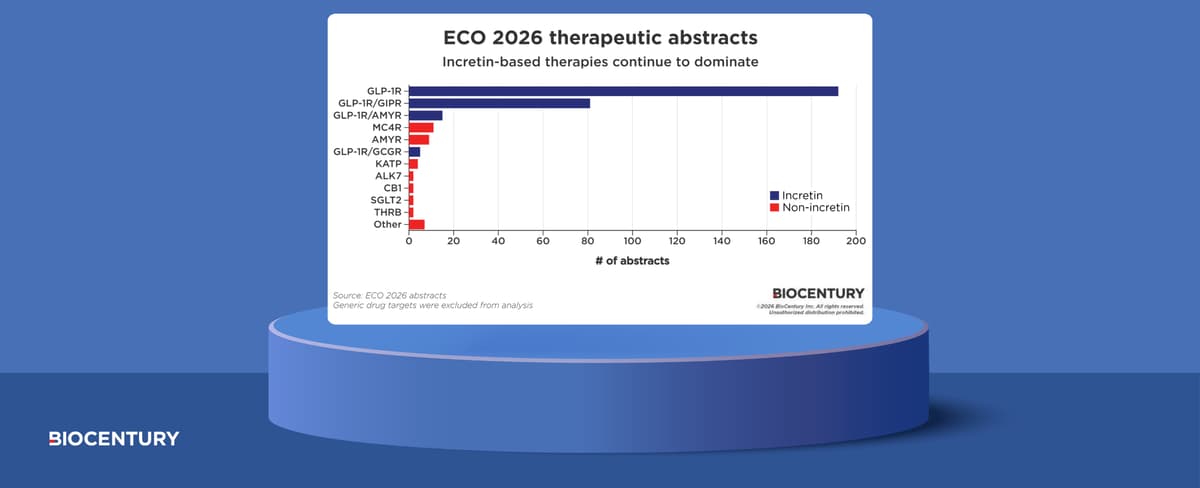
Incretins Continue to Dominate Obesity Conferences
Recent obesity conferences were dominated by incretin‑based therapies, with GLP‑1 receptor agonists such as semaglutide and the dual GIP/GLP‑1 agent tirzepatide taking center stage. New trial data presented showed weight‑loss results of 15‑20 percent, reinforcing the clinical potency of these...

Pharmaceutical Executive Daily: FDA Approves AstraZeneca's Fasenra
The FDA has approved AstraZeneca’s Fasenra (benralizumab) for patients 12 and older with hypereosinophilic syndrome (HES), adding a rare‑disease indication to a drug already used for severe eosinophilic asthma. The approval rests on the Phase III Natron trial, which showed a...
Novel CAR T Cell Therapy Moves Into Clinical Studies
The University of Texas MD Anderson Cancer Center and its joint venture CTMC received FDA clearance to proceed with an Investigational New Drug application for a novel CAR‑T cell therapy targeting CD94‑positive T/NK‑cell lymphomas. The therapy will enter a Phase 1...

Regenxbio's RGX-202 Hits Phase 3 Primary Endpoint, Eyes FDA Filing
Regenxbio announced that its RGX-202 gene therapy met the primary endpoint of its pivotal Phase 3 trial, with 28 of 30 patients achieving at least 10% normal dystrophin levels and an average expression of 71.1%. The company says the data...
ECO 2026: Indirect Comparison Favours Wegovy Pill over Foundayo
At the 33rd European Congress on Obesity, Novo Nordisk presented a post‑hoc indirect comparison of oral semaglutide (Wegovy pill) versus orforglipron (Foundayo). Using simulated treatment comparison and matching‑adjusted indirect methods, the analysis showed Wegovy achieved roughly 3 percentage‑points greater weight...
New mRNA Therapy Destroys Cancer by Improving T Cell Priming
MIT researchers engineered lipid‑nanoparticle mRNA encoding NF‑κB‑inducing kinase (NIK) or IRF8 to reprogram immature dendritic cells into the cDC1 phenotype. The immune‑remodeling mRNAs amplified CD8+ T‑cell priming, leading to complete tumor regression in 70‑80% of mice across colorectal and metastatic...

FDA CDER Acting Director Tracy Hoeg Set to Leave
FDA Center for Drug Evaluation & Research (CDER) Acting Director Tracy Beth Hoeg expected to depart from agency - Reuters
Bacterial Energy Enzyme Reveals Dual-Trigger Sodium Pump Mechanism, Offering Antibiotic Clues
Researchers used modified AI tools and supercomputer‑scale molecular dynamics to capture the hidden motions of the bacterial sodium‑pumping enzyme Na⁺‑NQR. The simulations revealed a dual‑trigger mechanism where sodium binding and electron transfer drive conformational changes in subunits NqrD and NqrE,...

Psilocybin Offers Fast-Acting Alternative to Traditional Antidepressants
A phase‑2, double‑blind trial in Sweden found that a single 25 mg dose of psilocybin produced rapid antidepressant effects, cutting MADRS scores by an average of 9.7 points within eight days versus 2.4 points for an active placebo. The benefit persisted...

Translational Intelligence: The CDMO's Unspoken Role in the ATMP Journey
Advanced therapy medicinal product (ATMP) developers face a critical translation gap between research‑stage processes and GMP‑compliant manufacturing, where the process itself becomes the product. This gap, not a funding issue, creates structural risk that can halt programs and deter investors....

Candel Reports Prostate Cancer Drug's Long-Term Data Ahead of FDA Filing
Candel Therapeutics released long‑term follow‑up results for its investigational prostate cancer therapy, showing durable efficacy and a favorable safety profile. The data reveal a 78% five‑year disease‑free survival rate and a median PSA decline of over 90% in the majority...

Aardvark's Next Move After Clinical Hold; Alumis' Verdict on Acelyrin Asset
Aardvark Therapeutics announced that the FDA clinical hold on its rare‑disease HERO and OLE studies has been lifted, allowing the company to unblind the trial data. The unblinding will provide critical efficacy and safety readouts for the targeted condition. In...
Cogent Biosciences Targets Transformational 2026 with Bezuclastinib
Cogent Biosciences Inc. says its lead drug candidate Bezuclastinib will hit several regulatory and clinical checkpoints in 2026, positioning the firm for a pivotal year. The tyrosine‑kinase inhibitor targets the KIT D816V mutation that drives most gastrointestinal stromal tumors and...

CAR-T Therapy for Stiff Person Syndrome Nears Approval
Kyverna Therapeutics is close to securing regulatory approval for its CAR‑T cell therapy aimed at treating stiff person syndrome (SPS), a rare autoimmune neurological disorder with no approved drugs. The therapy uses engineered T cells to eliminate the B‑cell populations...

Resurge Therapeutics Presents Clinical Results on Its Intraprostatic Drug Elution (IPDE) Platform for Benign Prostatic Hyperplasia (BPH) at AUA 2026
Resurge Therapeutics presented data from two early‑phase studies of its intraprostatic drug elution (IPDE) platform for benign prostatic hyperplasia (BPH) at the AUA 2026 meeting. In the transperineal cohort of 20 patients, IPSS dropped 15 points and peak urinary flow...

#ASGCT26: A Zillow-Like Marketplace for Abandoned Gene Therapies Goes Live
Two nonprofit groups, the American Society of Gene & Cell Therapy and the Orphan Therapeutics Accelerator, unveiled a Zillow‑style online marketplace that lists abandoned cell and gene therapy assets. The platform, launched at the ASGCT26 conference, aggregates more than 30...

Arna Pharma and Slate Run Complete JV for New US Company
Arna Pharma and Slate Run Pharmaceuticals have completed a joint venture to create a U.S.-based specialty pharmaceutical company focused on branded products, 505(b)(2) medicines, and specialized generics. The partnership launched its first collaborative product, Aridol, and plans to add more...

NICE Backs Stomach Cancer, PAH Drugs for NHS Use
The UK’s health technology assessment body NICE has endorsed AstraZeneca’s immunotherapy Imfinzi for peri‑operative treatment of aggressive stomach and gastro‑oesophageal junction cancers, and approved Merck & Co.’s Winrevair as an add‑on therapy for pulmonary arterial hypertension (PAH). More than 1,500 patients in...
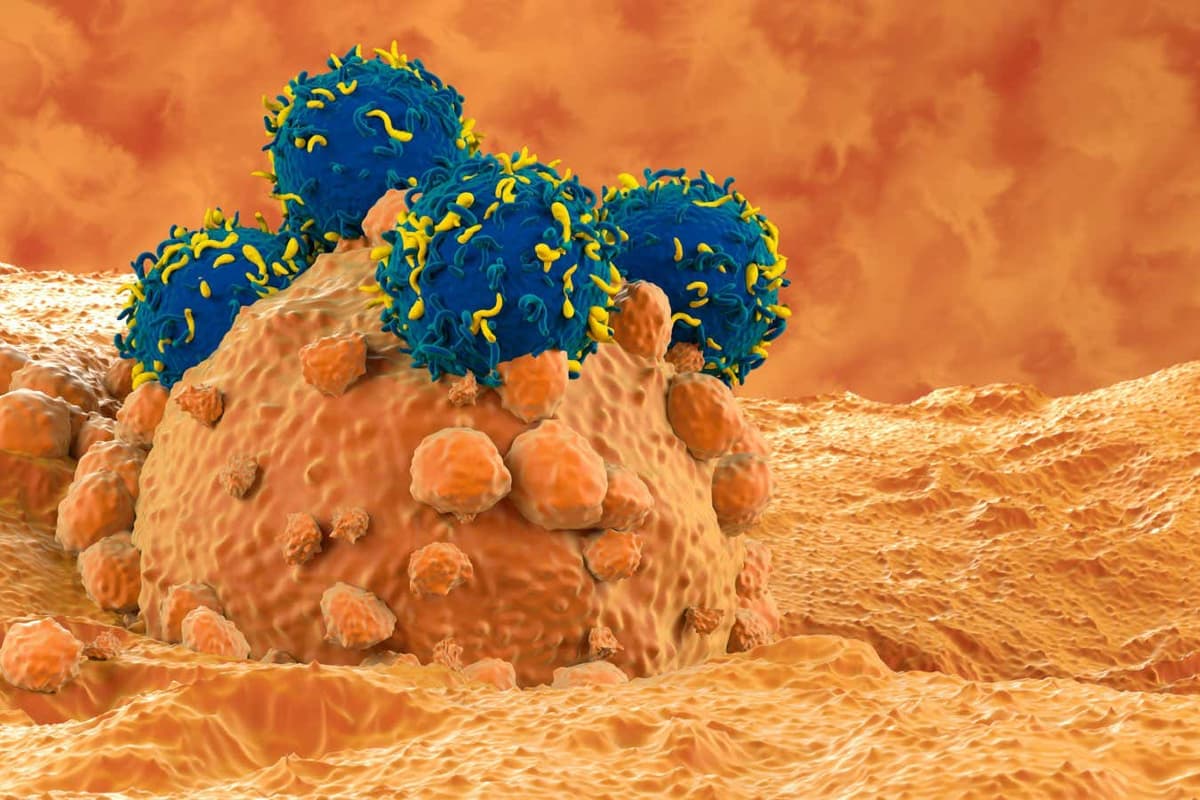
CAR T-Cell Therapy Bolstered by Stiffening up Cancer Cells First
Researchers have discovered that pre‑treating cancer cells to increase their stiffness markedly improves the effectiveness of CAR T‑cell therapy in mouse models of aggressive melanoma. The physical alteration of tumor mechanics enhances immune cell infiltration and tumor killing, offering a novel...

Kyowa Kirin Secures FDA Approval for Crysvita Dosing Update
Kyowa Kirin has secured FDA approval to expand Crysvita’s dosing regimen for adults with X‑linked hypophosphataemia (XLH). The update allows a 0.5 mg/kg dose up to 90 mg every two weeks, with the option to increase to 1 mg/kg after four weeks if serum...

Kyowa Kirin Secures FDA Approval for Crysvita Dosing Update
Kyowa Kirin has secured FDA approval for a dosing update to Crysvita (burosumab‑twza) for adults with X‑linked hypophosphataemia (XLH). The new regimen permits a 0.5 mg/kg dose every two weeks, with the option to increase to 1 mg/kg after four weeks if serum...

Biogen Reports P-II (CELIA) Trial Results on Diranersen in Early Alzheimer’s Disease
Biogen disclosed phase‑II (CELIA) results for intrathecal diranersen in 416 patients with early Alzheimer’s disease. The trial did not meet its primary dose‑response endpoint on CDR‑SB at week 76, but all dosing arms, especially 60 mg every‑four‑weeks, showed a slowing of clinical...

AstraZeneca’s Fasenra Receives the US FDA Approval for Hypereosinophilic Syndrome
The U.S. FDA has granted approval for AstraZeneca’s biologic Fasenra (benralizumab) to treat patients aged 12 and older with hypereosinophilic syndrome (HES) lacking a non‑hematologic secondary cause. Approval follows the Phase III NATRON trial, which enrolled 133 patients and compared 30 mg...

As New Rules Take Hold, What Does ‘Good AI Practice’ Look Like in Drug Development?
On January 14 2026 the EMA and FDA published joint guiding principles that define good AI practice for drug development. The technology‑neutral framework emphasizes context of use, risk‑based controls, data governance, documentation and lifecycle management rather than adding new regulatory requirements. It...

Racura Oncology Advances CPACS Trial to Next Dose Level
Racura Oncology (ASX:RAC) received Safety Review Committee clearance to double the RC220 dose from 40 mg/m² to 80 mg/m² for Cohort 2 of its CPAPS Phase 1 trial, pairing the drug with doxorubicin. The first three Cohort 1 patients showed no dose‑limiting toxicities, and all...

Predicting Alzheimers & Dementia (and Minimizing Risk)
A wave of new research links environmental and immunological factors to dementia risk. Epidemiological data show heat‑related illness raises long‑term dementia incidence, while regular sauna exposure appears to cut Alzheimer’s risk by roughly two‑thirds. Observational studies also suggest the recombinant...

Biogen Finds Alzheimer’s Path for Tau ASO Despite Mixed Data
Biogen reported Phase 2 data for its tau‑targeting antisense oligonucleotide (ASO) in Alzheimer’s disease, showing a roughly 30% reduction in cerebrospinal fluid tau levels. Cognitive endpoints, however, delivered only modest, statistically non‑significant improvements versus placebo. The safety profile remained clean,...